Abstract
Background/Objectives:
This study investigates age-dependent changes in different components of energy expenditure (EE) within the longitudinal study on nutrition and health status in an aging population in Giessen, Germany (GISELA).
Subjects/Methods:
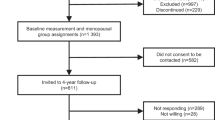
Between 1994 and 2006, data obtained at a total of 3033 visits from 363 women and 153 men with a mean initial age of 67.4±5.9 and 66.9±5.2 years, respectively, were evaluated. The mean duration of follow-up was 8 years. Resting metabolic rate (RMR) was assessed by indirect calorimetry and physical activity patterns were assessed by questionnaire. EE of physical activity and total EE (TEE) were calculated using multipliers for RMR. Energy intake was determined through a validated 3-day estimated dietary record. Linear mixed models were used to analyze the influence of age on EE adjusted for covariates.
Results:
Resting metabolic rate decreased in women and men by 158 and 326 kJ/d per decade, respectively; after considering changes in body composition and fat distribution, respective decreases were 81 and 286 kJ/d per decade. EE of physical activity decreased similarly in both sexes (472 kJ/d per decade). TEE dropped in women and men by 540 and 823 kJ/d per decade, respectively. No statistically significant changes in energy intake and body weight were observed in the course of follow-up.
Conclusions:
The age-dependent decrease in TEE is mainly due to a decrease in physical activity. The stable energy intake and body weight of the GISELA subjects may be indicators for a relatively good health status.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bender R, Lange S (2001). Adjusting for multiple testing—when and how? J Clin Epidemiol 54, 343–349.
Bergmann U, Wiholm BE (1981). Patient medication on admission to a medical clinic. Eur J Clin Pharmacol 20, 185–191.
Berke EM, Gardner AW, Goran MI, Poehlman ET (1992). Resting metabolic rate and the influence of the pretesting environment. Am J Clin Nutr 55, 626–629.
Black AE, Coward WA, Cole TJ, Prentice AM (1996). Human energy expenditure in affluent societies: an analysis of 574 doubly-labelled water measurements. Eur J Clin Nutr 50, 72–92.
Blanc S, Schoeller DA, Bauer D, Danielson ME, Tylavsky F, Simonsick EM et al. (2004). Energy requirements in the eighth decade of life. Am J Clin Nutr 79, 303–310.
Bosy-Westphal A, Eichhorn C, Kutzner D, Illner K, Heller M, Müller MJ (2003). The age-related decline in resting energy expenditure in humans is due to the loss of fat-free mass and to alterations in its metabolically active components. J Nutr 133, 2356–2362.
Cnaan A, Laird NM, Slasor P (1997). Tutorial in biostatistics: using the general linear mixed model to analyse unbalanced repeated measures and longitudinal data. Stat Med 16, 2349–2380.
Conley KE, Jubrias SA, Esselman PC (2000). Oxidative capacity and ageing in human muscle. J Physiol 526, 203–210.
Conway JM, Seale JL, Jacobs DR, Irwin ML, Ainsworth BE (2002). Comparison of energy expenditure estimates from doubly labelled water, a physical activity questionnaire, and physical activity records. Am J Clin Nutr 75, 519–525.
Elia M, Ritz P, Stubbs RJ (2000). Total energy expenditure in the elderly. Eur J Clin Nutr 54 (Suppl, 3), S92–S103.
Gallagher D, Allen A, Wang Z, Heymsfield SB, Krasnow N (2000). Smaller organ tissue mass in the elderly fails to explain lower resting metabolic rate. Ann N Y Acad Sci 904, 449–455.
Gallagher D, Belmonte D, Deurenberg P, Wang Z, Krasnow N, Pi-Sunyer FX et al. (1998). Organ-tissue mass measurement allows modeling of REE and metabolically active tissue mass. Am J Physiol 275, E249–E258.
