Abstract
Background:
Despite recent therapeutic and diagnostic advances, prostate cancer remains the second leading cause of cancer-related deaths among men in the Western world. Oncolytic viruses that replicate selectively in tumour cells represent a novel treatment candidate for these malignancies.
Methods:
We analysed infectivity of avirulent Semliki Firest virus SFV-VA7 in human prostate cancer cell lines VCaP, LNCaP and 22Rv1 and in nonmalignant prostate epithelial cell line RWPE-1. Therapeutic potency of SFV-VA7 was evaluated in subcutaneous and orthotopic mouse LNCaP xenograft models.
Results:
SFV-VA7 infected and killed the tested human prostate cancer cell lines irrespective of their hormone response status, while the nonmalignant prostate epithelial cell line RWPE-1 proved highly virus resistant. Notably, a single peritoneal dose of SFV-VA7 was sufficient to eradicate all subcutaneous and orthotopic LNCaP tumours.
Conclusions:
Our results indicate that SFV-VA7 is a novel, promising therapeutic virus against prostate cancer warranting further testing in early clinical trials.
Similar content being viewed by others
Main
Prostate cancer is by far the most common malignancy in men, and after lung- and bronchial cancer, the second leading cause of cancer-related male deaths in the developed world (Siegel et al, 2016). Approximately 1 out of 7 men will be diagnosed with prostate cancer during his lifetime (ACS cancer statistics, 2016). Despite improvements in early detection, advanced disease is still incurable (Siegel et al, 2016). Oncolytic virotherapy with tumour-specific viruses represents a novel treatment option for such urological malignancies. Indeed, vaccinia-, adeno- and reoviruses have shown promise against prostate cancer in early-phase clinical trials (Delwar et al, 2016). Oncolytic Semliki Forest virus SFV-VA7 has demonstrated significant promise in a variety of murine cancer models, including human melanoma (Vähä-Koskela et al, 2006), osteosarcoma (Ketola et al, 2008), lung carcinoma (Määttä et al, 2008), glioma (Heikkilä et al, 2010) and syngeneic colorectal carcinoma (Ruotsalainen et al, 2015), but its potential against prostate cancer has thus far remained unexplored.
Here we have studied antiviral type I interferon (IFN-I) responses of VCaP, LNCaP and 22Rv1 prostate cancer cell lines and their susceptibility to SFV-VA7-mediated oncolysis in comparison to nonmalignant RWPE-1 prostate endothelial cells. We show that prostate cancer cell lines were readily infected and killed by SFV-VA7, whereas nonmalignant prostate cells displayed resistance to infection. Of the tested cell lines, malignant VCaP and nonmalignant RWPE-1 showed functional IFN-I response indicated by STAT1 phosphorylation. Peritoneal administration of SFV-VA7 eradicated 100% of the large, established subcutaneous and orthotopic tumours without observed toxicities.
Materials and methods
Cell lines and viruses
RWPE-1 (ATCC) cells were cultured in keratinocyte serum-free medium supplemented with bovine pituitary extract and human recombinant epidermal growth factor (GIBCO). 22Rv1 (ATCC) and LNCaP cells were cultured in RPMI-1640 (Sigma-Aldrich, St Louis, MO, USA) supplemented with 10% fetal bovine serum (FBS, Autogen Bioclear, Calne, UK) and 1% L-glutamine (Sigma-Aldrich). VCaP cells were cultured in Dulbecco's Modified Eagle's Medium (Sigma-Aldrich) supplemented with 10 FBS and 1% L-glutamine. All the used culture media were supplemented with 1% penicillin, streptomycin (Sigma-Aldrich). Production of replicative SFV VA7-EGFP and VA7-Rluc have been described earlier (Vähä-Koskela et al, 2003; Heikkilä et al, 2010).
Measuring of virus production
A total of 80 000 RWPE-1, 22Rv1 or VCaP cells were seeded on 12-well plate and infected 3 days later with VA7-EGFP (MOI=0.01) in 1 ml volume. Cells were washed with PBS 2 h post infection (pi.) and fresh medium was added. Medium (200 μl) from two parallel wells was collected 48 and 72 h pi. for plaque titration (Ruotsalainen et al, 2015). Cell infections were carried out with SFV-VA7 viruses expressing either EGFP or Rluc transgene, which do not influence the infection (Heikkilä et al, 2010).
