Abstract
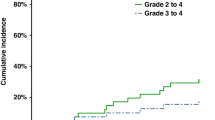
I.v. BU plus fludarabine is an effective conditioning regimen for myeloid neoplasias with low treatment-related mortality. At standard doses, cutaneous toxicity has been reported in <5% of cases. As we observed a much higher incidence of cutaneous toxicity in patients who received predominantly pharmacokinetically based doses of BU, we performed a retrospective analysis of 61 patients who received i.v. BU plus fludarabine (+/− antithymocyte globulin; ATG) as a conditioning regimen before allogeneic PBSC transplant. Of the 58 evaluable patients, 33 (57%) developed cutaneous toxicity that fell within the spectrum of toxic erythema of chemotherapy (TEC). The median onset of TEC was 22 days and most patients had multiple sites of involvement, with the groin, axillae and palms/soles being the favored sites. In men, scrotal involvement, sometimes severe, was also commonly observed. Initially, allergic reactions to antibiotics, fungal infections and GVHD were also considered until the clinical presentation of TEC became well recognized. In all patients, the skin healed without specific therapy but resolution often required several weeks. This series suggests that TEC is common after BU/fludarabine+/− ATG and it is important for transplant physicians to recognize, particularly as misdiagnosis could lead to inappropriate treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Andersson BS, de Lima M, Thall PF, Wang X, Couriel D, Korbling M et al. Once daily i.v. busulfan and fludarabine (i.v. Bu-Flu) compares favorably with i.v. busulfan and cyclophosphamide (i.v. BuCy2) as pretransplant conditioning therapy in AML/MDS. Biol Blood Marrow Transplant 2008; 14: 672–684.
Bredeson CN, Zhang MJ, Agovi MA, Bacigalupo A, Bahlis NJ, Ballen K et al. Outcomes following HSCT using fludarabine, busulfan, and thymoglobulin: a matched comparison to allogeneic transplants conditioned with busulfan and cyclophosphamide. Biol Blood Marrow Transplant 2008; 14: 993–1003.
Chunduri S, Dobogai LC, Peace D, Saunthararajah Y, Quigley J, Chen YH et al. Fludarabine/i.v. BU conditioning regimen: myeloablative, reduced intensity or both? Bone Marrow Transplant 2008; 41: 935–940.
de Lima M, Couriel D, Thall PF, Wang X, Madden T, Jones R et al. Once-daily intravenous busulfan and fludarabine: clinical and pharmacokinetic results of a myeloablative, reduced-toxicity conditioning regimen for allogeneic stem cell transplantation in AML and MDS. Blood 2004; 104: 857–864.
Russell JA, Tran HT, Quinlan D, Chaudhry A, Duggan P, Brown C et al. Once-daily intravenous busulfan given with fludarabine as conditioning for allogeneic stem cell transplantation: study of pharmacokinetics and early clinical outcomes. Biol Blood Marrow Transplant 2002; 8: 468–476.
Russell JA, Irish W, Balogh A, Chaudhry MA, Savoie ML, Turner AR et al. The addition of 400 cGY total body irradiation to a regimen incorporating once-daily intravenous busulfan, fludarabine, and antithymocyte globulin reduces relapse without affecting nonrelapse mortality in acute myelogenous leukemia. Biol Blood Marrow Transplant 2010; 16: 509–514.
Kröger N, Bornhäuser M, Ehninger G, Schwerdtfeger R, Biersack H, Sayer HG et al. Allogeneic stem cell transplantation after a fludarabine/busulfan-based reduced-intensity conditioning in patients with myelodysplastic syndrome or secondary acute myeloid leukemia. Ann Hematol 2003; 82: 336–342.
Santarone S, Pidala J, Di Nicola M, Field T, Alsina M, Ayala E et al. Fludarabine and pharmacokinetic-targeted busulfan before allografting for adults with acute lymphoid leukemia. Biol Blood Marrow Transplant 2011; 17: 1505–1511.
Kerbauy FR, Rodrigues M, de Souza Santos FP, Sobrinho JN, Kutner JM, Torres MA et al. Allogeneic hematopoietic stem cell transplant after intravenous busulfan and fludarabine conditioning. Leuk Lymphoma 2011; 52: 321–324.
Geddes M, Kangarloo SB, Naveed F, Quinlan D, Chaudhry MA, Stewart D et al. High busulfan exposure is associated with worse outcomes in a daily i.v. busulfan and fludarabine allogeneic transplant regimen. Biol Blood Marrow Transplant 2008; 14: 220–228.
