Abstract
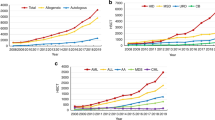
This study presents the pediatric hematopoietic SCT (HSCT) activity in Iran between 1991 and 2012. Overall, 1105 fifteen-year-old or younger patients have undergone HSCT (975 allogeneic and 130 autologous). Annual HSCTs have been increasing steadily since 2007. HLA-matched siblings and other related donors were the main source of HSCs, although since 2008 a national HLA registry has been established to fill the gap for patients lacking a related donor. Inherited abnormalities of RBCs (45.88%), leukemias (27.6%) and BM failure syndromes (11.94%) constituted the majority of HSCTs during this period. Two-year overall survival and disease-free survival rates for all patients were 74.2% (95% confidence interval (CI): 71.6–77) and 66.3% (95% CI: 63.5–69.3), respectively. Leading cause of death in allogeneic group was TRM (165 deaths) and relapse caused the majority of deaths in the autologous group (39 deaths). All HSCTs from the beginning have been performed exclusively with TBI-free-conditioning regimens, which provides unique data for comparison with activities of other centers. Encouraging survival rates provide a basis for future studies on the extensive applicability of TBI-free-conditioning regimens in pediatric HSCT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Lorenz E, Uphoff D, Reid TR, Shelton E . Modification of irradiation injury in mice and guinea pigs by bone marrow injections. J Natl Cancer Inst 1951; 12: 197–201.
Storb R . History of pediatric stem cell transplantation. Pediatr Transplant 2004; 8: 5–11.
Vos O, Davids JA, Weyzen WW, Van Bekkum DW . Evidence for the cellular hypothesis in radiation protection by bone marrow cells. Acta Physiol Pharmacol Neerl 1956; 4: 482–486.
Aljurf MD, Zaidi SZ, El Solh H, Hussain F, Ghavamzadeh A, Mahmoud HK et al. Special issues related to hematopoietic SCT in the Eastern Mediterranean region and the first regional activity report. Bone Marrow Transplant 2009; 43: 1–12.
Benchekroun S, Harif M, Madani A, Quessar A, Zafad S, Rachid R . Present and future of hematology and stem cell transplantation in Morocco. Bone Marrow Transplant 2008; 42: S106–S108.
Ghavamzadeh A, Alimogaddam K, Jahani M, Mousavi S, Iravani M, Bahar B et al. Hematopoietic stem cell transplantation in Iran: 1991 to 2008. Hematol Oncol Stem Cell Ther 2008; 1: 231–238.
Miano M, Labopin M, Hartmann O, Angelucci E, Cornish J, Gluckman E et al. Haematopoietic stem cell transplantation trends in children over the last three decades: a survey by the paediatric diseases working party of the European Group for Blood and Marrow Transplantation. Bone Marrow Transplant 2007; 39: 89–99.
Wachowiak J, Labopin M, Miano M, Chybicka A, Stary J, Sterba J et al. Haematopoietic stem cell transplantation in children in eastern European countries 1985-2004: development, recent activity and role of the EBMT/ESH Outreach Programme. Bone Marrow Transplant 2008; 41: S112–S117.
Yoshimi A, Suzuki R, Atsuta Y, Iida M, Lu DP, Tong W et al. Hematopoietic SCT activity in Asia: a report from the Asia-Pacific Blood and Marrow Transplantation Group. Bone Marrow Transplant 2010; 45: 1682–1691.
Rocha V, Locatelli F . Searching for alternative hematopoietic stem cell donors for pediatric patients. Bone Marrow Transplant 2008; 41: 207–214.
Baldomero H, Gratwohl M, Gratwohl A, Tichelli A, Niederwieser D, Madrigal A et al. The EBMT activity survey 2009: trends over the past 5 years. Bone Marrow Transplant 2011; 46: 485–501.
Pession A, Rondelli R, Paolucci P, Pastore G, Dini G, Bonetti F et al. Hematopoietic stem cell transplantation in childhood: report from the bone marrow transplantation group of the Associazione Italiana Ematologia Oncologia Pediatrica (AIEOP). Haematologica 2000; 85: 638–646.
Wiener LS, Steffen-Smith E, Fry T, Wayne AS . Hematopoietic stem cell donation in children: a review of the sibling donor experience. J Psychosoc Oncol 2007; 25: 45–66.