Cell viability assay
A total of 40 000 RWPE-1, 22Rv1, VCaP or LNCaP cells were seeded on 96 –well plate and infected with VA7-EGFP using MOI=0.01−10. Cell viability was measured from 3 to 4 parallel wells 4 days pi. with Cell proliferation Kit I (Roche Applied Science, Penzberg, Germany). LNCaP viability was measured with Calcein AM assay.
Western blot
To induce STAT1 phosphorylation, cells were treated with 1000 units per ml of human recombinant IFN-β (Nordic Biosite, Täby, Sweden) and incubated at +37 °C for 20 min. Expression of viral proteins and infection-induced STAT1 phosphorylation was detected from VA7-EGFP-infected cells MOI=10, 18 h pi. Western blotting was performed as described before (Martikainen et al, 2015) using the following antibodies: Rabbit polyclonal antibody against nsP3 (kindly provided by Tero Ahola, Helsinki) or viral structural proteins (produced in house), rabbit anti-STAT1 (61011; BD Transduction Laboratories, Franklin Lakes, NJ, USA), rabbit anti-Tyr701-phosphorylated STAT1 (D4A7; Cell Signaling Technology, Danvers, MA, USA) and mouse anti-β-actin (C4, Santa Cruz Biotechnology, Santa Cruz, CA, USA). Anti-rabbit–Cy5 and anti-mouse–Cy3 (ECL Plex, GE Healthcare, Chicago, IL, USA) were used as secondary antibodies. Imaging was performed with a Typhoon scanner (GE Healthcare).
Immunohistochemistry
Brain and tumour tissue samples were collected from mice 72 h after intraperitoneal injection of 1 × 106 PFU VA7-EGFP virus or PBS. Paraformaldehyde-fixed and paraffin-embedded tissue sections were stained using the Vectastain ABC kit (rabbit IgG; Vector Laboratories, Burlingame, CA, USA) and a polyclonal rabbit antibody reactive against SFV structural proteins (produced in-house). 3,3′-diaminobenzidine (SigmaFast DAB; Sigma-Aldrich) was used for colour reaction.
In vivo experiments
CAnN.Cg-Foxn1nu/Crl Balb/c nude mice (Charles River) were used for the animal studies. Work was conducted following Finnish Board of Animal Experimentation guidelines, licence number ESAVI/3257/04.10.07/2014.
For subcutaneous tumours, 5 × 106 LNCaP cells were injected in 100 μl volume of PBS/Matrigel (50 out of 50) into left flanks of mice. Tumour growth was stimulated with implanted testosterone pellet (10 mg, Innovative Research of America, Sarasota, FL, USA). Upon palpable tumours (100–300 mm3 as measured with caliper ((length × width2)/2), mice received a single intraperitoneal injection of 1 × 106 PFU VA7-GFP in 100 μl PBS. PBS was used as the vehicle treatment. Mice were killed when tumour size reached 1000 mm2.
Orthotopic prostate tumours were induced as previously described (Tuomela et al, 2009). Briefly, a small incision was made above pubic symphysis and the bladder and seminal vesicles were carefully moved to expose the prostate. 5 × 105 LNCaP cells in 20 μl volume of PBS/Matrigel were injected into the prostate. Prostate-specific antigen (PSA) secretion into serum was measured with PSA detection kit (Roche, Applied Science). Mice received a single intraperitoneal injection of 1 × 106 PFU VA7-GFP in 100 μl PBS, when PSA was detectable.
Results
VA7-EGFP infects and kills human prostate cancer cell lines but not nonmalignant prostate epithelial cells
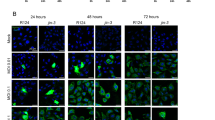
To test the feasibility of SFV-VA7 against prostate cancer, we first analysed its infectivity in vitro. Interestingly, even low multiplicity of infection (MOI) of SFV-VA7 efficiently infected and killed all tested VCaP, 22Rv1 and LNCaP prostate cancer cell lines (Figure 1A), whereas the nonmalignant RWPE-1 cells were significantly more resistant and displayed more than 100-fold reduction in supernatant viral titers (Figure 1B).
