Abstract
Background:
Pancreatic cancer risk is elevated among testicular cancer (TC) survivors. However, the roles of specific treatments are unclear.
Methods:
Among 23 982 5-year TC survivors diagnosed during 1947–1991, doses from radiotherapy to the pancreas were estimated for 80 pancreatic cancer patients and 145 matched controls. Chemotherapy details were recorded. Logistic regression was used to estimate odds ratios (ORs).
Results:
Cumulative incidence of second primary pancreatic cancer was 1.1% at 30 years after TC diagnosis. Radiotherapy (72 (90%) cases and 115 (80%) controls) was associated with a 2.9-fold (95% confidence interval (CI) 1.0–7.8) increased risk. The OR increased linearly by 0.12 per Gy to the pancreas (P-trend<0.001), with an OR of 4.6 (95% CI 1.9–11.0) for ⩾25 Gy vs <25 Gy. Radiation-related risks remained elevated ⩾20 years after TC diagnosis (P=0.020). The risk increased with the number of cycles of chemotherapy with alkylating or platinum agents (P=0.057), although only one case was exposed to platinum.
Conclusions:
A dose–response relationship exists between radiation to the pancreas and subsequent cancer risk, and persists for over 20 years. These excesses, although small, should be considered when radiotherapy with exposure to the pancreas is considered for newly diagnosed patients. Additional data are needed on the role of chemotherapy.
Similar content being viewed by others
Main
The incidence of testicular cancer (TC), the most common malignancy affecting males aged 15–34 years in the United States and Europe (McGlynn et al, 2003; Garner et al, 2005), has steadily increased over the past 30 years (Chia et al, 2010). As a result of the introduction of radiotherapy in the 1950s and cisplatin-based combination chemotherapy in the 1970s (Einhorn and Donohue, 1977), TC is currently among the most curable solid tumours, with 10-year relative survival reaching 95% (Biggs et al, 2007; Verdecchia et al, 2007).
Previous studies of TC survivors have demonstrated increased risks for treatment-related second solid malignancies, beginning 10–15 years after initial diagnosis. Pancreatic cancer is of particular concern among TC survivors as standardised incidence ratios from registry-based studies have been consistently elevated by two- to four-fold (Van Leeuwen et al, 1993; Travis et al, 1997; Kollmannsberger et al, 1999; Travis et al, 2005; Robinson et al, 2007; Horwich et al, 2014), with patterns of risk consistent with the late effects of radiotherapy. Pancreatic cancer is the fourth most common cause of cancer death in the general US population, with an overall 5-year relative survival of 5.8% (Siegel et al, 2015). This high fatality rate and the lack of data on the effect of radiation doses from treatment for prior cancers highlight the importance of assessing pancreatic cancer risk after abdominal radiotherapy (Carr et al, 2002; Dores et al, 2014). However, no previous study has examined the effects of radiation dose and specific chemotherapy agents on pancreatic cancer risk after TC. Therefore, we performed a case–control study nested in an international cohort of 5-year survivors of TC to evaluate treatment-related pancreatic cancer risk based on estimated radiation doses to the pancreas and cumulative amounts of chemotherapeutic agents.
Materials and methods
Patient selection
We studied 23 982 5-year survivors of histologically confirmed TC as their first primary cancer who were diagnosed between 1947 and 1991 and identified from 6 population-based cancer registries (Sweden, Denmark, Norway, Ontario (Canada), Finland, and Iowa (USA)) or diagnosed at one of the main hospitals in the Netherlands (Van den Belt-Dusebout et al, 2009). The TC patients with a prior history of non-melanoma skin cancer were not excluded as such cancers were not consistently recorded in the cancer registries during the study period. We observed 98 cases of second primary invasive pancreatic cancer diagnosed during 1965–2004. Medical records were obtained for 81 cases (83%). Most of the 17 pancreatic cancer patients without medical records were diagnosed before 1970. We randomly selected two controls per case (N=162) who survived TC without a second cancer at least as long as the corresponding case and individually matched the case by registry, birth date, and calendar year of TC diagnosis (both within 5 years). Medical records were located for 135 controls (83%). To reach the target of 2 controls per case, we selected additional controls, relaxing the matching criteria when necessary – with partial success as very old hospital records had often been destroyed. Eventually, we included a total of 145 controls for 80 cases (one additional case was excluded because no matched controls were available) (Table 1).
