Abstract
Background/Objectives:
Prebiotics have attracted interest for their ability to positively affect the colonic microbiota composition, thus increasing resistance to infection and diarrhoeal disease. This study assessed the effectiveness of a prebiotic galacto-oligosaccharide mixture (B-GOS) on the severity and/or incidence of travellers' diarrhoea (TD) in healthy subjects.
Subjects/Methods:
The study was a placebo-controlled, randomized, double blind of parallel design in 159 healthy volunteers, who travelled for minimum of 2 weeks to a country of low or high risk for TD. The investigational product was the B-GOS and the placebo was maltodextrin. Volunteers were randomized into groups with an equal probability of receiving either the prebiotic or placebo. The protocol comprised of a 1 week pre-holiday period recording bowel habit, while receiving intervention and the holiday period. Bowel habit included the number of bowel movements and average consistency of the stools as well as occurrence of abdominal discomfort, flatulence, bloating or vomiting. A clinical report was completed in the case of diarrhoeal incidence. A post-study questionnaire was also completed by all subjects on their return.
Results:
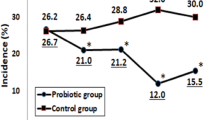
Results showed significant differences between the B-GOS and the placebo group in the incidence (P<0.05) and duration (P<0.05) of TD. Similar findings occurred on abdominal pain (P<0.05) and the overall quality of life assessment (P<0.05).
Conclusions:
Consumption of the tested galacto-oligosaccharide mixture showed significant potential in preventing the incidence and symptoms of TD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Black RE (1990). Epidemiology of travellers' diarrhea and relative importance of various pathogens. Rev Infect Dis 12, S73–S79.
Buddington KK, Danohoo JB, Buddington RK (2002). Dietary oligofructose and inulin protect mice from enteric and systemic pathogens and tumour inducers. J Nutr 132, 472–477.
Center for Disease Control and Prevention (CDC) (2008). Prevention of specific infectious diseases. In: CDC Health Information for International Travel. Atlanta, GA.
Cremonini F, Di Caro S, Nista EC (2002). Meta-analysis: the effect of probiotic administration on antibiotic associated diarrhea. Aliment Pharmacol Ther 16, 1461–1467.
Cummings JH, Christie S, Cole JT (2001). A study of fructooligosacharides in the prevention of travellers' diarrhea. Aliment Pharmacol Ther 15, 1139–1145.
Depeint F, Tzortzis G, Vulevic J, I'anson K, Gibson GR (2008). Prebiotic evaluation of a novel galacto-oligosaccharide mixture produced by the enzymatic activity of Bifidobacterium bifidum NCIM 41171, in healthy humans. Am J Clin Nutr 87 (Suppl, 3), S785–S791.
DuPont HL (2001). Treatment of travellers' diarrhea. J Travel Med 8, S31–S33.
DuPont HL, Capsuto EG (1996). Persistent diarrhea in travellers. Clin Infect Dis 22, 124–128.
Gibson GR, McCartney AL, Rastall RA (2005). Prebiotics and resistance to gastrointestinal infections. Br J Nutr 93, S31–S34.
Gibson GR, Roberfroid MB (1995). Dietary modulation of the human colonic microbiota: introducing the concept of prebiotics. J Nutr 125, 1401–1412.
Huang JS, Bousvaros A, Lee JW, Diaz A, Davidson EJ (2002). Efficacy of probiotic use in acute diarrhea in children: a meta-analysis. Dig Dis Sci 47, 2625–2634.
Kamat D, Mathur A (2006). Prevention and management of travellers' diarrhea. Travel Med 52 (Suppl, 7), 289–302.
Kunz C, Rudloff S, Baier W, Klein N, Strobel S (2000). Oligosaccharides in human milk: structural functional, and metabolic aspects. Ann Rev Nutr 20, 699–722.
Paredes P, Campbell-Forrester S, Mathewson JJ, Ashley D, Thompson S, Steffen R et al. (2000). Etiology of travellers' diarrhea on a Caribbean Island. J Travel Med 7, 15–18.
