Abstract
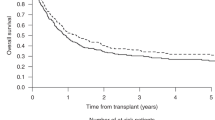
Secondary AML (sAML) has a poor prognosis with conventional chemotherapy alone. Allogeneic hematopoietic cell transplantation (HCT) is beneficial for high-risk AML. Data comparing outcomes of transplants for patients with de novo and sAML are limited. We compared outcomes of patients transplanted for de novo and sAML in first complete remission and investigated the effect of age, HCT comorbidity index (HCT-CI) and karyotype in both groups. A total of 264 patients with de novo (n=180) and sAML (n=84) underwent allogeneic HCT between 1999 and 2013. Median age at transplant was 51 years (range 18–71), median follow-up of survivors was 77 months. Evaluation of all patients demonstrated no significant difference between de novo and sAML for overall survival (P=0.18), leukemia-free survival (P=0.17), cumulative incidence of relapse (P=0.51) and non-relapse mortality (P=0.42). Multivariable and propensity score analyses confirmed the comparable outcomes between de novo and sAML post transplant. Although sAML demonstrates outcomes inferior to de novo AML treated with chemotherapy alone, outcomes following allogeneic HCT are comparable between the two groups.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Leone G, Mele L, Pulsoni A, Equitani F, Pagano L . The incidence of secondary leukemias. Haematologica 1999; 84: 937–945.
Witherspoon RP, Deeg HJ . Allogeneic bone marrow transplantation for secondary leukemia or myelodysplasia. Haematologica 1999; 84: 1085–1087.
Pagana L, Pulsoni A, Tosti ME, Avvisati G, Mele L, Mele M et al. Clinical and biological features of acute myeloid leukaemia occurring as second malignancy: GIMEMA archive of adult acute leukaemia. Br J Haematol 2001; 112: 109–117.
Vardiman JW, Thiele J, Arber DA, Brunning RD, Borowitz MJ, Porwit A et al. The 2008 revision of the World Health Organization (WHO) classification of myeloid neoplasms and acute leukemia: rationale and important changes. Blood 2009; 114: 937–951.
Abdelhameed A, Pond GR, Mitsakakis N, Brandwein J, Chun K, Gupta V et al. Outcome of patients who develop acute leukemia or myelodysplasia as a second malignancy after solid tumors treated surgically or with strategies that include chemotherapy and/or radiation. Cancer 2008; 112: 1513–1521.
Anderson JE, Gooley TA, Schoch G, Anasetti C, Bensinger WI, Clift RA et al. Stem cell transplantation for secondary acute myeloid leukemia: evaluation of transplantation as initial therapy or following induction chemotherapy. Blood 1997; 89: 2578–2585.
Ostgård LS, Kjeldsen E, Holm MS, Brown Pde N, Pedersen BB, Bendix K et al. Reasons for treating secondary AML as de novo AML. Eur J Haematol 2010; 85: 217–226.
Preiss BS, Bergmann OJ, Friis LS, Sørensen AG, Frederiksen M, Gadeberg OV et al. Cytogenetic findings in adult secondary acute myeloid leukemia (AML): frequency of favorable and adverse chromosomal aberrations do not differ from adult de novo AML. Cancer Genet Cytogenet 2010; 202: 108–122.
Dann EJ, Rowe JM . Biology and therapy of secondary leukaemias. Best Pract Res Clin Haematol 2001; 14: 119–137.
Josting A, Wiedenmann S, Franklin J, May M, Sieber M, Wolf J et al. Secondary myeloid leukemia and myelodysplastic syndromes in patients treated for Hodgkin's disease: a report from the German Hodgkin's Lymphoma Study Group. J Clin Oncol 2003; 21: 3440–3446.
Kayser S, Döhner K, Krauter J, Köhne CH, Horst HA, Held G et al. The impact of therapy-related acute myeloid leukemia (AML) on outcome in 2853 adult patients with newly diagnosed AML. Blood 2011; 117: 2137–2145.
Della Porta MG . Prognosis of secondary acute myeloid leukemia. Leuk Res 2013; 37: 857–858.
Park SH, Chi HS, Cho YU, Jang S, Park CJ . Evaluation of prognostic factors in patients with therapy-related acute myeloid leukemia. Blood Res 2013; 48: 185–192.
Ornstein MC, Mukherjee S, Mohan S, Elson P, Tiu RV, Saunthararajah Y et al. Predictive factors for latency period and a prognostic model for survival in patients with therapy-related AML. Am J Hematol 2014; 89: 168–173.
Sorror ML, Sandmaier BM, Storer BE, Maris MB, Baron F, Maloney DG et al. Comorbidity and disease status based risk stratification of outcomes among patients with acute myeloid leukemia or myelodysplasia receiving allogeneic hematopoietic cell transplantation. J Clin Oncol 2007; 25: 4246–4254.