The study was approved by either the institutional review boards in each centre or by the Data Inspectorate of participating countries, and exempted from review by The Netherlands Cancer Institute and the National Cancer Institute because only existing de-identified data were used.
Data collection
Details on TC diagnosis and treatment as well as patient demographics were abstracted from available records using standardised forms. Medical and pathology records were reviewed for pancreatic cancer cases to confirm the diagnosis and determine tumour location (head, body, tail). Data on TC chemotherapy were abstracted for dates and routes of administration, regimens, number of cycles, drugs, and doses. Cumulative doses (mg m−2) were calculated for individual agents. Because of similarities in mechanisms of action, platinum compounds were combined with alkylating agents into a category of alkylating-like agents, although they form covalent metal DNA adducts instead of alkylating DNA (Brunton et al, 2011).
Abstracted radiotherapy details included dates of administration, beam energy, delivered dose, field location, and configuration. Patients were generally treated with dog-leg fields (para-aortic and ipsilateral iliac nodes) or para-aortic fields only. Daily target doses were 1.8–2.0 Gy resulting in cumulative doses ranging between 25 and 50 Gy. Dose was calculated to 129 points in the pancreas (divided as 54, 50, and 25 points in the head, body, and tail, respectively) based on a typical pancreas configuration (Perez et al, 2008), using a custom-designed dose program, based on measurements in water and anthropomorphic phantoms constructed of tissue-equivalent material (Stovall et al, 2006). Analyses of radiotherapy risks used the mean dose to the pancreas tumour location (same location in matched controls), specified as head, body, and tail. For 13 (16%) cases with unknown tumour location, analyses used mean dose to the pancreas head where the majority of pancreatic tumours with known subsite (82%) were located.
Statistical analysis
Cumulative incidence of second primary invasive pancreatic cancer in the population-based cohort (that is, excluding the Netherlands) was calculated with death and other second cancers (except non-melanoma skin cancer) as competing risks (Gooley et al, 1999). The relative risk of pancreatic cancer was estimated using odds ratios (ORs) and 95% confidence intervals (CIs) derived from conditional logistic regression (Breslow and Day, 1980), comparing exposure histories among cases with those of matched controls. Radiotherapy received within 5 years of pancreatic cancer diagnosis (or equivalent date in controls) was not included because it was unlikely to have contributed to the pancreatic cancer. The radiation dose–response relationship was evaluated using dose as a categorical variable. In addition, the excess odds ratio (EOR) per Gy was estimated by the linear additive dose–response model OR=EXP(ΣjαjXj)[1+βD], where D is radiation dose in Gy, β is the EOR per Gy, and the Xj are covariates (for example, chemotherapy) with corresponding log ORs αj. Departure from linearity was evaluated by a likelihood ratio test of the null hypothesis γ=0 in a model including dose as an exponential factor OR=EXP(ΣjαjXj)[1+βD × EXP(γD)], where γ indicates downward (γ<0) or upward curvature (γ>0) in the EOR per Gy. Patients with missing radiotherapy dose were included as a separate category.
The ORs for chemotherapy were assessed by having ever vs never received any chemotherapy or any alkylating agent-containing chemotherapy adjusted for radiation dose (0, >0–24.9, 25.0–29.9, 30.0–34.9, 35.0–39.9, 40.0–44.9, and ⩾45.0 Gy). The ORs were also calculated according to the number of alkylating agent-containing chemotherapy cycles (categorical variable), and trend tests were based on the number of alkylating agent-containing cycles (continuous variable) in an additive model, like for continuous dose as shown above. Heterogeneity in radiation-related risks among patient subgroups under a multiplicative model was evaluated by comparing the goodness of fit of models including separate ORs and EORs for each subgroup with models including a single estimate, respectively. To evaluate the joint effect of radiotherapy (radiation dose <25 vs >=25 Gy) and chemotherapy (no vs yes), deviances of multiplicative and additive models were compared with those of more general models that included interaction terms. Attributable risks were calculated by averaging the quantities [dose × EOR per Gy]/[1+(dose × EOR per Gy)] over cases with known dose. The SAS (version 9.2; SAS Institute, Cary, NC, USA) and EPICURE (Preston et al, 1993) software were used.