Hunger SP, Loh KM, Baker KS, Schultz KR . Controversies of and unique issues in hematopoietic cell transplantation for infant leukemia. Biol Blood Marrow Transplant 2009; 15: 79–83.
Atsuta Y, Suzuki R, Yoshimi A, Gondo H, Tanaka J, Hiraoka A et al. Unification of hematopoietic stem cell transplantation registries in Japan and establishment of the TRUMP System. Int J Hematol 2007; 86: 269–274.
Chen PM, Hsiao LT, Chen MH, Chang PM, Liu CY, Hong YC et al. Current status of hematopoietic stem cell transplantation in Taiwan. Bone Marrow Transplant 2008; 42: S133–S136.
Gratwohl A, Baldomero H, Frauendorfer K, Urbano-Ispizua A . EBMT activity survey 2004 and changes in disease indication over the past 15 years. Bone Marrow Transplant 2006; 37: 1069–1085.
Shamsi T, Hashmi K, Adil S, Ahmad P, Irfan M, Raza S et al. The stem cell transplant program in Pakistan—the first decade. Bone Marrow Transplant 2008; 42: S114–S117.
Ahmed SO, Ghavamzadeh A, Zaidi SZ, Baldomero H, Pasquini MC, Hussain F et al. Trends of hematopoietic stem cell transplantation in the Eastern Mediterranean region, 1984-2007. Biol Blood Marrow Transplant 2011; 17: 1352–1361.
Rihn C, Cilley J, Naik P, Pedicano AV, Mehta J . Definition of myeloid engraftment after allogeneic hematopoietic stem cell transplantation. Haematologica 2004; 89: 763–764.
Atkinson K, Horowitz MM, Gale RP, Lee MB, Rimm AA, Bortin MM . Consensus among bone marrow transplanters for diagnosis, grading and treatment of chronic graft-versus-host disease. Committee of the International Bone Marrow Transplant Registry. Bone Marrow Transplant 1989; 4: 247–254.
Schultz KR, Baker KS, Boelens JJ, Bollard CM, Egeler RM, Cowan M et al. Challenges and opportunities for international cooperative studies in pediatric hematopoeitic cell transplantation: priorities of the Westhafen Intercontinental Group. Biol Blood Marrow Transplant 2013; 19: 1279–1287.
Mohamed SY, Fadhil I, Hamladji RM, Hamidieh AA, Fahmy O, Ladeb S et al. Hematopoietic stem cell transplantation in the Eastern Mediterranean Region (EMRO) 2008-2009: report on behalf of the Eastern Mediterranean Bone Marrow Transplantation (EMBMT) Group. Hematol Oncol Stem Cell Ther 2011; 4: 81–93.
Bazarbachi A, Labopin M, Ghavamzadeh A, Giebel S, Al-Zahrani H, Ladeb S et al. Allogeneic matched-sibling hematopoietic cell transplantation for AML: comparable outcomes between Eastern Mediterranean (EMBMT) and European (EBMT) centers. Bone Marrow Transplant 2013; 48: 1065–1069.
Gratwohl A, Baldomero H, Frauendorfer K, Rocha V, Apperley J, Niederwieser D . The EBMT activity survey 2006 on hematopoietic stem cell transplantation: focus on the use of cord blood products. Bone Marrow Transplant 2008; 41: 687–705.
Hamidieh A, Kargar M, Jahani M, Alimoghaddam K, Bahar B, Mousavi SA et al. The outcome of allogeneic hematopoietic stem cell transplants without total body irradiation in pediatric patients with acute lymphoblastic leukemia: single centre experience. J Pediatr Hematol Oncol 2012; 34: 101–107.
Hamidieh AA, Alimoghaddam K, Jahani M, Bahar B, Mousavi SA, Iravani M et al. Non-TBI hematopoietic stem cell transplantation in pediatric AML patients: a single-center experience. J Pediatr Hematol Oncol 2012; 35: e239–e245.
Hamidieh AA, Alimoghaddam K, Jahani M, Mousavi SA, Iravani M, Bahar B et al. Long-term results of non-fludarabine versus fludarabine-based stem cell transplantation without total body irradiation in Fanconi anemia patients. Hematol Oncol Stem Cell Ther 2011; 4: 109–115.
