Abstract
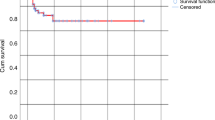
There is little data comparing the activity and toxicity of donor lymphocyte therapy with granulocyte (G)-CSF-mobilized cells (G-donor lymphocyte infusion (DLI)) with the conventionally collected DLI (C-DLI) after allogeneic blood or marrow transplantation. We retrospectively evaluated 67 patients to compare the efficacy and toxicity of GCSF-mobilized DLI with C-DLI in the treatment of relapse of malignant disease or poor donor engraftment post transplant. We assessed clinical outcomes that may represent the immunological outcome of DLI. The median OS was 210 days (range 3–2436 days), 291 days (range 17–1491 days) in the G-DLI group (15 patients) and 207.5 days (range 3–2436 days) in the C-DLI group (52 patients). The median PFS time was 72 days (range 8–1491 days) in the G-DLI group vs 82 days (range 1–2436 days) in the C-DLI group. Rates of post DLI GVHD and improvement in donor engraftment were similar in the G-DLI and C-DLI groups. We conclude that G-DLI appears to have similar therapeutic activity to that seen with C-DLI, and where such cells are available they may be substituted for conventional donor lymphocytes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Copelan EA, Biggs JC, Thompson JM, Crilley P, Szer J, Kapoor N et al. Treatment for acute myelocytic leukemia with allogeneic bone marrow transplantation following preparation with BuCy2. Blood 1991; 78: 838–843.
Frassoni F, Barrett AJ, Grañena A, Ernst P, Garthon G, Kolb HJ et al. Relapse after allogeneic bone marrow transplantation for acute leukaemia: a survey by the E.B.M.T. of 117 cases. Br J Haematol 1988; 70: 317–320.
Schmid C, Labopin M, Nagler A, Bornhäuser M, Fnke J, Fassas A et al. Donor lymphocyte infusion in the treatment of first hematological relapse after allogeneic stem-cell transplantation in adults with acute myeloid leukemia: a retrospective risk factors analysis and comparison with other strategies by the EBMT Acute Leukemia Working Party. J Clin Oncol 2007; 25: 4938–4945.
Mortimer J, Blinder MA, Schulman S, Appelbaum FR, Buckner CD, Clift RA et al. Relapse of acute leukemia after marrow transplantation: natural history and results of subsequent therapy. J Clin Oncol 1989; 7: 50–57.
Slavin S, Naparstek E, Nagler A, Ackerstein A, Samuel S, Kapelushnik J et al. Allogeneic cell therapy with donor peripheral blood cells and recombinant human interleukin-2 to treat leukemia relapse after allogeneic bone marrow transplantation. Blood 1996; 87: 2195–2204.
Kolb HJ, Schattenberg A, Goldman JM, Hertenstein B, Jacobsen N, Arcese W et al. Graft-versus-leukemia effect of donor lymphocyte transfusions in marrow grafted patients. Blood 1995; 86: 2041–2050.
Bensinger WI, Martin PJ, Storer B, Clift R, Forman SJ, Negrin R et al. Transplantation of bone marrow as compared with peripheral-blood cells from HLA-identical relatives in patients with hematologic cancers. N Engl J Med 2001; 344: 175–181.
Couban S, Simpson DR, Barnett MJ, Bredeson C, Hubesch L, Howson-Jan K et al. A randomized multicenter comparison of bone marrow and peripheral blood in recipients of matched sibling allogeneic transplants for myeloid malignancies. Blood 2002; 100: 1525–1531.
Flowers ME, Parker PM, Johnston LJ, Matos AV, Storer B, Bensinger WI et al. Comparison of chronic graft-versus-host disease after transplantation of peripheral blood stem cells versus bone marrow in allogeneic recipients: long-term follow-up of a randomized trial. Blood 2002; 100: 415–419.
Klangsinsirikul P, Russell NH . Peripheral blood stem cell harvests from G-CSF-stimulated donors contain a skewed Th2 CD4 phenotype and a predominance of type 2 dendritic cells. Exp Hematol 2002; 30: 495–501.
Kolb HJ, Mittermuller J, Clemm C, Holler E, Ledderose G, Brehm G et al. Donor leukocyte transfusions for treatment of recurrent chronic myelogenous leukemia in marrow transplant patients. Blood 1990; 76: 2462–2465.
