Abstract
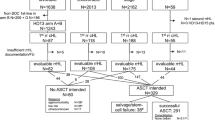
Obesity continues to be an increasing global health issue contributing to the complexity of chemotherapy dosing in the field of SCT. Investigation into the optimal dosing weight used to calculate chemotherapy doses in obese patients undergoing SCT is limited and inconclusive. Our single-center, retrospective study compared safety and efficacy outcomes by body mass index (BMI) for 476 adult lymphoma patients who underwent auto-SCT with a myeloablative chemotherapeutic regimen of BU, CY and etoposide dosed using adjusted body weight. Three weight groups categorized based on BMI were defined: normal/underweight ⩽24.9 kg/m2, overweight 25–29.9 kg/m2 and obese ⩾30 kg/m2. Severity of mucositis, incidence of secondary malignancy, incidence of bacteremia and median hospital length of stay did not differ among the groups. The median times to absolute neutrophil count and platelet recovery were 10 days (P=0.75) and 14 days (P=0.17), respectively. Obese patients had a lower 100-day mortality compared with other weight groups, although this did not translate into an OS benefit. OS and disease relapse were similar among the groups. Our study demonstrates that use of adjusted body weight to calculate chemotherapy doses does not negatively have an impact on outcomes in obese patients undergoing auto-SCT with BU, CY and etoposide.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Flegal KM, Carroll MD, Kit BK, Ogden CL . Prevalence of obesity and trends in the distribution of body mass index among US adults, 1999–2010. JAMA 2012; 307: 491–497.
Hunter RJ, Navo MA, Thaker PH, Bodurka DC, Wolf JK, Smith JA . Dosing chemotherapy in obese patients: actual versus assigned body surface area (BSA). Cancer Treat Rev 2009; 35: 69–78.
Kaestner SA, Sewell GJ . Chemotherapy dosing part I: scientific basis for current practice and use of body surface area. Clin Oncol (R Coll Radiol) 2007; 19: 23–37.
Lopes-Serrao MD, Ussery SM, Hall RG 2nd, Shah SR . Evaluation of chemotherapy-induced severe myelosuppression incidence in obese patients with capped dosing. J Oncol Pract 2011; 7: 13–17.
Field KM, Kosmider S, Jefford M, Michael M, Jennens R, Green M et al. Chemotherapy dosing strategies in the obese, elderly, and thin patient: results of a nationwide survey. J Oncol Pract 2008; 4: 108–113.
Griggs JJ, Mangu PB, Anderson H, Balaban EP, Dignam JJ, Hryniuk WM et al. Appropriate chemotherapy dosing for obese adult patients with cancer: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol 2012; 30: 1553–1561.
Tarella C, Caracciolo D, Gavarotti P, Argentino C, Zallio F, Corradini P et al. Overweight as an adverse prognostic factor for non-Hodgkin’s lymphoma patients receiving high-dose chemotherapy and autograft. Bone Marrow Transplant 2000; 26: 1185–1191.
Navarro WH, Agovi MA, Logan BR, Ballen K, Bolwell BJ, Frangoul H et al. Obesity does not preclude safe and effective myeloablative hematopoietic cell transplantation (HCT) for acute myelogenous leukemia (AML) in adults. Biol Blood Marrow Transplant 2010; 16: 1442–1450.
Navarro WH, Loberiza FR Jr ., Bajorunaite R, van Besien K, Vose JM, Lazarus HM et al. Effect of body mass index on mortality of patients with lymphoma undergoing autologous hematopoietic cell transplantation. Biol Blood Marrow Transplant 2006; 12: 541–551.
Bubalo J, Carpenter PA, Majhail N, Perales MA, Marks DI, Shaughnessy P et al. Conditioning chemotherapy dose adjustment in obese patients: a review and position statement by the american society for blood and marrow transplantation practice guideline committee. Biol Blood Marrow Transplant 2014; 20: 600–616.
Devine BJ . Gentamicin therapy. Drug Intel Clin Pharm 1974; 8: 650–655.
Obesity: preventing and managing the global epidemic. Report of a WHO consultation. World Health Organ Tech Rep Ser 2000; 894: i–xii, 1–253.
Sonis ST, Eilers JP, Epstein JB, LeVeque FG, Liggett WH Jr ., Mulagha MT et al. Validation of a new scoring system for the assessment of clinical trial research of oral mucositis induced by radiation or chemotherapy. Mucositis Study Group. Cancer 1999; 85: 2103–2113.
Horan TC, Andrus M, Dudeck MA . CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control 2008; 36: 309–332.
