Abstract
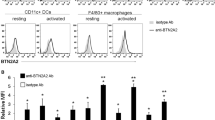
Tumor necrosis factor (TNF) and its receptors TNF receptor type 1 (TNFR1) and type 2 (TNFR2) have a central role in chronic inflammatory diseases. While TNFR1 mainly confers inflammation, activation of TNFR2 elicits not only pro-inflammatory but also anti-inflammatory effects. In this study, we wanted to investigate the anti-inflammatory therapeutic potential of selective activation of TNFR2 in mice with established collagen-induced arthritis. Mice with established arthritis induced by immunization with bovine collagen type II were treated with six injections of the TNFR2-specific agonist TNCscTNF80, given every second day. Two days after treatment cessation, the cell compositions of bone marrow, spleen and lymph nodes were analyzed. Mice were visually scored until day 30 after the start of therapy and the degree of joint inflammation was determined by histology. Treatment with TNCscTNF80 increased arthritis-induced myelopoiesis. Little effect was seen on the infiltration rate of inflammatory immature myeloid cells and on the reduction of lymphoid cells in secondary lymphoid organs. Upon treatment, frequency of regulatory T (Treg) cells in the CD4+ T-cell population was increased in both spleen and inguinal lymph nodes. In addition, the expression of TNFR2 on Treg cells was enhanced. The clinical score started to improve 1 week after cessation treatment and remained lower 30 days after initiation of therapy. The histological score also revealed amelioration of joint inflammation in TNCscTNF80-treated versus control mice. Activation of TNFR2 might provide a suitable therapeutic strategy in autoimmune arthritis by increasing the numbers of regulatory cell types, in particular Treg cells, and by attenuation of arthritis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Picerno V, Ferro F, Adinolfi A, Valentini E, Tani C, Alunno A. One year in review: the pathogenesis of rheumatoid arthritis. Clin Exp Rheumatol 2015; 33: 551–558.
Parks CG, D'Aloisio AA, DeRoo LA, Huiber K, Rider LG, Miller FW et al. Childhood socioeconomic factors and perinatal characteristics influence development of rheumatoid arthritis in adulthood. Ann Rheum Dis 2013; 72: 350–356.
Byng-Maddick R, Ehrenstein MR. The impact of biological therapy on regulatory T cells in rheumatoid arthritis. Rheumatology (Oxford) 2015; 54: 768–775.
Williams RO. Collagen-induced arthritis in mice: a major role for tumor necrosis factor-alpha. Methods Mol Biol 2007; 361: 265–284.
Monaco C, Nanchahal J, Taylor P, Feldmann M. Anti-TNF therapy: past, present and future. Int Immunol 2015; 27: 55–62.
Faustman D, Davis M. TNF receptor 2 pathway: drug target for autoimmune diseases. Nat Rev Drug Discov 2010; 9: 482–493.
Chen X, Baumel M, Mannel DN, Howard OM, Oppenheim JJ. Interaction of TNF with TNF receptor type 2 promotes expansion and function of mouse CD4+CD25+ T regulatory cells. J Immunol 2007; 179: 154–161.
Faustman DL, Davis M. TNF receptor 2 and disease: autoimmunity and regenerative medicine. Front Immunol 2013; 4: 478.
Chen X, Subleski JJ, Kopf H, Howard OM, Mannel DN, Oppenheim JJ. Cutting edge: expression of TNFR2 defines a maximally suppressive subset of mouse CD4+CD25+FoxP3+ T regulatory cells: applicability to tumor-infiltrating T regulatory cells. J Immunol 2008; 180: 6467–6471.
Chen X, Wu X, Zhou Q, Howard OM, Netea MG, Oppenheim JJ. TNFR2 is critical for the stabilization of the CD4+Foxp3+ regulatory T cell phenotype in the inflammatory environment. J Immunol 2013; 190: 1076–1084.
Okubo Y, Mera T, Wang L, Faustman DL. Homogeneous expansion of human T-regulatory cells via tumor necrosis factor receptor 2. Sci Rep 2013; 3: 3153.
He X, Landman S, Bauland SC, van den Dolder J, Koenen HJ, Joosten I. A TNFR2-agonist facilitates high purity expansion of human low purity treg cells. PLoS One 2016; 11: e0156311.