Collins RH, Rogers ZR, Bennett M, Kumar V, Nikein A, Fay JW et al. Hematologic relapse of chronic myelogenous leukemia following allogeneic bone marrow transplantation: apparent graft-versus-leukemia effect following abrupt discontinuation of immunosuppression. Bone Marrow Transplant 1992; 10: 391–395.
Mackinnon S, Papadopoulos EB, Carabasi MH, Reich L, Collins NH, Boulad F et al. Adoptive immunotherapy evaluating escalating doses of donor leukocytes for relapse of chronic myeloid leukemia after bone marrow transplantation: separation of graft-versus-leukemia responses from graft-versus-host disease. Blood 1995; 86: 1261–1268.
Siena S, Schiavo R, Pedrazzoli P, Carlo-Stella C . Therapeutic relevance of CD34 cell dose in blood cell transplantation for cancer therapy. J Clin Oncol 2000; 18: 1360–1377.
Kiss JE, Rybka WB, Winkelstein A, de Magalhaes-Silverman M, Lister J, D’Andrea P et al. Relationship of CD34+ cell dose to early and late hematopoiesis following autologous peripheral blood stem cell transplantation. Bone Marrow Transplant 1997; 19: 303–310.
Guglielmi C, Arcese W, Dazzi F, Brand R, Bunjes D, Verdonck LF et al. Donor lymphocyte infusion for relapsed chronic myelogenous leukemia: prognostic relevance of the initial cell dose. Blood 2002; 100: 397–405.
Collins RHJ, Shpilberg O, Drobyski WR, Porter DL, Giralt S, Champlin R et al. Donor leukocyte infusions in 140 patients with relapsed malignancy after allogeneic bone marrow transplantation. J Clin Oncol 1997; 15: 433–444.
Dazzi F, Szydlo RM, Craddock C, Cross NC, Kaeda J, Chase A et al. Comparison of single-dose and escalating-dose regimens of donor lymphocyte infusion for relapse after allografting for chronic myeloid leukemia. Blood 2000; 95: 67–71.
Dazzi F, Szydlo RM, Cross NC, Craddock C, Kaeda J, Kanfer E et al. Durability of responses following donor lymphocyte infusions for patients who relapse after allogeneic stem cell transplantation for chronic myeloid leukemia. Blood 2000; 96: 2712–2716.
Schattenberg A, Schaap N, Van De Wiel-Van Kemenade E, Bär B, Preijers F, Van Der Maazen R et al. In relapsed patients after lymphocyte depleted bone marrow transplantation the percentage of donor T lymphocytes correlates well with the outcome of donor leukocyte infusion. Leuk Lymphoma 1999; 32: 317–325.
Dazzi F, Szydlo RM, Apperley JF, Goldman JM . Prognostic factors for acute graft-versus-host disease after donor lymphocyte infusions. Blood 2002; 100: 2673–2674.
Huff CA, Fuchs EJ, Smith BD, Blackford A, Garrett-Mayer E, Brodsky RA et al. Graft-versus-host reactions and the effectiveness of donor lymphocyte infusions. Biol Blood Marrow Transplant 2006; 12: 414–421.
Fozza C, Szydlo RM, Abdel-Rehim MM, Nadal E, Goldman JM, Apperley JF et al. Factors for graft-versus-host disease after donor lymphocyte infusions with an escalating dose regimen: lack of association with cell dose. Br J Haematol 2007; 136: 833–836.
Verdonck LF, Petersen EJ, Lokhorst HM, Nieuwenhuis HK, Dekker AW, Tilanus MG et al. Donor leukocyte infusions for recurrent hematologic malignancies after allogeneic bone marrow transplantation: impact of infused and residual donor T cells. Bone Marrow Transplant 1998; 22: 1057–1063.
Lokhorst HM, Schattenberg A, Cornelissen JJ, Van Oers MH, Fibbe W, Russell L et al. Donor lymphocyte infusions for relapsed multiple myeloma after allogeneic stem-cell transplantation: predictive factors for response and long-term outcome. J Clin Oncol 2000; 18: 3031–3037.