Peltola H, Gorbach SL (2001). Travellers' diarrhea epidemiology and clinical aspects. In: Dupont H, Steffen R (eds). Textbook of Travel Medicine and Health. BC Decker Inc, Hamilton: Canada. pp 151–159.
Saavedra JM, Bauman NA, Oung I, Perman JA, Yolken RH (1994). Feeding of Bifidobacterium bifidum and Streptococcus thermophilus to infants in hospital for prevention of diarrhea and shedding of rotavirus. Lancet 344, 1046–1049.
Searle LEJ, Best A, Nunez A, Salguero FJ, Johnson L, Weyer U et al. (2009). A galacto-oligosaccharide mixture, produced by the enzymatic activity of Bifidobacterium bifidum NCIMB 41171, reduces Salmonella Typhimurium infection in mice. J Med Microb 58, 37–48.
Silk DBA, Davis A, Vulevic J, Tzortzis G, Gibson GR (2009). Clinical trial: the effect of a trans-galactooligosacharide prebiotic on faecal microbiota and symptoms in irritable bowel syndrome. Aliment Pharmacol Ther 29, 508–518.
Steffen R, Van Der Linde F, Gyr K, Schär M (1983). Epidemiology of diarrhea in travellers. JAMA 249, 1176–1180.
Tojo M, Oikawa T, Morikawa Y, Yamashita N, Iwata S, Satoh Y (1987). The effects of Bifidobacterium breve administration on Campylobacter enteritis. Acta Pediatr Jpn 29, 160–167.
Tzortzis G, Goulas AK, Gee JM, Gibson GR (2005). A novel galactooligosaccharide mixture increases the bifidobacterial population numbers in a continuous in vitro fermentation system and in the proximal colon of pigs in vivo. J Nutr 135, 1726–1731.
U.S. Food and Drug Administration (2008). Center for Food Safety and Applied Nutrition. Office of Food Additive Safety. Version 28 July, 2008. GRAS Notice No. CRN 000236. Internet: http://www.cfsan.fda.gov/∼rdb/opa-g236.html (accessed 06 November 2008).
Vila J, Vargas M, Ruiz J, Corachan M, Jimenez De Anta MT, Gascon J (2000). Quinolone resistance in enterotoxigenic Escherichia coli causing diarrhea in travellers to India in comparison with other geographical areas. Antimicrob Agents Chemother 44, 1731–1733.
Vulevic J, Drakoularakou A, Yaqoob P, Tzortzis G, Gibson GR (2008). Modulation of the fecal microflora profile and immune function by a novel trans-galactooligosacharide mixture (B-GOS) in healthy elderly volunteers. Am J Clin Nutr 88, 1438–1446.
World Health Organization (2004). WHOQOL Study Protocol. WHO 2004 MNH7PSF/93.9.
Acknowledgements
Financial support was offered through a grant from Clasado Ltd. All data were analysed by Dr Drakoularakou.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Drakoularakou, A., Tzortzis, G., Rastall, R. et al. A double-blind, placebo-controlled, randomized human study assessing the capacity of a novel galacto-oligosaccharide mixture in reducing travellers' diarrhoea. Eur J Clin Nutr 64, 146–152 (2010). https://doi.org/10.1038/ejcn.2009.120
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2009.120
Keywords
This article is cited by
-
Quantitative trait loci and genomic prediction for grain sugar and mineral concentrations of cowpea [Vigna unguiculata (L.) Walp.]
Scientific Reports (2024)
-
The current state and future direction of DoD gut microbiome research: a summary of the first DoD gut microbiome informational meeting
Standards in Genomic Sciences (2018)
-
ACG Clinical Guideline: Diagnosis, Treatment, and Prevention of Acute Diarrheal Infections in Adults
American Journal of Gastroenterology (2016)
-
Pathogen-induced secretory diarrhea and its prevention
European Journal of Clinical Microbiology & Infectious Diseases (2016)
-
The Traveling Microbiome
Current Infectious Disease Reports (2016)