Abstract
Background:
Phosphoglycerate kinase-1 (PGK1) has been recently documented in various malignancies; however, the molecular mechanisms of the variable PGK1 expression and its clinical significance in terms of survival status remain unclear.
Methods:
Real-time quantitative PCR (real-time qPCR) and western blotting were used to verify PGK1 expression in 46 fresh breast cancer tissues and matched normal tissues. A tissue microarray (TMA) comprising 401 breast cancer tissues and 123 matched normal tissues was investigated by immunohistochemistry for PGK1 expression. Then, the correlation between PGK1 expression and the clinicopathologic features was analysed.
Results:
PGK1 mRNA and protein expression were significantly increased in breast cancer tissues compared with that in normal breast tissues. High PGK1 expression was significantly associated with higher histologic grade (P=0.009) and positive status of ER (P=0.004), Her-2 (P=0.026) and P53 (P=0.012). High levels of PGK1 expression were associated with worse overall survival (OS, P=0.02). Furthermore, patients who underwent paclitaxel chemotherapy with high levels PGK1 expression had shorter OS than did those with low levels of PGK1 expression (P<0.001). Multivariate analysis indicated that PGK1 (P=0.001) was an independent predictor in the patients treated with paclitaxel.
Conclusions:
PGK1 is a prognostic biomarker of chemoresistance to paclitaxel treatment in breast cancer.
Similar content being viewed by others
Main
Breast cancer is the most prevalent cancer in women. It accounted for 29% (232 670) of all new cancers and 15% (40 000) of total cancer deaths in the United States in 2014 (Siegel et al, 2014). The death rate for breast cancer is down from its peak rate by 34% as a result of improvements in early detection and treatment (Berry et al, 2005; Etzioni et al, 2008; Edwards et al, 2010). Despite developments in surgery and new chemotherapy regimens over the last few decades, disease progression typically occurs in advanced breast cancers, likely due to the presence or the development of chemoresistant tumours (Pommerenke et al, 1994). Therefore, the ability to predict a response before commencing chemotherapy would be beneficial to patients who are unlikely to respond to the drugs and could save them from unnecessary drug toxicity. Although mechanisms of chemoresistance are multiple, it has long been recognised that hypoxic cells, such as those found in the highly hypoxic core areas of tumours, are the most chemoresistant cells (Kessler et al, 2010; Monti and Gariboldi, 2011). To survive these difficult conditions, lacking O2, cells necessarily adopt strategies of resistance, such as enhanced glycolysis or overexpression of anti-apoptotic factors (Vaupel and Mayer, 2007; Bertout et al, 2008).
Among the glycolytic enzymes, phosphoglycerate kinase-1 (PGK1) catalyses a crucial step of glycolysis, transferring a phosphate group from 1,3-biphosphoglycerate to ADP, forming ATP and 3-phosphoglycerate (Wang et al, 2007). PGK1 is regulated by hypoxia-inducible factor-1α (Dayan et al, 2006). In addition to its function as a glycolytic pathway enzyme, PGK1 is also believed to be involved in tumour biology (Kabbage et al, 2008), angiogenesis (Lay et al, 2000), DNA replication and repair in mammalian nuclei (Vishwanatha et al, 1992; Popanda et al, 1998) and cancer metastasis (Zieker et al, 2010b). Further, PGK1 is involved in the onset of malignancies, such as pancreatic cancer (Hwang et al, 2006), liver cancer (Ai et al, 2011) and gastric cancer (Zieker et al, 2008; Zieker et al, 2010a). In patients suffering from breast cancer, PGK1 expression has been reported to be increased (Kabbage et al, 2008) and to be associated with Her-2 status (Zhang et al, 2005). Nevertheless, the molecular mechanisms of the variable PGK1 expression and its clinical significance in terms of survival status remain unclear.
Real-time quantitative PCR (real-time qPCR) and western blotting were used to verify PGK1 expression in fresh cancer tissues and their matched normal tissues. To further elucidate the issues, we established a tissue microarray (TMA) comprising 401 breast cancer tissues and 123 matched normal tissues. Then, the correlation between PGK1 expression and the clinicopathologic features was analysed. This study was performed in accordance with the REMARK criteria (McShane et al, 2005).