Polz J, Remke A, Weber S, Schmidt D, Weber-Steffens D, Pietryga-Krieger A et al. Myeloid suppressor cells require membrane TNFR2 expression for suppressive activity. Immun Inflamm Dis 2014; 2: 121–130.
Zhang L, Zhang Z, Zhang H, Wu M, Wang Y. Myeloid-derived suppressor cells protect mouse models from autoimmune arthritis via controlling inflammatory response. Inflammation 2014; 37: 670–677.
Chopra M, Biehl M, Steinfatt T, Brandl A, Kums J, Amich J et al. Exogenous TNFR2 activation protects from acute GvHD via host T reg cell expansion. J Exp Med 2016; 213: 1881–1900.
Mates L, Chuah MK, Belay E, Jerchow B, Manoj N, Acosta-Sanchez A et al. Molecular evolution of a novel hyperactive Sleeping Beauty transposase enables robust stable gene transfer in vertebrates. Nat Genet 2009; 41: 753–761.
Bevaart L, Vervoordeldonk MJ, Tak PP. Collagen-induced arthritis in mice. Methods Mol Biol 2010; 602: 181–192.
Bozic CR, Gerard NP, von Uexkull-Guldenband C, Kolakowski JrLF, Conklyn MJ, Breslow R et al. The murine interleukin 8 type B receptor homologue and its ligands. Expression and biological characterization. J Biol Chem 1994; 269: 29355–29358.
Ohmori Y, Wyner L, Narumi S, Armstrong D, Stoler M, Hamilton TA. Tumor necrosis factor-alpha induces cell type and tissue-specific expression of chemoattractant cytokines in vivo. Am J Pathol 1993; 142: 861–870.
Kim EY, Priatel JJ, Teh SJ, Teh HS. TNF receptor type 2 (p75) functions as a costimulator for antigen-driven T cell responses in vivo. J Immunol 2006; 176: 1026–1035.
Coquet JM, Ribot JC, Babala N, Middendorp S, van der Horst G, Xiao Y et al. Epithelial and dendritic cells in the thymic medulla promote CD4+Foxp3+ regulatory T cell development via the CD27-CD70 pathway. J Exp Med 2013; 210: 715–728.
Mahmud SA, Manlove LS, Schmitz HM, Xing Y, Wang Y, Owen DL et al. Costimulation via the tumor-necrosis factor receptor superfamily couples TCR signal strength to the thymic differentiation of regulatory T cells. Nat Immunol 2014; 15: 473–481.
Movahedi K, Guilliams M, Van den Bossche J, Van den Bergh R, Gysemans C, Beschin A et al. Identification of discrete tumor-induced myeloid-derived suppressor cell subpopulations with distinct T cell-suppressive activity. Blood 2008; 111: 4233–4244.
Youn JI, Collazo M, Shalova IN, Biswas SK, Gabrilovich DI. Characterization of the nature of granulocytic myeloid-derived suppressor cells in tumor-bearing mice. J Leukoc Biol 2012; 91: 167–181.
Cuenca AG, Delano MJ, Kelly-Scumpia KM, Moreno C, Scumpia PO, Laface DM et al. A paradoxical role for myeloid-derived suppressor cells in sepsis and trauma. Mol Med 2011; 17: 281–292.
Zhao X, Rong L, Zhao X, Li X, Liu X, Deng J et al. TNF signaling drives myeloid-derived suppressor cell accumulation. J Clin Invest 2012; 122: 4094–4104.
Zhang H, Wang S, Huang Y, Wang H, Zhao J, Gaskin F et al. Myeloid-derived suppressor cells are proinflammatory and regulate collagen-induced arthritis through manipulating Th17 cell differentiation. Clin Immunol 2015; 157: 175–186.
Crook KR, Jin M, Weeks MF, Rampersad RR, Baldi RM, Glekas AS et al. Myeloid-derived suppressor cells regulate T cell and B cell responses during autoimmune disease. J Leukoc Biol 2015; 97: 573–582.
Wang W, Jiao Z, Duan T, Liu M, Zhu B, Zhang Y et al. Functional characterization of myeloid-derived suppressor cell subpopulations during the development of experimental arthritis. Eur J Immunol 2015; 45: 464–473.
Reynolds G, Gibbon JR, Pratt AG, Wood MJ, Coady D, Raftery G et al. Synovial CD4+ T-cell-derived GM-CSF supports the differentiation of an inflammatory dendritic cell population in rheumatoid arthritis. Ann Rheum Dis 2016; 75: 899–907.