Results
The cumulative incidence of second primary invasive pancreatic cancer in the population-based cohort was 0.14% (95% CI 0.07–0.20%) and 1.08% (95% CI 0.83–1.34%), respectively, at 15 and 30 years after TC diagnosis. Of all pancreatic cancers (median age at diagnosis, 61 years; range, 41–81 years), 48% occurred ⩾20 years after TC diagnosis (median, 20 years; range, 6–38 years), and the majority were located in the head of the pancreas (69%). The median age at diagnosis is lower than that reported in the US population (73 years) during 1973–2002 (Lau et al, 2010) or peak occurrence reported in Denmark (70–74 years) during 1978–2003 among males and females (Teiblum et al, 2009). In both the United States and Denmark, tumours of the pancreatic head predominated in the general population (Teiblum et al, 2009; Lau et al, 2010).
Among pancreas cancer cases and controls, median age at TC diagnosis was 40 years (range, 19–73 years), 68% had been treated for seminoma, and 94% had stage I or II disease (Table 1). The TC treatment included surgery and radiotherapy (81% cases, 74% controls); surgery, radiotherapy, and chemotherapy (8% cases, 6% controls); surgery only (6% cases, 15% controls); or surgery and chemotherapy (4% cases, 6% controls).
Two common fields resulted in average radiation doses of ∼30 Gy to the head and body of the pancreas: dog-leg (40% of patients who received radiotherapy) and para-aortic fields (35%) (Table 2). Abdominal (13%) and non-central para-aortic fields (11%) resulted in average doses to the head and body of the pancreas of 15–20 Gy. For all other radiation fields (including mediastinum, pelvis, mantle, testes, neck, or supraclavicular area), the pancreas received on average <2 Gy to any pancreas subsite.
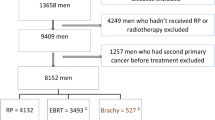
Patients who received radiotherapy had a 2.9-fold (95% CI 1.0–7.8) increased risk of pancreatic cancer compared with patients who did not receive radiotherapy (Table 3). Risk increased with increasing dose to the pancreatic tumour site (P-trend<0.001), with ORs of 0.9 (95% CI 0.2–3.2), 2.5 (95% CI 0.6–11.4), 4.5 (95% CI 1.3–15.6), 8.1 (95% CI 1.8–35.5), 2.3 (95% CI 0.6–9.7), and 7.1 (95% CI 1.5–33.2) for >0–24.9, 25.0–29.9, 30.0–34.9, 35.0–39.9, 40.0–44.9, and ⩾45.0 Gy, respectively, compared with no radiotherapy. The EOR per Gy was 0.12 (95% CI 0.03–0.42) and consistent with linearity (test of nonlinearity, P=0.127), although power to detect nonlinearity is limited (Figure 1). Among patients treated with radiotherapy, the estimated percentage of pancreatic cancers attributable to radiotherapy was 70% (95% CI 44–83%) that increased to 81% (95% CI 52–94%) for patients who received ⩾25 Gy to the pancreas.
Radiation dose–response relationship for pancreatic cancer following testicular cancer based on 80 cases and 145 controls. Filled circles and error bars indicate odds ratios and 95% confidence intervals for categories of dose to the pancreatic tumour location in cases and a corresponding location in controls (as shown in Table 3) plotted at the mean dose per category. The slope of the solid line is the linear excess odds ratio (EOR) per Gy (0.12, 95% CI 0.03–0.42).
Radiation-related risk estimates were similar or increased slightly when adjusted for the number of alkylating agent-containing chemotherapy cycles, with an EOR per Gy of 0.20 (95% CI 0.05–0.88) (Supplementary Table 1). In addition, there was no evidence that radiation-related risks were modified by chemotherapy or vice versa. Interaction terms between both binary and continuous indicators of chemotherapy and radiotherapy were nonsignificant (P>0.5; Tables 3 and 4).