Hanel M, Kroger N, Sonnenberg S, Bornhauser M, Kruger W, Kroschinsky F et al. Busulfan, cyclophosphamide, and etoposide as high-dose conditioning regimen in patients with malignant lymphoma. Ann Hematol 2002; 81: 96–102.
Copelan EA, Penza SL, Pohlman B, Avalos BR, Goormastic M, Andresen SW et al. Autotransplantation following busulfan, etoposide and cyclophosphamide in patients with non-Hodgkin's lymphoma. Bone Marrow Transplant 2000; 25: 1243–1248.
Kim JE, Lee DH, Yoo C, Kim S, Kim SW, Lee JS et al. BEAM or BuCyE high-dose chemotherapy followed by autologous stem cell transplantation in non-Hodgkin's lymphoma patients: a single center comparative analysis of efficacy and toxicity. Leuk Res 2011; 35: 183–187.
Hadjibabaie M, Iravani M, Taghizadeh M, Ataie-Jafari A, Shamshiri AR, Mousavi SA et al. Evaluation of nutritional status in patients undergoing hematopoietic SCT. Bone Marrow Transplant 2008; 42: 469–473.
Jaime-Perez JC, Colunga-Pedraza PR, Gutierrez-Gurrola B, Brito-Ramirez AS, Gutierrez-Aguirre H, Cantu-Rodriguez OG et al. Obesity is associated with higher overall survival in patients undergoing an outpatient reduced-intensity conditioning hematopoietic stem cell transplant. Blood Cells Mol Dis 2013; 51: 61–65.
Nikolousis E, Nagra S, Paneesha S, Delgado J, Holder K, Bratby L et al. Allogeneic transplant outcomes are not affected by body mass index (BMI) in patients with haematological malignancies. Ann Hematol 2010; 89: 1141–1145.
Deeg HJ, Seidel K, Bruemmer B, Pepe MS, Appelbaum FR . Impact of patient weight on non-relapse mortality after marrow transplantation. Bone Marrow Transplant 1995; 15: 461–468.
Leger F, Nguyen L, Puozzo C . Exposure equivalence between IV (0.8 mg/kg) and oral (1 mg/kg) busulfan in adult patients. Eur J Clin Pharmacol 2009; 65: 903–911.
Nguyen L, Leger F, Lennon S, Puozzo C . Intravenous busulfan in adults prior to haematopoietic stem cell transplantation: a population pharmacokinetic study. Cancer Chemother Pharmacol 2006; 57: 191–198.
Radich JP, Gooley T, Bensinger W, Chauncey T, Clift R, Flowers M et al. HLA-matched related hematopoietic cell transplantation for chronic-phase CML using a targeted busulfan and cyclophosphamide preparative regimen. Blood 2003; 102: 31–35.
Aggarwal C, Gupta S, Vaughan WP, Saylors GB, Salzman DE, Katz RO et al. Improved outcomes in intermediate- and high-risk aggressive non-Hodgkin lymphoma after autologous hematopoietic stem cell transplantation substituting intravenous for oral busulfan in a busulfan, cyclophosphamide, and etoposide preparative regimen. Biol Blood Marrow Transplant 2006; 12: 770–777.
Zhang H, Graiser M, Hutcherson DA, Dada MO, McMillan S, Ali Z et al. Pharmacokinetic-directed high-dose busulfan combined with cyclophosphamide and etoposide results in predictable drug levels and durable long-term survival in lymphoma patients undergoing autologous stem cell transplantation. Biol Blood Marrow Transplant 2012; 18: 1287–1294.
Slattery JT, Clift RA, Buckner CD, Radich J, Storer B, Bensinger WI et al. Marrow transplantation for chronic myeloid leukemia: the influence of plasma busulfan levels on the outcome of transplantation. Blood 1997; 89: 3055–3060.
Andersson BS, Thall PF, Madden T, Couriel D, Wang X, Tran HT et al. Busulfan systemic exposure relative to regimen-related toxicity and acute graft-versus-host disease: defining a therapeutic window for i.v. BuCy2 in chronic myelogenous leukemia. Biol Blood Marrow Transplant 2002; 8: 477–485.
Acknowledgements
We thank the Taussig Bone Marrow Transplantation program, all those involved with the Unified Transplant Database and the Cleveland Clinic Department of Pharmacy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Lau, J., Weber, C., Earl, M. et al. Outcomes after autologous SCT in lymphoma patients grouped by weight. Bone Marrow Transplant 50, 652–657 (2015). https://doi.org/10.1038/bmt.2014.327
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2014.327
This article is cited by
-
Patient eligibility for hematopoietic stem cell transplantation: a review of patient-associated variables
Bone Marrow Transplantation (2019)
-
A time to stop, a time to start: high-dose chemotherapy in overweight and obese patients
Bone Marrow Transplantation (2015)