Grimes DA, Schulz KF (2002). Descriptive studies: what they can and cannot do. Lancet 359, 145–149.
Guo SS, Zeller C, Chumlea WC, Siervogel RM (1999). Aging, body composition, and lifestyle: the Fels Longitudinal Study. Am J Clin Nutr 70, 405–411.
Hallfrisch J, Muller D, Drinkwater D, Tobin J, Andres R (1990). Continuing diet trends in men: the Baltimore Longitudinal Study of Aging. J Gerontol 45, M186–M191.
Heinsimer JA, Lefkowitz RJ (1986). The impact of aging on adrenergic receptor function: clinical and biochemical aspects. J Am Geriatr Soc 33, 184–188.
Hughes VA, Frontera WR, Roubenoff R, Evans WJ, Singh MAF (2002). Longitudinal changes in body composition in older men and women: role of body weight change and physical activity. Am J Clin Nutr 76, 473–481.
Hughes VA, Roubenoff R, Wood M, Frontera WR, Evans WJ, Singh MAF (2004). Anthropometric assessment of 10-y changes in body composition in the elderly. Am J Clin Nutr 80, 475–482.
Hunter GR, Weinsier RL, Gower BA, Wetzstein C (2001). Age-related decrease in resting energy expenditure in sedentary white women: effects of regional differences in lean and fat mass. Am J Clin Nutr 73, 333–337.
Institute of Medicine (2002). Dietary Reference Intakes: Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids. National Academy Press: Washington, DC. (Internet: http://www.nap.edu/books/0309085373/html).
Jungjohann SM, Lührmann PM, Bender R, Blettner M, Neuhäuser-Berthold M (2005). Eight-year trends in food, energy and macronutrient intake in an elderly German population. Br J Nutr 93, 361–378.
Keys A, Taylor HL, Grande F (1973). Basal metabolism and age of adult man. Metabolism 22, 579–587.
Klausen B, Toubro S, Astrup A (1997). Age and sex effects on energy expenditure. Am J Clin Nutr 65, 895–907.
Krems C, Lührmann PM, Neuhäuser-Berthold M (2004). Physical activity in young and elderly subjects. J Sports Med Phys Fitness 44, 71–76.
Krems C, Lührmann PM, Straßburg A, Hartmann B, Neuhäuser-Berthold M (2005). Lower resting metabolic rate in the elderly may not be entirely due to changes in body composition. Eur J Clin Nutr 59, 255–262.
Kyle UG, Genton L, Karsegard L, Slosman DO, Pichard C (2001). Single prediction equation for bioelectrical impedance analysis in adults aged 20–94 years. Nutrition 17, 248–253.
Littell RC, Henry PR, Ammermann CB (1998). Statistical analysis of repeated measures data using SAS procedures. J Anim Sci 76, 1216–1231.
Lührmann PM, Herbert BM, Gaster C, Neuhäuser-Berthold M (1999). Validation of a self-administered 3-day estimated dietary record for use in the elderly. Eur J Nutr 38, 235–240.
Lührmann PM, Herbert BM, Neuhäuser-Berthold M (2001a). Effects of fat mass and body fat distribution on resting metabolic rate in the elderly. Metabolism 50, 972–975.
Lührmann PM, Herbert BM, Neuhäuser-Berthold M (2001b). Underreporting of energy intake in an elderly German population. Nutrition 17, 912–916.
Mensink GBM (2002). Beiträge zur Gesundheitsberichterstattung des Bundes. Was essen wir heute? Ernährungsverhalten in Deutschland. Robert-Koch-Institut: Berlin.
Moreiras O, Van Staveren WA, Amorim Cruz JA, Carbajal A, De Henauw S, Grunenberger F et al. (1996). Longitudinal changes in the intake of energy and macronutrients of elderly Europeans. Eur J Clin Nutr 50 (Suppl, 2), S67–S76.