The OR for pancreas cancer among TC patients given chemotherapy (Supplementary Table 2), adjusted for radiation dose, was 1.4 (95% CI 0.5–3.7; Table 3). For patients treated with alkylating-agent containing chemotherapy, the OR was 2.2 (95% CI 0.7–6.9), based on 8 cases and 11 controls. Risk reached 3.5-fold for ⩾5 cycles (2 cases, 4 controls, 95% CI 0.4–32.4), with a borderline significant trend (P-trend=0.057). Six of 154 seminoma patients (4%, chemotherapy unknown for one seminoma patient) and 18 of 65 non-seminoma patients (28%) received chemotherapy. For alkylating agent-containing chemotherapy, corresponding numbers were 4% and 18%, respectively. For the number of alkylating agent-containing chemotherapy cycles, the association was pronounced among non-seminoma patients, where ORs increased up to 10.9 (95% CI 1.0–117.6) for ⩾5 cycles (P-trend=0.034), whereas there was no apparent association for patients with other histology (P-trend=0.298) (data not shown).
We observed no evidence of heterogeneity of radiation-related risks for cancers in the head of the pancreas (EOR per Gy=0.10) vs those in the body or tail (EOR per Gy=0.02, P-homogeneity=0.330; Table 4). Power was limited as only 11 pancreatic cancers were located in the body or tail. Furthermore, risks appeared homogeneous by age at and year of diagnosis of TC or pancreatic cancer and by TC histology. There was a significant radiation dose–response among patients who did not receive chemotherapy (EOR per Gy=0.15, P<0.001), the largest treatment group (89% of cases, 89% of controls). Risks remained significantly increased ⩾20 years after exposure (EOR per Gy=0.07, P=0.020).
We performed sensitivity analyses to evaluate the robustness of our findings. Results were similar when each registry was excluded one at a time (range EOR per Gy, 0.08–0.21). All major results were only minimally affected when we excluded controls who did not strictly match the case within 5 years for date of birth (N=2), year of TC diagnosis (N=1), or follow-up period (N=3). We also evaluated obesity, a pancreatic cancer risk factor (Ryan et al, 2014; Maisonneuve and Lowenfels, 2015), among patients with recorded body mass index (BMI; 44 cases and 60 controls). The unadjusted EOR per Gy (0.10) was similar after adjustment for continuous or categorical BMI (kg m−2) at TC diagnosis (0.10 and 0.11, respectively).
Discussion
In an international nested case–control study within a cohort of 23 982 5-year survivors of TC treated between 1947 and 1991, we observed a significant dose–response relationship between cumulative radiation dose to the pancreas and risk of pancreatic cancer. Elevated radiation-associated risk persisted for more than two decades. The TC survivor population is of interest for the high proportion of patients who received abdominal radiotherapy in the absence of chemotherapy, thus permitting an unconfounded evaluation of the role of high-dose ionising radiation in pancreatic carcinogenesis.
Our study is among the first to establish a radiation dose–response relationship for second primary pancreatic cancer among cancer patients not treated with alkylating agent-containing chemotherapy, with an EOR per Gy of 0.15 (95% CI 0.03–0.66). In an earlier report of pancreatic cancer among Hodgkin’s lymphoma survivors (Dores et al, 2014), the number of patients treated with radiation in the absence of alkylating agent-containing chemotherapy was too small (10 cases and 33 controls) to establish a dose–response in this group alone. The overall EOR per Gy of 0.10 (95% CI 0.02–0.42) was similar between studies. Our current results add to the evidence for a causal association between radiation and pancreatic cancer.
Significant radiation dose-response relationships for pancreatic cancer have been observed among patients exposed to medical radiation for indications other than cancer treatment (Weiss et al, 1994; Ryan et al, 2014; Maisonneuve and Lowenfels, 2015) with doses to the pancreas varying widely but typically less than ∼15 Gy. Among atomic bomb survivors, who received doses generally under 4 Gy, a nonsignificant positive association (EOR per Gy=0.26, 90% CI −0.07–0.68) was observed in the most recent analysis of those data (Travis et al, 2003).
Although numbers were quite small, we observed a suggestive association between alkylating agent-containing chemotherapy and subsequent pancreatic cancer, particularly among non-seminoma patients who were more likely to receive chemotherapy. Radiation-related risks did not appear to differ according to the receipt of alkylating agents. However, these findings should be interpreted cautiously as numbers were too small to compare patients with substantial exposure to radiation and alkylating agents with patients with neither treatment. In the only other study of second primary pancreatic cancer with detailed information on antecedent radiation and chemotherapy, Dores et al (2014) observed especially high risks (18-fold) of pancreatic cancer among Hodgkin’s lymphoma survivors who received both subdiaphragmatic radiotherapy and ⩾6 cycles of alkylating agent-containing chemotherapy.