Grimwade D, Hills RK, Moorman AV, Walker H, Chatters S, Goldstone AH et al. Refinement of cytogenetic classification in acute myeloid leukemia: determination of prognostic significance of rare recurring chromosomal abnormalities among 5876 younger adult patients treated in the United Kingdom Medical Research Council trials. Blood 2010; 116: 354–365.
Armand P, Kim HT, Zhang MJ, Perez WS, Dal Cin PS, Klumpp TR et al. Classifying cytogenetics in patients with acute myelogenous leukemia in complete remission undergoing allogeneic transplantation: a Center for International Blood and Marrow Transplant Research study. Biol Blood Marrow Transplant 2012; 18: 280–288.
Khabori MA, El-Emary M, Xu W, Guyatt G, Galal A, Kuruvilla J et al. Impact of intensity of conditioning therapy in patients aged 40-60 years with AML/myelodysplastic syndrome undergoing allogeneic transplantation. Bone Marrow Transplant 2011; 46: 516–522.
Bacigalupo A, Ballen K, Rizzo D, Giralt S, Lazarus H, Ho V et al. Defining the intensity of conditioning regimens: working definitions. Biol Blood Marrow Transplant 2009; 15: 1628–1633.
Korn EL . Censoring distributions as a measure of follow-up in survival analysis. Stat Med 1986; 5: 255–260.
Pepe MS, Mori M . Kaplan-Meier marginal or conditional probability curves in summarizing competing risks failure time data? Stat Med 1993; 12: 737–751.
Fine J, Gray R . A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc 1999; 94: 496–509.
D'Agostino RB Jr . Propensity score methods for bias reduction in the comparison of a treatment to a non-randomized control group. Stat Med 1998; 17: 2265–2281.
Liem YS, Wong JB, Hunink MM, de Charro FT, Winkelmayer WC . Propensity scores in the presence of effect modification: a case study using the comparison of mortality on hemodialysis versus peritoneal dialysis. Emerg Themes Epidemiol 2010; 7: 1.
Gyurkocza B, Storb R, Storer BE, Chauncey TR, Lange T, Shizuru JA et al. Nonmyeloablative allogeneic hematopoietic cell transplantation in patients with acute myeloid leukemia. J Clin Oncol 2010; 28: 2859–2867.
Grimwade D, Walker H, Oliver F, Wheatley K, Harrison C, Harrison G et al. The importance of diagnostic cytogenetics on outcome in AML: analysis of 1,612 patients entered into the MRC AML 10 trial. The Medical Research Council Adult and Children's Leukaemia Working Parties. Blood 1998; 92: 2322–2333.
Schoch C, Kern W, Schnittger S, Hiddemann W, Haferlach T . Karyotype is an independent prognostic parameter in therapy-related acute myeloid leukemia (t-AML): an analysis of 93 patients with t-AML in comparison to 1091 patients with de novo AML. Leukemia 2004; 18: 120–125.
Borthakur G, Lin E, Jain N, Estey EE, Cortes JE, O'Brien S et al. Survival is poorer in patients with secondary core-binding factor acute myelogenous leukemia compared with de novo core-binding factor leukemia. Cancer 2009; 115: 3217–3221.
Acknowledgements
We thank Otsuka Pharmaceuticals Inc for their generous financial support used for data collection and analysis. Funding provided by Otsuka Pharmaceuticals Inc for data collection and analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Funding provided by Otsuka Pharmaceuticals Inc for data collection and analysis. The authors have no other conflict of interest or financial disclosures to declare.
Rights and permissions
About this article
Cite this article
Michelis, F., Atenafu, E., Gupta, V. et al. Comparable outcomes post allogeneic hematopoietic cell transplant for patients with de novo or secondary acute myeloid leukemia in first remission. Bone Marrow Transplant 50, 907–913 (2015). https://doi.org/10.1038/bmt.2015.59
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2015.59
This article is cited by
-
Allogeneic hematopoietic cell transplantation in patients with therapy-related myeloid neoplasm after breast cancer: a study of the Chronic Malignancies Working Party of the EBMT
Bone Marrow Transplantation (2022)
-
Allogeneic hematopoietic cell transplantation can overcome the adverse prognosis indicated by secondary-type mutations in de novo acute myeloid leukemia
Bone Marrow Transplantation (2022)
-
ELN risk stratification and outcomes in secondary and therapy-related AML patients consolidated with allogeneic stem cell transplantation
Bone Marrow Transplantation (2021)
-
Inferior outcome of allogeneic stem cell transplantation for secondary acute myeloid leukemia in first complete remission as compared to de novo acute myeloid leukemia
Blood Cancer Journal (2020)
-
The leukemia strikes back: a review of pathogenesis and treatment of secondary AML
Annals of Hematology (2019)