Abstract
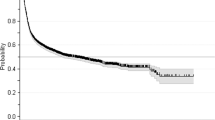
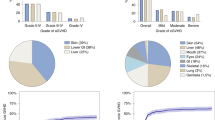
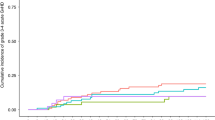
Allogeneic hematopoieteic stem cell transplantation (HSCT) is the only curative treatment for myelofibrosis (MF), but it is still associated with significant risks and complications. One of these complications is poor graft function, but incidence and risk factors have not been studied yet. We retrospectively studied a cohort of 100 patients with primary MF or post-ET/PV MF who received a reduced-intensity HSCT in our center. The cumulative incidence of primary leukocyte engraftment was 98%. The cumulative incidence of poor graft function was 17% and all of the cases occurred before day 100 after HSCT at a median of 49 days (range 24–99 days). In the univariate analysis, age as continuous parameter (P=0.05; hazard ratio 1042) and persistence of significant splenomegaly (defined as palpable splenomegaly of ⩾10 cm under costal margin) at d+30 after HSCT (33% vs 12%; P=0.05) showed an increased cumulative incidence of poor graft function. In conclusion, the incidence of poor graft function after HSCT for MF is rather high, but did not influence survival. Persistence of splenomegaly after transplantation is a significant factor for poor graft function in myelofibrosis patients. Whether therapeutic reduction of splenomegaly before HSCT would result in a lower incidence of poor graft function should be investigated in future studies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Mattsson J, Ringden O, Storb R . Graft failure after allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant 2008; 14 (Supplement 1): 165–170.
Petersdorf EW, Hansen JA, Martin PJ, Woolfrey A, Malkki M, Gooley T et al. Major-histocompatibility-complex class I alleles and antigens in hematopoietic-cell transplantation. N Engl J Med 2001; 345: 1794–1800.
Champlin RE, Horowitz MM, van Bekkum DW, Camitta BM, Elfenbein GE, Gale RP et al. Graft failure following bone marrow transplantation for severe aplastic anemia: risk factors and treatment results. Blood 1989; 73: 606–613.
Storb R, Prentice RL, Thomas ED, Appelbaum FR, Deeg HJ, Doney K et al. Factors associated with graft rejection after HLA-identical marrow transplantation for aplastic anaemia. Br J Haematol 1983; 55: 573–585.
Marmont AM, Horowitz MM, Gale RP, Sobocinski K, Ash RC, van Bekkum DW et al. T-cell depletion of HLA-identical transplants in leukemia. Blood 1991; 78: 2120–2130.
Le Blanc K, Remberger M, Uzunel M, Mattsson J, Barkholt L, Ringdén O . A comparison of nonmyeloablative and reduced-intensity conditioning for allogeneic stem-cell transplantation. Transplantation 2004; 78: 1014–1020.
Remberger M, Watz E, Ringdén O, Mattsson J, Shanwell A, Wikman A . Major ABO blood group mismatch increases the risk for graft failure after unrelated donor hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2007; 13: 675–682.
Storb R, Prentice RL, Thomas ED . Marrow transplantation for treatment of aplastic anemia. An analysis of factors associated with graft rejection. N Engl J Med 1977; 296: 61–66.
Davies SM, Weisdorf DJ, Haake RJ, Kersey JH, McGlave PB, Ramsay NK et al. Second infusion of bone marrow for treatment of graft failure after allogeneic bone marrow transplantation. Bone Marrow Transplant 1994; 14: 73–77.
Wolff SN . Second hematopoietic stem cell transplantation for the treatment of graft failure, graft rejection or relapse after allogeneic transplantation. Bone Marrow Transplant 2002; 29: 545–552.
McCann SR, Bacigalupo A, Gluckman E, Hinterberger W, Hows J, Ljungman P et al. Graft rejection and second bone marrow transplants for acquired aplastic anaemia: a report from the Aplastic Anaemia Working Party of the European Bone Marrow Transplant Group. Bone Marrow Transplant 1994; 13: 233–237.
Klyuchnikov E, El-Cheikh J, Sputtek A, Lioznov M, Calmels B, Furst S et al. CD34(+)-selected stem cell boost without further conditioning for poor graft function after allogeneic stem cell transplantation in patients with hematological malignancies. Biol Blood Marrow Transplant 2014; 20: 382–386.