Although the proportion of TC patients receiving radiotherapy has decreased substantially during recent decades, our results apply to the large number of TC survivors who have been effectively treated with radiotherapy in the past and remain alive. In view of the increasing incidence of TC in the past decades (Chia et al, 2010) and the availability of curative therapy, TC survivors currently comprise ∼4% of all male US cancer survivors (DeSantis et al, 2010) and ∼5% of all male cancer survivors in the Nordic countries (Engholm et al, 2010). In addition, currently up to one-third of seminoma patients may receive radiotherapy (DeSantis et al, 2010; Vossen et al, 2012; Kohut et al, 2014). The cumulative radiotherapy target volume dose decreased from 40 to 30 Gy after 1980 and further to 20 Gy since ∼1990 (Jones et al, 2005; Hoffman et al, 2008; Yu et al, 2009; Schmoll et al, 2009; Arvold et al, 2012; National Comprehensive Cancer Network (NCCN), 2013; Comprehensive Cancer Center Netherlands, 2014). Among non-seminoma patients, cumulative radiation doses were 45–50 Gy before 1980, whereas radiotherapy was rarely used after 1980, when cisplatin became available (Einhorn and Donohue, 1977). Alkylating agent-containing chemotherapy may also increase pancreatic cancer risk, as observed in our study and one other series (Dores et al, 2014), although results here are based on small numbers.
A major strength of our study is the case–control design nested in an international cohort of 23 982 TC patients, most of them followed for more than three decades, with collection of detailed clinical and demographic data. We performed individual dosimetry and estimated the radiation dose to the tumour subsite that likely led to accurate dose estimates, although uncertainties remain because of, among others, computed tomography-based plans for radiotherapy. Our study has several limitations. Despite the large study base, the small number of patients treated with platinum-based chemotherapy (1 case and 7 controls) did not permit adequate statistical power to evaluate this modality. Furthermore, the inability to obtain medical records was more common for patients diagnosed before 1970, and thus a larger number of cases from registries that were established in earlier years could not be included. However, differential ascertainment of medical records is unlikely to introduce bias as controls were matched to cases on year of TC diagnosis, registry, and birth date. Adjustment for BMI at TC diagnosis among patients with available data did not substantially change radiation risk estimates. As information on other established pancreatic cancer risk factors such as smoking, Helicobacter pylori infection, blood group, diabetes mellitus, and chronic pancreatitis (International Agency for Research on Cancer, 2004; Ryan et al, 2014; Maisonneuve and Lowenfels, 2015) was not available in this retrospective study, we were unable to adjust our analyses accordingly. It is unlikely, however, that confounding of therapy-related risks by the aforementioned factors exists, as to our knowledge they do not influence clinical decisions with regard to TC treatments. The effect of increased BMI as an intermediate factor in increased pancreas cancer risk caused by therapy for TC could not be evaluated because of the lack of post-treatment BMI data.
Our findings add to the knowledge of potential adverse sequelae associated with TC treatment. Although second pancreatic cancer is a rare complication of TC therapy, it is highly fatal. In our study, median survival was 4 months among the 77 pancreatic cancer cases who were known to have died. The results may also be applicable to patients with cancers at other sites in whom similar abdominal regions may be irradiated today (Halperin et al, 2013; Teepen et al, 2016). Consideration of administering radiotherapy with curative intent should include an evaluation of the radiation-related pancreatic cancer risk that may persist for >20 years, although the small magnitude of any excess risk must be weighed against the potential benefits of radiotherapy.
Change history
27 September 2016
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Arvold ND, Catalano PJ, Sweeney CJ, Hoffman KE, Nguyen PL, Balboni TA, Fossa SD, Travis LB, Beard CJ (2012) Barriers to the implementation of surveillance for stage I testicular seminoma. Int J Radiat Oncol Biol Phys 84 (2): 383–389.