Jun HX, Jun CY, Yu ZX . In vivo induction of T-cell hyporesponsiveness and alteration of immunological cells of bone marrow grafts using granulocyte colony-stimulating factor. Haematologica 2004; 89: 1517–1524.
Chen SH, Li X, Huang XJ . Effect of recombinant human granulocyte colony-stimulating factor on T-lymphocyte function and the mechanism of this effect. Int J Hematol 2004; 79: 178–184.
Arpinati M, Green CL, Heimfeld S, Heuser JE, Anasetti C . Granulocyte-colony stimulating factor mobilizes T helper 2-inducing dendritic cells. Blood 2000; 95: 2484–2490.
Pan L, Delmonte J, Jalonen CK, Ferrara JL . Pretreatment of donor mice with granulocyte colony-stimulating factor polarizes donor T lymphocytes toward type-2 cytokine production and reduces severity of experimental graft-versus-host disease. Blood 1995; 86: 4422–4429.
Zeng D, Jbakhsh-Jones S, Strober S . Granulocyte colony-stimulating factor reduces the capacity of blood mononuclear cells to induce graft-versus-host disease: impact on blood progenitor cell transplantation. Blood 1997; 90: 453–463.
Krenger W, Snyder KM, Byon JC, Falzarano G, Ferrara JL . Polarized type 2 alloreactive CD4+ and CD8+ donor T cells fail to induce experimental acute graft-versus-host disease. J Immunol 1995; 155: 585–593.
Krenger W, Cooke KR, Crawford JM, Sonis ST, Simmons R, Pan L et al. Transplantation of polarized type 2 donor T cells reduces mortality caused by experimental graft-versus-host disease. Transplantation 1996; 62: 1278–1285.
Fowler DH, Kurasawa K, Smith R, Eckhaus MA, Gress RE . Donor CD4-enriched cells of Th2 cytokine phenotype regulate graft-versus-host disease without impairing allogeneic engraftment in sublethally irradiated mice. Blood 1994; 84: 3540–3549.
Imami N, Brookes PA, Lombardi G, Hakooz B, Johns M, Goldman JM et al. Association between interleukin-4-producing T lymphocyte frequencies and reduced risk of graft-versus-host disease. Transplantation 1998; 65: 979–988.
Pan L, Teshima T, Hill GR, Bungard D, Brinson YS, Reddy VS et al. Granulocyte colony-stimulating factor-mobilized allogeneic stem cell transplantation maintains graft-versus-leukemia effects through a perforin-dependent pathway while preventing graft-versus-host disease. Blood 1999; 93: 4071–4078.
Reyes E, Garcia-Castro L, Esquivel F, Hornedo J, Cortes-Funes H, Solovera J et al. Granulocyte colony-stimulating factor (G-CSF) transiently suppresses mitogen-stimulated T-cell proliferative response. Br J Cancer 1999; 80: 229–235.
Mielcarek M, Graf L, Johnson G, Torok-Storb B . Production of interleukin-10 by granulocyte colony-stimulating factor-mobilized blood products: a mechanism for monocyte-mediated suppression of T-cell proliferation. Blood 1998; 92: 215–222.
Eckardt JR, Roodman GD, Boldt DH, Clark GM, Alvarez R, Page C et al. Comparison of engraftment and acute GVHD in patients undergoing cryopreserved or fresh allogeneic BMT. Bone Marrow Transplant 1993; 11: 125–131.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Abbi, K., Zhu, J., Ehmann, W. et al. G-CSF mobilized vs conventional donor lymphocytes for therapy of relapse or incomplete engraftment after allogeneic hematopoietic transplantation. Bone Marrow Transplant 48, 357–362 (2013). https://doi.org/10.1038/bmt.2012.144
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2012.144
Keywords
This article is cited by
-
New strategies of DLI in the management of relapse of hematological malignancies after allogeneic hematopoietic SCT
Bone Marrow Transplantation (2016)
-
Mobilized peripheral blood grafts include more than hematopoietic stem cells: the immunological perspective
Bone Marrow Transplantation (2015)
-
Preemptive DLI without withdrawal of immunosuppression to promote complete donor T-cell chimerism results in favorable outcomes for high-risk older recipients of alemtuzumab-containing reduced-intensity unrelated donor allogeneic transplant: a prospective phase II trial
Bone Marrow Transplantation (2014)