Abstract
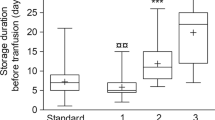
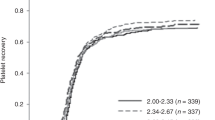
Hematopoietic stem cell transplantation is usually performed without considering the ABO compatibility between donor and recipient. There are few studies analyzing ABO matching impact on transfusion outcome of umbilical cord blood transplantation (UCBT) recipients. The aim of this study was to analyze factors influencing transfusion outcome, highlighting the ABO matching between donor and recipient. This study has reviewed data from 318 patients who underwent single unit UCBT at la Fe University Hospital from January 2000 to December 2014. There were no differences between RBC and platelet (PLT) requirements or RBC and PLT transfusion independence according to ABO matching between donor and recipient. RBC and PLT requirements were statistically correlated (ρ=0,841, P<0.001). A total of 170 and 188 patients achieved RBC and PLT independence, respectively, within 180 days after UCBT. Persistence of recipient isoagglutinins was detected in 6.8% of patients with major ABO incompatibility at median of 176 days (103–269) after UCBT. Autoimmune haemolytic anemia was diagnosed in 15 patients, 12 of them due to cold antibodies. In conclusion, ABO matching has not influenced transfusion requirements of patients undergoing UCBT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Sanz J, Sanz MA, Saavedra S, Lorenzo I, Montesinos P, Senent L et al. Cord blood transplantation from unrelated donors in adults with high-risk acute myeloid leukemia. Biol Blood Marrow Transplant 2010; 16: 86–94.
Solh M, Brunstein C, Morgan S, Weisdorf D . Platelet and red blood cell utilization and transfusion independence in umbilical cord blood and allogeneic peripheral blood hematopoietic cell transplants. Biol Blood Marrow Transplant 2011; 17: 710–716.
Booth GS, Gehrie EA, Bolan CD, Savani BN . Clinical guide to ABO-incompatible allogeneic stem cell transplantation. Biol Blood Marrow Transplant 2013; 19: 1152–1158.
Rowley SD, Donato ML, Bhattacharyya P . Red blood cell-incompatible allogeneic hematopoietic progenitor cell transplantation. Bone Marrow Transplant 2011; 46: 1167–1185.
Watz E, Remberger M, Ringden O, Lundahl J, Ljunqman P, Mattson P et al. Analysis of donor and recipient ABO incompatibility and antibody-associated complications alter allogeneic stem cell transplantation with reduced-intensity conditioning. Biol Blood Marrow Transplant 2014; 20: 264–271.
Wang Z, Sorror ML, Leisenring W, Schoch G, Maloney DG, Sandmaier BM et al. The impact of donor type and ABO incompatibility on transfusion requirements after nonmyeloablative hematopoietic cell Transplantation. Br J Haematol 2010; 149: 101–110.
Blin N, Traineau R, Houssin S, Peffault de Latour R, Petropoulou A, Robin M et al. Impact of donor-recipient major ABO mismatch on allogeneic transplantation outcome according to stem cell source. Biol Blood Marrow Transplant 2010; 16: 1315–1323.
Le Viellez A, P´Ng S, Buffery S, Wright M, Cooney J, Cannell P et al. Red cell and platelet transfusion burden following myeloablative allogeneic haematopoietic stem cell transplantation. Intern Med J 2015; 45: 1286–1292.
Tomonari A, Takahashi S, Ooi J, Tsikada N, Konuma T, Kobayashi T et al. Impact of ABO incompatibility on engraftment and transfusion outcome after unrelated cord blood transplantation: a single institute experience in japan. Bone Marrow Transplantation 2007; 40: 523–528.
Konuma T, Kato S, Ooi J, Oiwa-Monna M, Ebihara Y, Mochizuki S et al. Effect of ABO blood group incompatibility on the outcome of single-unit cord blood transplantation after myeloablative conditioning. Biol Blood Marrow Transplant 2014; 20: 577–592.
Petz LD. . Immune hemolysis associated with transplantation. Semin Hematol 2005; 42: 145–155.