Biggs ML, Schwartz SM (2007) Cancer of the testis. In SEER Survival Monograph: Cancer Survival Among Adults: U.S. SEER Program, 1988–2001, Patient and Tumor Characteristics LA Ries, JL Young, GE Keel, MP Eisner, YD Lin, MJ Horner, (eds) National Cancer Institute, SEER Program, NIH Pub. No. 07-6215: Bethesda, MD.
Breslow NE, Day NE (1980) Statistical Methods in Cancer Research Vol. 1 The Analysis of Case-Control Studies. International Agency for Research on Cancer: Lyon, France.
Brunton LL, Chabner BA, Knollmann BC (eds) (2011) Goodman and Gilman's The Pharmacological Basis of Therapeutics 12th edn. McGraw Hill.
Carr ZA, Kleinerman RA, Stovall M, Weinstock RM, Griem ML, Land CE. (2002) Malignant neoplasms after radiation therapy for peptic ulcer. Radiat Res 157: 668–677.
Chia VM, Quraishi SM, Devesa SS, Purdue MP, Cook MB, McGlynn KA (2010) International trends in the incidence of testicular cancer, 1973-2002. Cancer Epidemiol Biomarkers Prev 19 (5): 1151–1159.
Comprehensive Cancer Center Netherlands (2014) Kankerzorg in beeld – rapportage testiscarcinoom. Available at:. https://www.iknl.nl/docs/default-source/KIB-rapportages/portfolio_kib_testiscarcinoom.pdf?sfvrsn=2 [in Dutch].
DeSantis CE, Lin CC, Mariotto AB, Siegel RL, Stein KD, Kramer JL, Alteri R, Robbins AS, Jemal A (2010) Cancer treatment and survivorship statistics, 2014. CA Cancer J Clin 64 (4): 252–271.
Dores GM, Curtis RE, van Leeuwen FE, Stovall M, Hall P, Lynch CF, Smith SA, Weathers RE, Storm HH, Hodgson DC, Kleinerman RA, Joensuu H, Johannesen TB, Andersson M, Holowaty EJ, Kaijser M, Pukkala E, Vaalavirta L, Fossa SD, Langmark F, Travis LB, Fraumeni JF Jr, Aleman BM, Morton LM, Gilbert ES (2014) Pancreatic cancer risk after treatment of Hodgkin lymphoma. Ann Oncol 25 (10): 2073–2079.
Einhorn LH, Donohue J (1977) Cis-diamminedichloroplatinum, vinblastine, and bleomycin combination chemotherapy in disseminated testicular cancer. Ann Intern Med 87 (3): 293–298.
Engholm G, Ferlay J, Christensen N, Bray F, Gjerstorff ML, Klint A, Kotlum JE, Olafsdottir E, Pukkala E, Storm HH (2010) NORDCAN — a Nordic tool for cancer information, planning, quality control and research. Acta Oncol 49 (5): 725–736.
Garner MJ, Turner MC, Ghadirian P, Krewski D (2005) Epidemiology of testicular cancer: an overview. Int J Cancer 116 (3): 331–339.
Gooley TA, Leisenring W, Crowley J, Storer BE (1999) Estimation of failure probabilities in the presence of competing risks: new representations of old estimators. Stat Med 18 (6): 695–706.
Halperin EC, Brady LW, Perez CA, Wazer DE (2013) Perez & Brady's Principles and Practice of Radiation Oncology 6th edn Wolters Kluwer: Amsterdam, The Netherlands.
Hoffman KE, Chen MH, Punglia RS (2008) Influence of year of diagnosis, patient age, and sociodemographic status on recommending adjuvant radiation treatment for stage I testicular seminoma. J Clin Oncol 26: 3937–3942.
Horwich A, Fossa S, Huddart R, Dearnaley DP, Stenning S, Aresu M, Bliss JM, Hall E (2014) Second cancer risk and mortality in men treated with radiotherapy for stage I seminoma. Br J Cancer 110: 256–263.
International Agency for Research on Cancer (2004) Tobacco smoke and involuntary smoking. Monographs on the Evaluation of Carcinogenic Risks to Humans Vol. 83. World Health Organization: Lyon, France.
