Abstract
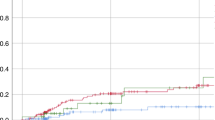
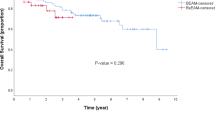
We performed a retrospective study in patients who underwent high-dose chemotherapy and auto-SCT because of haematological malignancies. Forty patients were treated with palifermin while 80 were controls selected after being matched for diagnosis and length of neutropenia. Patients treated with BEAM or BU-CY or THIO-CY (BEAM/BUS) displayed, after palifermin, a lower rate of severe oral mucositis (P=0.03). This beneficial effect of palifermin was not evident in the stratum of patients treated with high-dose melphalan (HD-PAM). After palifermin, we observed in the whole treated population a reduced rate of ‘fever of unknown origin’ (FUO, P=0.02) and of severe infections not related to Gram-positive bacteria (FUO, Gram-negative bacteremia or pneumonia) (P=0.003). This effect of palifermin on infections not related to Gram-positive bacteria was evident only in patients receiving BEAM/BUS (P=0.01) and not in patients treated with HD-PAM (P=0.11). Fibrinogen peak in plasma was found to be reduced after palifermin in the whole population (P=0.01) and in the stratum who received BEAM/BUS (P=0.02) but not in the stratum of HD-PAM. In conclusion, anti-infectious beneficial effects of palifermin are more evident in BEAM/BUS-treated patients and toward some types of infections. Reduction of fibrinogen level after palifermin suggests that this agent reduces not only the rate of infections but also their severity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Gil L, Styczynski G, Komarnicki M . Infectious complication in 314 patients after high dose therapy and autologous hematopoietic stem cell transplantation: risk factor analysis and outcome. Infection 2007; 35: 421–427.
Kolbe K, Domkin D, Hg Derigs, Bhakdi S, Huber C, Aulitzky WE . Infectious complications during neutropenia subsequent to peripheral blood stem cell transplantation. Bone Marrow Transplant 1997; 19: 143–147.
Mossad SB, Longworth D, Goormastic M, Serkey JM, Keys TF, Bolwell BJ . Early infectious complications in autologous bone marrow transplantation: a review of 219 patients. Bone Marrow Transplant 1996; 18: 265–271.
Spielberger R, Stiff P, Bensinger W, Gentile T, Weisdorf D, Kewalramani T et al. Palifermin for oral mucositis afterintensive therapy for hematologic cancers. New Engl J Med 2004; 351: 2590–2598.
Tsirigotis P, Triantafyllou K, Girkas K, Giannopoulou V, Ioannidou E, Chondropoulos S et al. Keratinocyte growth factor is effective in theprevention of intestinal mucositis in patients with hematological malignanciestreated with high-dose chemotherapy and autologous hematopoietic SCT: a video-capsule endoscopy study. Bone Marrow Transplant 2008; 42: 337–343.
Nasilowska-Adamska B, Rzepecki P, Manko J, Czyz A, Markiewicz M, Federowicz I et al. The influence of palifermin (Kepivance) on oral mucositis and acute graft versus host disease in patients with hematological diseases undergoing hematopoietic stem cell transplant. Bone Marrow Transplant 2007; 40: 983–988.
Horsley P, Bauer JD, Mazkowiack R, Gardner R, Bashford J . Palifermin improves severe mucositis, swallowing problems, nutrition impact symptoms, and length of stay in patients undergoing hematopoietic stem cell transplantation. Support Care Cancer 2007; 15: 105–109.
Langner S, Staber P, Schub N, Gramatzki M, Grothe W, Behre G et al. Palifermin reduces incidence and severity oforal mucositis in allogeneic stem-cell transplant recipients. Bone Marrow Transplant 2008; 42: 275–279.
Herbers AH, van der Velden WJ, de Haan AF, Donnelly JP, Blijlevens NM . Impact of palifermin on intestinal mucositis of HSCT recipients after BEAM. Bone Marrow Transplant 2014; 49: 8–10.
Blijlevens N, de Château M, Krivan G, Rabitsch W, Szomor A, Pytlik R et al. In a high-dose melphalan setting, palifermin compared with placebo had no effect on oral mucositis or related patient's burden. Bone Marrow Transplant 2013; 48: 966–971.
Milone G, Leotta S, Pinto V, Avola G, Camuglia MG, Tripepi G . Palifermin in high dose chemotherapy and autologous stem cell transplantation reduces infection rate. Drugs Cell Ther Hematol 2013; 2: 133–146.
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood 2005; 106: 2912–2919.
Donnelly JP, Muus P, Schattenberg A, De Witte T, Horrevorts A, De Pauw BE . A scheme for daily monitoring of oral mucositis in allogeneic BMT recipients. Bone Marrow Transplant 1992; 9: 409–413.
Wardley AM, Jayson GC, Swindell R, Morgenstern GR, Chang J, Bloor R et al. Prospective evaluation of oral mucositis in patients receiving myeloablative conditioning regimens and haemopoietic progenitor rescue. Br J Haematol 2000; 110: 292–299.
Lissalde-Lavigne G, Combescure C, Muller L, Bengler C, Raillard A, Lefrant JY et al. Simple coagulation tests improve survival prediction in patients with septic shock. J Thromb Haemost 2008; 6: 645–653.
Wyleżoł I, Snarski E, Markiewicz M, Kyrcz-Krzemień S, Jędrzejczak WW, Walewski J . Comparison of benefits of early, delayed, and no administration of G-CSF after autologous peripheral blood stem cell transplantation in lymphoma patients. Ann Transplant 2013; 18: 336–341.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Milone, G., Leotta, S., Cupri, A. et al. Palifermin reduces infection rate and hyperfibrinogenemia in patients treated with high-dose chemotherapy based on beam or BU-thiothepa. Bone Marrow Transplant 49, 1193–1197 (2014). https://doi.org/10.1038/bmt.2014.140
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2014.140
This article is cited by
-
Italian consensus conference for the outpatient autologous stem cell transplantation management in multiple myeloma
Bone Marrow Transplantation (2016)