Materials and methods
Patients and clinical samples
This study used archival material from the Department of Pathology at the Harbin Medical University Cancer Hospital, including tissues from 401 consecutive patients with histologically confirmed breast cancer and 123 matched normal tissue samples, all from 2006. Fresh cancer tissues and their matched normal tissues from 46 patients were collected and stored at −80 °C immediately after resection to extract protein and RNA. The cancer tissues and their matched normal tissues were examined by two pathologists. All of the patients were invasive breast cancer patients. The patients who presented with recurrent tumour, metastatic disease at presentation, bilateral tumours, other previous tumours or those who had previously received neoadjuvant treatment were excluded. All protocols were reviewed and approved by the Ethical Committee of Harbin Medical University in Harbin, China. All patients gave informed consent for the diagnostic procedures and the proposed treatment.
The clinicopathologic characteristics of the 401 breast cancer patients were described in Table 1. The median age of the patients was 49 years (range, 28–78). Among all of the patients, lymph node metastasis (LNM) was present in 224 patients (55.9%) and absent in 177 patients (44.1%). A total of 267 (66.6%) patients were classified as stage I and II and 134 (33.4%) were stage III. Of 401 patients, 129 received standard paclitexel-based combination chemotherapy after surgery consisting of six courses of treatment.
All breast cancer patients were tested for ER, PR, Her-2, P53 and Ki67, assayed in paraffin-embedded, formation-fixed tissue using antibodies against the proteins ER, PR, Her-2, P53 and Ki67 (Zhong shan-Bio Co., Beijing, China). Immunohistochemical (IHC) staining for ER and PR was performed using a conventional detection method and was considered positive if 1% or more of the nuclei in the invasive component of the tumour were stained (Hammond et al, 2010). Positive staining for Her-2 was defined based on the percentage of tumour cells and the intensity of membrane staining. No staining observed or membrane staining of fewer than 10% of the tumour cells was scored as 0. Faint or barely perceptible incomplete membrane staining detected in more than 10% of the tumour cells was scored as 1+. Weak to moderately complete membrane staining observed in more than 10% of the tumour cells was scored as 2+. Strong complete membrane staining observed in more than 10% of the tumour cells was scored as 3+. Scores of 0 to 1+ were regarded as negative and 3+ were regarded as positive (Hsu et al, 2002). We selected a Ki67 index of 14% as the optimal cut point for human visual assessment (Cheang et al, 2009). For P53, positive staining of more than 10% of the tumour cells was defined as positive tumour expression and staining of 10% or fewer of the cells as negative tumour expression (Millar et al, 2011). The patients were categorised into four molecular subtypes based upon previously validated clinicopathological criteria (Goldhirsch et al, 2013).
Follow-up
The clinical and pathological records of all the patients in the study were reviewed periodically. Examinations were performed every 4–6 months for the first 5 years and every 12 months thereafter during the follow-up period. Patients were followed regularly for a minimum of 5 years of follow-up at the Harbin Medical University Cancer Hospital. The clinical records were obtained from the departments providing follow-up care. Survival was calculated in months from the date of diagnosis to the date of whichever of the following occurred first: death, last known to be alive or 20 November 2012, which was the follow-up cut-off date used in our analysis.
RNA preparation and reverse transcription
Total RNA was extracted according to the protocol of TRIzol reagent (Invitrogen Corporation, Beijing, China) after ensuring that the tumour tissue contained more than 75% of tumour cells and had been approved by pathologists in the pathology department. RNA quality and concentration were measured by GeneQuant Pro (GE Healthcare, Buckinghamshire, United Kingdom). cDNA was synthesised from 1.0 μg of total RNA in a 20 μl reaction mixture using the PrimeScript RT Reagent Kit with gDNA eraser (Takara Bio Inc., Otsu, Japan).
Real-time qPCR
Real-time qPCR was performed using the ABI 7500 Fast sequence detection system (Applied Biosystems, Foster City, CA, USA) and FastStart Universal SYBR Green Master (ROX) reagent (Roche Applied Science, Mannheim, Germany) according to the manufacturers’ instructions. The primers of PGK1 were designed as follows: forward, 5′AACCAGAGGATTAAGGCTGC3′; and reverse, 5′GCCTACACAGTCCTTCAAGA3′. GAPDH was applied as the internal reference; its primers were as follows: 5′AACGACCCCTTCATTGAC3′; and reverse, 5′TCCACGACATACTCAGCAC3′. Amplification was performed under the following conditions: 95 °C for 10 min, followed by 40 cycles of 95 °C for 15 s, 60 °C for 60 s. The results of the real-time qPCR experiments were calculated using the 2−ΔΔCt method. Experiments were performed in triplicate for the same reaction.