Jones WG, Fossa SD, Mead GM, Roberts JT, Sokal M, Horwich A, Stenning SP (2005) Randomized trial of 30 versus 20 Gy in the adjuvant treatment of stage I testicular seminoma: a report on Medical Research Council Trial TE18, European Organisation for the Research and Treatment of Cancer Trial 30942 (ISRCTN18525328). J Clin Oncol 23 (6): 1200–1208.
Kohut RM Jr, Minnillo BJ, Kypriotakis G, Abouassaly R, Zhu H (2014) Changes in adjuvant therapy utilization in stage I seminoma: are they enough to prevent overtreatment? Urology 84 (6): 1319–1324.
Kollmannsberger C, Hartmann JT, Kanz L, Bokemeyer C (1999) Therapy-related malignancies following treatment of germ cell cancer. Int J Cancer 83: 860–863.
Lau MK, Davila JA, Shaib YH (2010) Incidence and survival of pancreatic head and body and tail cancers: a population-based study in the United States. Pancreas 39 (4): 458–462.
Maisonneuve P, Lowenfels AB (2015) Risk factors for pancreatic cancer: a summary review of meta-analytical studies. Int J Epidemiol 44 (1): 186–198.
McGlynn KA, Devesa SS, Sigurdson AJ, Brown LM, Tsao L, Tarone RE (2003) Trends in the incidence of testicular germ cell tumors in the United States. Cancer 97 (1): 63–70.
National Comprehensive Cancer Network (NCCN) (2013) Clinical Practice Guidelines in Oncology Testicular Cancer (Version 1). Available at: www.NCCN.org.
Perez CA, Brady LW, Halperin EC (2008) Perez & Brady's Principles and Practice of Radiation Oncology 5th edn. pp 1574–1575. Lippincott Williams & Wilkins: Philadelphia, PA.
Preston DL, Lubin JH, Pierce DA, McConney ME (1993) Epicure User's Guide. HiroSoft International Corporation: Seattle, WA.
Robinson D, Moller H, Horwich A (2007) Mortality and incidence of second cancers following treatment for testicular cancer. Br J Cancer 96: 529–533.
Ryan DP, Hong TS, Bardeesy N (2014) Pancreatic adenocarcinoma. N Engl J Med 371 (11): 1039–1049.
Schmoll HJ, Jordan K, Huddart R, Laguna MP, Horwich A, Fizazi K, Kataja V ESMO Guidelines Working Group (2009) Testicular seminoma: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol 20 (Suppl 4): 83–88.
Siegel RL, Miller KD, Jemal A (2015) Cancer statistics, 2015. CA Cancer J Clin 65 (1): 5–29.
Stovall M, Weathers R, Kasper C, Smith SA, Travis L, Ron E, Kleinerman R (2006) Dose reconstruction for therapeutic and diagnostic radiation exposures: use in epidemiological studies. Radiat Res 166: 141–157.
Teepen JC, de Vroom SL, van Leeuwen FE, Tissing WJ, Kremer LC, Ronckers CM (2016) Risk of subsequent gastrointestinal cancer among childhood cancer survivors: a systematic review. Cancer Treat Rev 43: 92–103.
Teiblum S, Thygesen LC, Johansen C (2009) Sixty-one years of pancreatic cancer in Denmark from 1943 to 2003: a nationwide study. Pancreas 38 (4): 374–378.
Travis LB, Curtis RE, Storm H, Hall P, Holowaty E, Van Leeuwen FE, Kohler BA, Pukkala E, Lynch CF, Andersson M, Bergfeldt K, Clarke EA, Wiklund T, Stoter G, Gospodarowicz M, Sturgeon J, Fraumeni JF Jr, Boice Jr JD (1997) Risk of second malignant neoplasms among long-term survivors of testicular cancer. J Natl Cancer Inst 89: 1429–1439.
Travis LB, Hauptmann M, Gaul LK, Storm HH, Goldman MB, Nyberg U, Berger E, Janower ML, Hall P, Monson RR, Holm LE, Land CE, Schottenfeld D, Boice JD Jr, Andersson M (2003) Site-specific cancer incidence and mortality after cerebral angiography with radioactive thorotrast. Radiat Res 160 (6): 691–706.