SFV-VA7 Infects and kills prostate cancer cell lines VCaP, LNCaP, 22Rv1, while replicating poorly in nonmalignant RWPE-1 prostate cells. (A) Cell viability (mean+s.d.) measured 72 h after infection with different MOI of SFV-VA7. The data from two experiments is presented. (B) virus titer (mean+s.d.) in cell culture medium measured at 48 h and 72 h post infection with SFV-VA7 MOI 0.01. The data from two experiments presented. (C) Western analysis of STAT1, P-STAT1 and β-actin in cells treated with recombinant IFN-β. (D) Western analysis of viral proteins and P-STAT1 in IFN-responsive VCaP and RWPE-1 cells infected with SFV-VA7 (MOI=10, 18 h pi).
LNCaP and 22Rv1 prostate cancer cells have dysfunctional JAK/STAT signaling pathway
We have observed previously that poorly IFN-I responsive tumours represent susceptible targets to SFV-VA7 (Ruotsalainen et al, 2012, 2015). Thus, we next studied IFN-I responsiveness of the malignant and nonmalignant prostate cells. As expected, IFN-I treatment induced robust STAT1 phosphorylation in RWPE-1 and positive control cell line Vero(B). In contrast, out of the three studied prostate cancer cell lines LNCaP, VCaP and 22Rv1 only VCaP displayed STAT1 phosphorylation upon IFN-β treatment (Figure 1C). Whereas both RWPE-1 and VCaP cells were type I IFN responsive, only the nonmalignant cells were resistant to oncolytic infection. Thus, we proceeded to further analyse SFV-VA7 replication in these cells. Interestingly, SFV-VA7 infection itself did not induce STAT1 phosphorylation in RWPE-1 cells and only weakly in VCaP cells (Figure 1D). In line with the poor productive infection (Figure 1A), RWPE-1 cells also contained markedly less viral envelope proteins after infection, but interestingly highly similar levels of non-structural protein 3 (nsp3) (Figure 1D).
SFV-VA7 eradicates subcutaneous and orthotopic LNCaP tumours in vivo
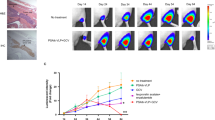
We next analysed the oncolytic potency of SFV-VA7 in the LNCaP prostate cancer model in vivo. LNCaP was chosen due to its unresponsiveness to type I IFN, which was found to be the predominant phenotype in the tested malignant cells. In addition, according to literature, LNCaP is the most widely used cell line tested in oncolytic virotherapy in vivo and hence the obtained results were deemed readily comparable to those reported from earlier studies. Strikingly, progressively growing subcutaneous tumours in nude mice started shrinking after intraperitoneal injection of SFV-VA7 and disappeared in 24 days after treatment (Figure 2A and B). Importantly, high virus loads could be detected in the tumours 72 h after infection both by immunohistochemistry (Figure 2C) and plaque titration (Figure 2D). Despite the known neurotropism of SFV, viral loads in brain were low and the infected mice did not display clinical symptoms (Figure 2D).
Intraperitoneally administered SFV-VA7 homes into subcutaneous LNCaP tumours and eradicates them. Tumour volumes (mean+range) measured in (A) untreated mice (n=5) and (B) mice treated with single intraperitoneal dose of SFV-VA7 (n=8, 1 × 106 PFU). (C) Representative immunohistochemical staining of LNCaP tumour samples collected 72 h pi. Virus antigen stained brown. Scale bar 200 μm. (D) Titration of SFV-VA7 from collected tumour and brain samples 72 h pi. Star: not detected.
Finally, we tested SFV-VA7 therapy efficacy in a clinically relevant setting by treating orthotopic LNCaP –tumours in nude mice. Tumour burden was quantified by measuring serum levels of secreted human prostate-specific antigen (PSA). Experiment end point was set to 20 ng ml−1 of PSA, which was reached without treatment in ∼30 days after first detectable PSA correlating with notable tumour mass in the prostate. Compellingly, all peritoneally infected mice experienced rapid and stable reduction in the serum PSA (Figure 3). SFV-VA7-treated mice were killed 90 days after LNCaP cell inoculation, and post mortem examination revealed no evidence of residual tumour mass.