Hamadani M, Craig M, Phillips GS, Abraham J, Tse W, Cumpston A et al. Higher busulfan dose intensity does not improve outcomes of patients undergoing allogeneic haematopoietic cell transplantation following fludarabine, busulfan-based reduced toxicity conditioning. Hematol Oncol 2011; 29: 202–210.
Pidala J, Kim J, Anasetti C, Kharfan-Dabaja MA, Nishihori T, Field T et al. Pharmacokinetic targeting of intravenous busulfan reduces conditioning regimen related toxicity following allogeneic hematopoietic cell transplantation for acute myelogenous leukemia. J Hematol Oncol 2010; 3: 36.
Ryu SG, Lee JH, Choi SJ, Lee YS, Seol M, Hur EH et al. Randomized comparison of four-times-daily versus once-daily intravenous busulfan in conditioning therapy for hematopoietic cell transplantation. Biol Blood Marrow Transplant 2007; 13: 1095–1105.
Russell JA, Duan Q, Chaudhry MA, Savoie ML, Balogh A, Turner AR et al. Transplantation from matched siblings using once-daily intravenous busulfan/fludarabine with thymoglobulin: a myeloablative regimen with low nonrelapse mortality in all but older patients with high-risk disease. Biol Blood Marrow Transplant 2008; 14: 888–895.
Bolognia JL, Cooper DL, Glusac EJ . Toxic erythema of chemotherapy: a useful clinical term. J Am Acad Dermatol 2008; 59: 524–529.
Ikeda H, Kawano H, Kitaura T, Kimura A, Kihira K . Acral erythema associated with high-dose methotrexate infusion. Ann Pharmacother 1999; 33: 646.
Hellier I, Bessis D, Sotto A, Margueritte G, Guilhou JJ . High-dose methotrexate-induced bullous variant of acral erythema. Arch Dermatol 1996; 132: 590–591.
Feizy V, Namazi MR, Barikbin B, Ehsani A . Methotrexate-induced acral erythema with bullous reaction. Dermatol Online J 2003; 9: 14.
Beri R, Chunduri S, Sweiss K, Peace DJ, Mactal-Haaf C, Dobogai LC et al. Reliability of a pretransplant i.v. BU test dose performed 2 weeks before myeloablative FluBu conditioning regimen. Bone Marrow Transplant 2010; 45: 249–253.
Morgan M, Dodds A, Atkinson K, Szer J, Downs K, Biggs J . The toxicity of busulphan and cyclophosphamide as the preparative regimen for bone marrow transplantation. Br J Haematol 1991; 77: 529–534.
Hoy SM, Lyseng-Williamson KA . Intravenous busulfan: in the conditioning treatment of pediatric patients prior to hematopoietic stem cell transplantation. Paediatr Drugs 2007; 9: 271–278.
Crider MK, Jansen J, Norins AL, McHale MS . Chemotherapy-induced acral erythema in patients receiving bone marrow transplantation. Arch Dermatol 1986; 122: 1023–1027.
Azurdia RM, Clark RE, Friedmann PS . Chemotherapy-induced acral erythema (CIAE) with bullous reaction. Clin Exp Dermatol 1999; 24: 64–66.
Burgdorf WH, Gilmore WA, Ganick RG . Peculiar acral erythema secondary to high-dose chemotherapy for acute myelogenous leukemia. Ann Intern Med 1982; 97: 61–62.
Levine LE, Medenica MM, Lorincz AL, Soltani K, Raab B, Ma A . Distinctive acral erythema occurring during therapy for severe myelogenous leukemia. Arch Dermatol 1985; 121: 102–104.
Lokich JJ, Moore C . Chemotherapy-associated palmar-plantar erythrodysesthesia syndrome. Ann Intern Med 1984; 101: 798–799.
Perkins JB, Kim J, Anasetti C, Fernandez HF, Perez LE, Ayala E et al. Maximally tolerated busulfan systemic exposure in combination with fludarabine as conditioning before allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant 2012; 18: 1099–1107.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Parker, T., Cooper, D., Seropian, S. et al. Toxic erythema of chemotherapy following i.v. BU plus fludarabine for allogeneic PBSC transplant. Bone Marrow Transplant 48, 646–650 (2013). https://doi.org/10.1038/bmt.2012.218
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2012.218
Keywords
This article is cited by
-
Dermatologic Conditions of the Early Post-Transplant Period in Hematopoietic Stem Cell Transplant Recipients
American Journal of Clinical Dermatology (2019)
-
Evaluation and Treatment of Stem Cell Transplant-Related Dermatoses
Current Dermatology Reports (2016)