Hamidieh AA, Pourpak Z, Hashemi S, Yari K, Fazlollahi MR, Movahedi M et al. Fludarabine-based reduced-intensity conditioning regimen for hematopoietic stem cell transplantation in primary hemophagocytic lymphohistiocytosis. Eur J Haematol 2014; 92: 331–336.
Hamidieh AA, Pourpak Z, Hosseinzadeh M, Fazlollahi MR, Alimoghaddam K, Movahedi M et al. Reduced-intensity conditioning hematopoietic SCT for pediatric patients with LAD-1: clinical efficacy and importance of chimerism. Bone Marrow Transplant 2011; 47: 646–650.
Hamidieh AA, Pourpak Z, Yari K, Fazlollahi MR, Hashemi S, Behfar M et al. Hematopoietic stem cell transplantation with a reduced-intensity conditioning regimen in pediatric patients with Griscelli syndrome type 2. Pediatr Transplant 2013; 17: 487–491.
Lin KH . Pediatric hematopoietic stem cell transplantation in Taiwan. Transplant Proc 1998; 30: 3477–3480.
Rodriguez-Romo L, Gonzalez-Llano O, Mancias-Guerra C, Jaime-Perez JC, Gomez-Pena A, Ruiz-Arguelles G et al. Pediatric hematopoietic SCT in Mexico: recent activity and main problems. Bone Marrow Transplant 2010; 46: 607–609.
Elbjeirami WM, Abdel-Rahman F, Hussein AA . Probability of finding an HLA-matched donor in immediate and extended families: the Jordanian experience. Biol Blood Marrow Transplant 2012; 19: 221–226.
Aghajanian A . A new direction in population policy and family planning in the Islamic Republic of Iran. Asia Pac Popul J 1995; 10: 3–20.
Lucarelli G, Andreani M, Angelucci E . The cure of thalassemia by bone marrow transplantation. Blood Rev 2002; 16: 81–85.
Gennery AR, Slatter MA, Grandin L, Taupin P, Cant AJ, Veys P et al. Transplantation of hematopoietic stem cells and long-term survival for primary immunodeficiencies in Europe: entering a new century, do we do better? J Allergy Clin Immunol 2010; 126: 602–10 e1-11.
Sevilla J, Fernandez-Plaza S, Diaz MA, Madero L . Hematopoietic transplantation for bone marrow failure syndromes and thalassemia. Bone Marrow Transplant 2005; 35: S17–S21.
Bacigalupo A, Socie G, Lanino E, Prete A, Locatelli F, Locasciulli A et al. Fludarabine, cyclophosphamide, antithymocyte globulin, with or without low dose total body irradiation, for alternative donor transplants, in acquired severe aplastic anemia: a retrospective study from the EBMT-SAA Working Party. Haematologica 2010; 95: 976–982.
Mitchell R, Nivison-Smith I, Anazodo A, Tiedemann K, Shaw PJ, Teague L et al. Outcomes of haematopoietic stem cell transplantation for inherited metabolic disorders: a report from the Australian and New Zealand Children's Haematology Oncology Group and the Australasian Bone Marrow Transplant Recipient Registry. Pediatr Transplant 2013; 17: 582–588.
Prasad VK, Kurtzberg J . Emerging trends in transplantation of inherited metabolic diseases. Bone Marrow Transplant 2008; 41: 99–108.
Boelens JJ . Trends in haematopoietic cell transplantation for inborn errors of metabolism. J Inherit Metab Dis 2006; 29: 413–420.
Aristei C, Santucci A, Corvo R, Gardani G, Ricardi U, Scarzello G et al. In haematopoietic SCT for acute leukemia TBI impacts on relapse but not survival: results of a multicentre observational study. Bone Marrow Transplant 2013; 48: 908–914.
Acknowledgements
We wish to acknowledge the contributions of all physicians, nursing staff, patients and their families and all who have made it possible to continue HSCT through many years.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interests.
Rights and permissions
About this article
Cite this article
Hamidieh, A., Behfar, M., Babaki, A. et al. Hematopoietic SCT in Iranian children 1991–2012. Bone Marrow Transplant 50, 517–522 (2015). https://doi.org/10.1038/bmt.2014.299
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2014.299