Discussion
Intratumoural administration of oncolytic virus has been shown to be effective against prostate cancer xenografts (Schenk et al, 2014; Mansfield et al, 2016). Systemic administration would be preferable in targeting advanced disease but has usually shown clearly limited efficacy (Berry et al, 2008; Li et al, 2013). Furthermore, preclinical oncolytic virotherapy research in prostate cancer has relied heavily on subcutaneous tumour models due to the high technical demands of orthotopic inoculation. In one of the few studies available, conditionally replicating adenovirus AxdAdB-3 was reported to modestly improve the survival of SCID mice bearing orthotopic DU145 tumours after intratumoural administration (Satoh et al, 2007). In another study, macrophage-based delivery of an oncolytic adenovirus in combination with docetaxel and radiation therapy showed significantly increased survival in orthotopic LNCaP model (Muthana et al, 2013).
Here we report that all tested prostate cancer cell lines (LNCaP, VCaP and 22Rv1) were efficiently infected and killed in vitro by SFV-VA7, but nonmalignant RWPE-1 cells proved resistant. While the mechanism of RWPE-1 resistance to SFV-VA7 infection warrants further study, high viral nsp3 protein levels after infection indicate that the inhibition takes place after viral entry and early expression of non-structural proteins.
A single peritoneal administration of SFV-VA7 resulted in a 100% cure rate both in subcutaneous and orthotopic LNCaP tumour models, thus suggesting that SFV-VA7 is a novel and highly promising option for treatment of prostate cancer and supporting early clinical testing. The fact that the used mice were immunosuppressed poses a potential caveat in clinical translation. However, earlier we have shown that SFV-VA7 could destroy type I IFN unresponsive mouse colorectal tumours with equal efficiency in immunocompetent and immunosuppressed hosts (Ruotsalainen et al, 2015). Notably, both hormone responsive LNCaP and hormone unresponsive 22Rv1 displayed poor IFN-I responsiveness and were highly susceptible to SFV-VA7 lysis. This result is in line with earlier reports of IFN-I defects in a proportion of human prostate tumours (Danziger et al, 2016) and implies that SFV-VA7 could possibly be utilised as a salvage therapy in castration resistant prostate cancer.
Change history
27 June 2017
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Berry LJ, Au GG, Barry RD, Shafren DR (2008) Potent oncolytic activity of human enteroviruses against human prostate cancer. Prostate 68: 577–587.
Danziger O, Shai B, Sabo Y, Bacharach E, Ehrlich M (2016) Combined genetic and epigenetic interferences with interferon signaling expose prostate cancer cells to viral infection. Oncotarget 7: 52115–52134.
Delwar Z, Zhang K, Rennie PS, Jia W (2016) Oncolytic virotherapy for urological cancers. Nat Rev Urol 13: 334–352.
Heikkilä JE, Vähä-Koskela MJV, Ruotsalainen JJ, Martikainen MW, Stanford MM, McCart JA, Bell JC, Hinkkanen AE (2010) Intravenously administered alphavirus vector VA7 eradicates orthotopic human glioma xenografts in nude mice. PLoS One 5: e8603.
Ketola A, Hinkkanen A, Yongabi F, Furu P, Määttä A-M, Liimatainen T, Pirinen R, Björn M, Hakkarainen T, Mäkinen K, Wahlfors J, Pellinen R (2008) Oncolytic Semliki forest virus vector as a novel candidate against unresectable osteosarcoma. Cancer Res 68: 8342–8350.
Li H, Nakashima H, Decklever TD, Nace RA, Russell SJ (2013) HSV-NIS, an oncolytic herpes simplex virus type 1 encoding human sodium iodide symporter for preclinical prostate cancer radiovirotherapy. Cancer Gene Ther 20: 478–485.
Mansfield DC, Kyula JN, Rosenfelder N, Chao-Chu J, Kramer-Marek G, Khan AA, Roulstone V, McLaughlin M, Melcher AA, Vile RG, Pandha HS, Khoo V, Harrington KJ (2016) Oncolytic vaccinia virus as a vector for therapeutic sodium iodide symporter gene therapy in prostate cancer. Gene Ther 23: 357–368.