Hoffman PC . Immune hemolytic anemia-selected topics. Hematology Am Soc Hematol Educ Program 2009, 80–86.
Sanz J, Arango M, Carpio N, Montesinos P, Moscardo F, Martin G et al. Autoimmune cytopenias after umbilical cord blood transplantation in adults with haematological malignancies: a single-center experience. Bone Marrow Transplant 2014; 49: 1084–1088.
Kanda Y . Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant 2013; 48: 452–458.
Muñoz J, Shah N, Rezvani K, Hosing C, Bollard CM, Oran B et al. Concise review: umbilical cord blood transplantation: past, present and future. Stem Cells Transl Med 2014; 3: 1435–1443.
Canals C, Muñiz-Diaz E, Martinez C, Martino R, Moreno I, Ramos A et al. Impact of the ABO incompatibility on allogeneic peripheral blood progenitor cell transplantation after reduced intensity conditioning. Transfusion 2004; 44: 1603–1611.
Ozkurt ZN, Yegin ZA, Yenicesu I, Aki SZ, Yagci M, Sucak GT. . Impact of ABO-incompatible donor on early and late outcome of hematopoietic stem cell transplantation. Transplantation Proceedings 2009; 41: 3851–3858.
Dahl D, Hahn A, Hoenecke C, Heuft HG, Dammann E, Stadler M et al. Prolonged isolated red blood cell transfusion requirement after allogeneic blood stem cell transplantation: identification of patients at risk. Transfusion 2010; 50: 649–655.
Holtan SG, Pasquini M, Weisdorf DJ . Acute graft-vs-host disease: a brench-to-bedside update. Blood 2014; 124: 363–373.
Ayas M, Iddiqui K, Al-Jefri A, El-Sohl H, Al-Ahmari A, Khairy A et al. Factors affecting the outcome of related allogeneic hematopoietic cell transplantation in patients with Fanconi anemia. Biol Blood Marrow Transplant 2014; 20: 1599–1603.
Christou G, Kekre N, Petrcich W, Tokessy M, Neurath D, Giulivi A et al. Impact of platelet transfusion on toxicity and mortality after hematopoietic progenitor cell transplantation. Transfusion 2015; 55: 253–258.
Berglund S, LeBlanc K, Remberger M, Gertow J, Uzunel M, Svenberg P et al. Factors with an impact on chimerism development and long-term survival after umbilical cord blood transplantation. Transplantation 2012; 94: 1066–1074.
Sokol RJ, Stamps R, Booker DJ, Scott FM, Laidlaw ST, Vandenberghe EA et al. Posttransplant immune-mediated hemolysis. Transfusion 2002; 42: 198–204.
Salmon JP, Michaux S, Hermanne JP, Baudoux E, Gerard C, Sontag-thull D et al. Delayed massive immune hemolysis mediated by minor ABO incompatibility after allogeneic peripheral blood progenitor cell transplantation. Transfusion 1999; 39: 824–827.
Snell M, Chau C, Hendrix D, Fox R, Downes KA, Creger R et al. Lack of isohemagglutinin production following minor ABO incompatible unrelated HLA mismatched umbilical cord blood transplantation. Bone Marrow Transplantation 2006; 38: 135–140.
Theilgaard-Monch K, Jensen KR, Palm H. . Flow cytometric assessment of lymphocytes subsets, lymphoid progenitors, and hematopoietic stem cell in allogeneic stem cell grafts. Bone Marrow Transplant 2001; 28: 1073–1082.
Acknowledgements
We thank to Mr P Gutteridge for his assistance in the revision of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Solves, P., Carpio, N., Carretero, C. et al. ABO incompatibility does not influence transfusion requirements in patients undergoing single-unit umbilical cord blood transplantation. Bone Marrow Transplant 52, 394–399 (2017). https://doi.org/10.1038/bmt.2016.264
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2016.264
This article is cited by
-
Red blood cell transfusion burden by day 30 predicts mortality in adults after single-unit cord blood transplantation
Bone Marrow Transplantation (2019)