Stasia A, Ghiso A, Galaverna F, Raiola AM, Gualandi F, Luchetti S et al. CD34 selected cells for the treatment of poor graft function after allogeneic stem cell transplantation. Biol Blood Marrow Transplant 2014; 20: 1435–1454.
Alchalby H, Badbaran A, Zabelina T, Kobbe G, Hahn J, Wolff D et al. Impact of JAK2V617F mutation status, allele burden, and clearance after allogeneic stem cell transplantation for myelofibrosis. Blood 2010; 116: 3572–3581.
Rajantie J, Sale GE, Deeg HJ, Amos D, Appelbaum F, Storb R et al. Adverse effect of severe marrow fibrosis on hematologic recovery after chemoradiotherapy and allogeneic bone marrow transplantation. Blood 1986; 67: 1693–1697.
Soll E, Massumoto C, Clift RA, Buckner CD, Appelbaum FR, Storb R et al. Relevance of marrow fibrosis in bone marrow transplantation: a retrospective analysis of engraftment. Blood 1995; 86: 4667–4673.
Dokal I, Jones L, Deenmamode M, Lewis SM, Goldman JM . Allogeneic bone marrow transplantation for primary myelofibrosis. Br J Haematol 1989; 71: 158–160.
Creemers GJ, Lowenberg B, Hagenbeek A . Allogeneic bone marrow transplantation for primary myelofibrosis. Br J Haematol 1992; 82: 772–773.
Kröger N, Holler E, Kobbe G, Bornhäuser M, Schwerdtfeger R, Baurmann H et al. Allogeneic stem cell transplantation after reduced-intensity conditioning in patients with myelofibrosis: a prospective, multicenter study of the Chronic Leukemia Working Party of the European Group for Blood and Marrow Transplantation. Blood 2009; 114: 5264–5270.
Bacigalupo A, Soraru M, Dominietto A, Pozzi S, Geroldi S, Van Lint MT et al. Allogeneic hemopoietic SCT for patients with primary myelofibrosis: a predictive transplant score based on transfusion requirement, spleen size and donor type. Bone Marrow Transplant 2010; 45: 458–463.
George B, Kerridge I, Gottlieb D, Huang G, Hertzberg M, Svennilson J et al. A reduced intensity conditioning protocol associated with excellent survival in patients with myelofibrosis. Bone Marrow Transplant 2008; 42: 567–568.
Kong Y, Chang YJ, Wang YZ, Chen YH, Han W, Wang Y et al. Association of an impaired bone marrow microenvironment with secondary poor graft function after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2013; 19: 1465–1473.
Dominietto A, Raiola AM, Van Lint MT, Lamparelli T, Gualandi F, Berisso G et al. Factors influencing haematological recovery after allogeneic haemopoietic stem cell transplantation: graft-versus-host disease, donor type, cytomegalovirus infections and cell dose. Br J Haematol 2001; 112: 219–227.
Kröger N, Zabelina T, Alchalby H, Stübig T, Wolschke C, Ayuk F et al. Dynamic of bone marrow fibrosis regression predicts survival after allogeneic stem cell transplantation for myelofibrosis. Biol Blood Marrow Transplant 2014; 20: 812–815.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Author contributions
HA and DY contributed equally to this work. HA designed study, analyzed patient data, performed statistical analysis and wrote the manuscript; DY collected and analyzed data and wrote the manuscript, TZ performed statistical analysis; FA analyzed data and approved the manuscript. NK designed the study and wrote the manuscript. All authors reviewed and approved the manuscript.
Rights and permissions
About this article
Cite this article
Alchalby, H., Yunus, DR., Zabelina, T. et al. Incidence and risk factors of poor graft function after allogeneic stem cell transplantation for myelofibrosis. Bone Marrow Transplant 51, 1223–1227 (2016). https://doi.org/10.1038/bmt.2016.98
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2016.98
This article is cited by
-
Placental expanded mesenchymal-like cells (PLX-R18) for poor graft function after hematopoietic cell transplantation: A phase I study
Bone Marrow Transplantation (2023)
-
Poor graft function after haploidentical stem cell transplantation with post-transplant cyclophosphamide
Annals of Hematology (2023)
-
Non-relapse cytopenias following allogeneic stem cell transplantation, a case based review
Bone Marrow Transplantation (2022)
-
The role of JAK inhibitors in hematopoietic cell transplantation
Bone Marrow Transplantation (2022)
-
Improving allogeneic stem cell transplantation in myelofibrosis
International Journal of Hematology (2022)