Welborn MB III, Van Zee K, Edwards PD, Pruitt JH, Kaibara A, Vauthey JN et al. A human tumor necrosis factor p75 receptor agonist stimulates in vitro T cell proliferation but does not produce inflammation or shock in the baboon. J Exp Med 1996; 184: 165–171.
Hu X, Li B, Li X, Zhao X, Wan L, Lin G et al. Transmembrane TNF-alpha promotes suppressive activities of myeloid-derived suppressor cells via TNFR2. J Immunol 2014; 192: 1320–1331.
Biton J, Semerano L, Delavallee L, Lemeiter D, Laborie M, Grouard-Vogel G et al. Interplay between TNF and regulatory T cells in a TNF-driven murine model of arthritis. J Immunol 2011; 186: 3899–3910.
Boschetti G, Nancey S, Sardi F, Roblin X, Flourie B, Kaiserlian D. Therapy with anti-TNFalpha antibody enhances number and function of Foxp3(+) regulatory T cells in inflammatory bowel diseases. Inflamm Bowel Dis 2011; 17: 160–170.
Huang B, Pan PY, Li Q, Sato AI, Levy DE, Bromberg J et al. Gr-1+CD115+ immature myeloid suppressor cells mediate the development of tumor-induced T regulatory cells and T-cell anergy in tumor-bearing host. Cancer Res 2006; 66: 1123–1131.
Nguyen DX, Ehrenstein MR. Anti-TNF drives regulatory T cell expansion by paradoxically promoting membrane TNF-TNF-RII binding in rheumatoid arthritis. J Exp Med 2016; 213: 1241–1253.
Farooq SM, Kumar A, Ashour HM. Eye-mediated immune tolerance to type II collagen in arthritis-prone strains of mice. J Cell Mol Med 2014; 18: 2512–2518.
Nagar M, Jacob-Hirsch J, Vernitsky H, Berkun Y, Ben-Horin S, Amariglio N et al. TNF activates a NF-kappaB-regulated cellular program in human CD45RA- regulatory T cells that modulates their suppressive function. J Immunol 2010; 184: 3570–3581.
Nie H, Zheng Y, Li R, Guo TB, He D, Fang L et al. Phosphorylation of FOXP3 controls regulatory T cell function and is inhibited by TNF-alpha in rheumatoid arthritis. Nat Med 2013; 19: 322–328.
Valencia X, Stephens G, Goldbach-Mansky R, Wilson M, Shevach EM, Lipsky PE. TNF downmodulates the function of human CD4+CD25hi T-regulatory cells. Blood 2006; 108: 253–261.
Acknowledgements
We thank B Echtenacher for help with the animal experimentation and Melanie Werner-Klein for help with the figures for the manuscript. Part of the work has been funded by the Deutsche Forschungsgemeinschaft with grants to RHS (STR 511/32-1) and HW (WA 1025/31-1). Additionally, the work has been funded by the respective institutions.
Author contributions
VM, TS, DW-S, DZ and ZJ-L generated data on collagen type II-induced arthritis and respective parameters thereof. HW generated the TNCscTNF80 construct. RHS and DNM analyzed and interpreted the data. All authors contributed to figures and the draft manuscript. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Lamontain, V., Schmid, T., Weber-Steffens, D. et al. Stimulation of TNF receptor type 2 expands regulatory T cells and ameliorates established collagen-induced arthritis in mice. Cell Mol Immunol 16, 65–74 (2019). https://doi.org/10.1038/cmi.2017.138
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cmi.2017.138
This article is cited by
-
Co-modulation of TNFR1 and TNFR2 in an animal model of multiple sclerosis
Journal of Neuroinflammation (2023)
-
Sequential treatment with a TNFR2 agonist and a TNFR1 antagonist improves outcomes in a humanized mouse model for MS
Journal of Neuroinflammation (2023)
-
Bivalent structure of a TNFR2-selective and agonistic TNF-α mutein Fc-fusion protein enhances the expansion activity of regulatory T cells
Scientific Reports (2023)
-
TNF and TNF receptors as therapeutic targets for rheumatic diseases and beyond
Nature Reviews Rheumatology (2023)
-
TNFR2 antagonist and agonist: a potential therapeutics in cancer immunotherapy
Medical Oncology (2022)