Martikainen M, Niittykoski M, von und zu Fraunberg M, Immonen A, Koponen S, van Geenen M, Vähä-Koskela M, Ylösmäki E, Jääskeläinen JE, Saksela K, Hinkkanen A (2015) MicroRNA-attenuated clone of virulent semliki forest virus overcomes antiviral type i interferon in resistant mouse CT-2A glioma. J Virol 89: 10637–10647.
Muthana M, Rodrigues S, Chen Y-Y, Welford A, Hughes R, Tazzyman S, Essand M, Morrow F, Lewis CE (2013) Macrophage delivery of an oncolytic virus abolishes tumor regrowth and metastasis after chemotherapy or irradiation. Cancer Res 73: 490–495.
Määttä A-M, Mäkinen K, Ketola A, Liimatainen T, Yongabi FN, Vähä-Koskela M, Pirinen R, Rautsi O, Pellinen R, Hinkkanen A, Wahlfors J (2008) Replication competent Semliki Forest virus prolongs survival in experimental lung cancer. Int J Cancer 123: 1704–1711.
Ruotsalainen J, Martikainen M, Niittykoski M, Huhtala T, Aaltonen T, Heikkilä J, Bell J, Vähä-Koskela M, Hinkkanen A (2012) Interferon-β sensitivity of tumor cells correlates with poor response to VA7 virotherapy in mouse glioma models. Mol Ther 20: 1529–1539.
Ruotsalainen JJ, Kaikkonen MU, Niittykoski M, Martikainen MW, Lemay CG, Cox J, De Silva NS, Kus A, Falls TJ, Diallo J-S, Le Boeuf F, Bell JC, Ylä-Herttuala S, Hinkkanen AE, Vähä-Koskela MJ (2015) Clonal variation in interferon response determines the outcome of oncolytic virotherapy in mouse CT26 colon carcinoma model. Gene Ther 22: 65–75.
Satoh M, Wang H, Ishidoya S, Abe H, Moriya T, Hamada H, Arai Y (2007) Oncolytic virotherapy for prostate cancer by E1A, E1B mutant adenovirus. Urology 70: 1243–1248.
Schenk E, Essand M, Kraaij R, Adamson R, Maitland NJ, Bangma CH (2014) Preclinical safety assessment of Ad[I/PPT-E1A], a novel oncolytic adenovirus for prostate cancer. Hum Gene Ther Clin Dev 25: 7–15.
Siegel RL, Miller KD, Jemal A (2016) Cancer statistics, 2016. CA Cancer J Clin 66: 7–30.
Tuomela J, Valta M, Seppänen J, Tarkkonen K, Väänänen HK, Härkönen P (2009) Overexpression of vascular endothelial growth factor C increases growth and alters the metastatic pattern of orthotopic PC-3 prostate tumors. BMC Cancer 9: 362.
Vähä-Koskela MJV, Kallio JP, Jansson LC, Heikkilä JE, Zakhartchenko VA, Kallajoki MA, Kähäri V-M, Hinkkanen AE (2006) Oncolytic capacity of attenuated replicative semliki forest virus in human melanoma xenografts in severe combined immunodeficient mice. Cancer Res 66: 7185–7194.
Vähä-Koskela MJV, Tuittila MT, Nygårdas PT, Nyman JK-E, Ehrengruber MU, Renggli M, Hinkkanen AE (2003) A novel neurotropic expression vector based on the avirulent A7(74) strain of Semliki Forest virus. J Neurovirol 9: 1–15.
Acknowledgements
This research was funded by Emil Aaltonen Foundation and Medicinska Understödsföreningen Liv och Hälsa.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License.
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Martikainen, M., Ruotsalainen, J., Tuomela, J. et al. Oncolytic alphavirus SFV-VA7 efficiently eradicates subcutaneous and orthotopic human prostate tumours in mice. Br J Cancer 117, 51–55 (2017). https://doi.org/10.1038/bjc.2017.151
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2017.151
Keywords
This article is cited by
-
Tutorial: design, production and testing of oncolytic viruses for cancer immunotherapy
Nature Protocols (2024)
-
Viruses as tools in gene therapy, vaccine development, and cancer treatment
Archives of Virology (2022)