Travis LB, Fossa SD, Schonfeld SJ, McMaster ML, Lynch CF, Storm H, Hall P, Holowaty E, Andersen A, Pukkala E, Andersson M, Kaijser M, Gospodarowicz M, Joensuu T, Cohen RJ, Boice JD Jr, Dores GM, Gilbert ES (2005) Second cancers among 40,576 testicular cancer patients: focus on long-term survivors. J Natl Cancer Inst 97 (18): 1354–1365.
Van den Belt-Dusebout AW, Aleman BM, Besseling G, De Bruin ML, Hauptmann M, Van 't Veer MB, De Wit R, Ribot JG, Noordijk EM, Kerst JM, Gietema JA, Van Leeuwen FE (2009) Roles of radiation dose and chemotherapy in the etiology of stomach cancer as a second malignancy. Int J Radiat Oncol Biol Phys 75 (5): 1420–1429.
Van Leeuwen FE, Stiggelbout AM, Van den Belt-Dusebout AW, Noyon R, Eliel MR, Van Kerkhoff EH, Delemarre JF, Somers R (1993) Second cancer risk following testicular cancer: a follow-up study of 1909 patients. J Clin Oncol 11: 415–424.
Verdecchia A, Francisci S, Brenner H, Gatta G, Micheli A, Mangone L, Kunkler I (2007) Recent cancer survival in Europe: a 2000-02 period analysis of EUROCARE-4 data. Lancet Oncol 8 (9): 784–796.
Vossen CY, Horwich A, Daugaard G, Van Poppel H, Osanto S (2012) Patterns of care in the management of seminoma stage I: results from a European survey. BJU Int 110 (4): 524–531.
Weiss HA, Darby SC, Doll R (1994) Cancer mortality following X-ray treatment for ankylosing spondylitis. Int J Cancer 59 (3): 327–338.
Yu HY, Madison RA, Setodji CM, Saigal CS (2009) Quality of surveillance for stage I testis cancer in the community. J Clin Oncol 27: 4327–4332.
Acknowledgements
We thank Diane Fuchs, Janet Lawler-Heavner, and their staff at Westat, Inc. (Rockville, MD, USA) for administrative assistance in conducting the field studies, and Jeremy Miller (Information Management Services, Silver Spring, MD, USA) for computer programming support. This work was supported by the Intramural Research Program of the National Cancer Institute, National Institutes of Health, Department of Health and Human Services, and National Cancer Institute contracts to Cancer Care Ontario, Toronto, Canada (N01-CP-31157); Danish Cancer Society, Copenhagen, Denmark (N01-CP-31019); Finnish Cancer Registry, Helsinki, Finland (N01-CP-31154); Information Management Services, Inc., Silver Spring, USA (N01-CP-31003); Karolinska Institute, Stockholm, Sweden (N01-CP-31156); University of Iowa, Iowa City, USA (N01-CP-31155); The University of Texas MD Anderson Cancer Center, Houston, USA (N02-CP-55503); and Westat, Inc., Rockville, USA (N02-CP-31136). The Dutch study was also supported by the Lance Armstrong Foundation and the Dutch Cancer Society (Grant No. NKI 04–3068). Dr Travis was supported by National Cancer Institute 1R01 CA 157823–01A1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License.
Supplementary Information accompanies this paper on British Journal of Cancer website
Supplementary information
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Hauptmann, M., Børge Johannesen, T., Gilbert, E. et al. Increased pancreatic cancer risk following radiotherapy for testicular cancer. Br J Cancer 115, 901–908 (2016). https://doi.org/10.1038/bjc.2016.272
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2016.272
Keywords
This article is cited by
-
Association between radiotherapy and risk of second primary malignancies in patients with resectable lung cancer: a population-based study
Journal of Translational Medicine (2023)
-
Quantifying cancer risk from exposures to medical imaging in the Risk of Pediatric and Adolescent Cancer Associated with Medical Imaging (RIC) Study: research methods and cohort profile
Cancer Causes & Control (2022)
-
Testicular cancer in Geneva, Switzerland, 1970–2012: incidence trends, survival and risk of second cancer
BMC Urology (2019)
-
Secondary malignancies after high-dose chemotherapy in germ cell tumor patients: a 34-year retrospective study of the European Society for Blood and Marrow Transplantation (EBMT)
Bone Marrow Transplantation (2018)
-
Risk of diabetes after para-aortic radiation for testicular cancer
British Journal of Cancer (2018)