Müller MJ, Bosy-Westphal A, Klaus S, Kreymann G, Lührmann M, Neuhäuser-Berthold M et al. (2004). WHO equations have shortcomings to predict resting energy expenditure in individuals from a modern affluent population—comparison with an actual German data base of resting energy expenditure. Am J Clin Nutr 80, 1379–1390.
Murray LA, Reilly JJ, Choudhry M, Durnin JVGA (1996). A longitudinal study of changes in body composition and basal metabolism in physically active elderly men. Eur J Appl Physiol 72, 215–218.
Nicolas AS, Faisant C, Lanzmann-Petithory D, Tome D, Vellas B (2000). The nutritional intake of a free-living healthy French population: a four-year follow-up. J Nutr Health Aging 4, 77–80.
Poehlman ET, Toth MJ, Webb GD (1993). Sodium-potassium pump activity contributes to the age-related decline in resting metabolic rate. J Clin Endocrinol Metab 76, 1054–1057.
Rothenberg EM, Bosaeus IG, Steen BC (2003). Energy expenditure at age 73 and 78—a five year follow-up. Acta Diabetol 40 (Suppl, 1), S134–S138.
SAS (1999). SAS/STAT® User's Guide, Version 8. SAS Institute Inc: Cary, NC.
Schiffman SS, Zevakis J (2002). Taste and smell perception in the elderly: effect of medications and disease. Adv Food Nutr Res 44, 247–346.
Sjögren A, Österberg T, Steen B (1994). Intake of energy, nutrients and food items in a ten-year cohort comparison and in a six-year longitudinal perspective: a population study of 70- and 76-year-old Swedish people. Age Ageing 23, 108–112.
Speakman JR (1998). The history and theory of the doubly labeled water technique. Am J Clin Nutr 68, 932S–938S.
Tooze JA, Schoeller DA, Subar AF, Kipnis V, Schatzkin A, Troiano RP (2007). Total daily energy expenditure among middle-aged men and women: the OPEN Study. Am J Clin Nutr 86, 382–387.
Tzankoff SP, Norris AH (1978). Longitudinal changes in basal metabolism in man. J Appl Physiol 45, 536–539.
Van den Akker M, Buntinx F, Metsemakers JF, Roos S, Knottnerus JA (1998). Multimorbidity in general practice: prevalence, incidence, and determinants of co-occurring chronic and recurrent diseases. J Clin Epidemiol 51, 367–375.
Weir JB de V (1949). New methods for calculating metabolic rate with special reference to protein metabolism. J Physiol 109, 1–9.
Welle S, Thornton C, Totterman S, Forbes G (1996). Utility of creatinine excretion in body-composition studies of healthy men and women older than 60 y. Am J Clin Nutr 63, 151–156.
WHO (1985). Energy and Protein Requirements. Report of a Joint FAO/WHO/UNU Expert Consultation. WHO Technical Report Series, no. 724. WHO: Geneva.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lührmann, P., Bender, R., Edelmann-Schäfer, B. et al. Longitudinal changes in energy expenditure in an elderly German population: a 12-year follow-up. Eur J Clin Nutr 63, 986–992 (2009). https://doi.org/10.1038/ejcn.2009.1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2009.1
Keywords
This article is cited by
-
Do we need race-specific resting metabolic rate prediction equations?
Nutrition & Diabetes (2019)
-
Changes in Body Composition, Comorbidities, and Nutritional Status Associated with Lower Weight Loss After Bariatric Surgery in Older Subjects
Obesity Surgery (2019)
-
The impact of multimorbidity on resting metabolic rate in community-dwelling women over a ten-year period: A cross-sectional and longitudinal study
The Journal of nutrition, health and aging (2017)
-
Laparoscopic Roux‐En‐Y Gastric Bypass is Effective and Safe in Over 55‐year‐old Patients: A Comparative Analysis
World Journal of Surgery (2014)
-
Best-fitting prediction equations for basal metabolic rate: informing obesity interventions in diverse populations
International Journal of Obesity (2013)