Western blotting analysis
Frozen tissue samples were homogenised in RIPA buffer consisting of 1% protease inhibitor mixture. The mixture was centrifuged at 12 000 g for 15 min at 4 °C and the supernatant was obtained. Total proteins were quantified. Per sample, 30 μg of protein was separated onto a denaturing polyacrylamide gel containing SDS and transferred to a methanol-activated PVDF filter membrane (Bio-Rad, Hercules, CA, USA). Before immunodetection, membranes were blocked with 5% non-fat dry milk. Primary antibodies, anti-PGK1 (1:500; rabbit polyclonal; Abcam, Cambridge, MA, USA) were diluted in the buffer and incubated at 4 °C overnight. After subsequently washing with TBST, the membranes were incubated with secondary antibody (horseradish peroxidase-conjugated anti-rabbit) for 1 h at room temperature. The experiment was repeated in triplicate. The bands were detected by enhanced chemiluminescence detection reagents (Applygen Technologies Inc., Beijing, China).
Tissue microarray
Tissue microarray (TMA) allowed the testing of a single biomarker in a high-throughput manner to test a large number of normal and cancerous tissues simultaneously. Tissue microarray (TMA) block was obtained by punching a 1.5 mm tissue cylinder (core) through a histologically representative area of each ‘donor’ tumour block, which was then inserted into an empty ‘recipient’ TMA paraffin block using a manual tissue arrayer as described (Parker et al, 2002). After the construction of the array block, all of the tissue blocks were sectioned with a microtome to 4 μm and affixed to the slide.
Immunohistochemical staining
The tissue sections were dried at 70 °C for 3 h. After deparaffinisation and hydration, the sections were washed in phosphate-buffered saline (PBS; 3 min × 3). The washed sections were treated with 3% H2O2 in the darkness for 5–20 min. After washing in distilled water, the sections were washed in PBS (5 min × 3). Antigen retrieval was performed in citrate buffer (pH 6.0) at 100 °C for 10 min. Each section was then treated with PGK1 rabbit polyclonal antibody (Abcam; at a dilution of 1:100) solution at 4 °C overnight. After washing in PBS (5 min × 3), each section was incubated with secondary antibody at room temperature for 30 min. After washing in PBS (5 min × 3), each section was treated with diaminobenzidine working solution at room temperature for 3–10 min and the slides were counterstained with haematoxylin. For negative controls, the primary antibody was substituted with PBS. The positive controls were lung cancer tissues with positive expression of PGK1 (Chen et al, 2003).
Evaluation of PGK1 protein expression by IHC
Semiquantitative expression levels were based on the intensity and the distribution of staining in a series of randomly selected 10 high-power fields, which were considered as representative of the average in a × 400 magnification field. Staining intensity was classified into four groups: level 0 (no staining), level 1 (weak staining=light yellow), level 2 (moderate staining=yellow brown) and level 3 (strong staining=brown). The percentage (0–100%) of the extent of reactivity was scored as follows: 0 (no positive tumour cells), 1 (fewer than 10% positive tumour cells), 2 (10–50% positive tumour cells) and 3 (more than 50% positive tumour cells). Next, the cytoplasmic expression score was obtained by multiplying the intensity and reactivity extension values. The score of 4 was used to distinguish between low and high expression of PGK1 (Remmele and Stegner, 1987).
Statistical analyses
All analyses were performed using statistical software (SPSS 17.0 for Windows; SPSS, Inc., Chicago, IL, USA). Associations between PGK1 expression and the patients’ clinicopathologic features, including age, tumour size, LNM, TNM stage, histologic grade, molecular subtypes and status of ER, PR, Her-2, Ki67 and P53 were assessed by the chi-square test. The Kaplan–Meier method was used to estimate OS. The influence of different variables on survival was assessed using Cox univariate and multivariate regression analyses. Risk ratios and their 95% confidence intervals were recorded for each marker. For continuous variables, Student’s t-test was performed. The level of significance was set at P<0.05.
Results
PGK1 mRNA and protein expression in breast cancer tissues
The analysis of real-time qPCR was used to confirm mRNA levels. The mean expression value of PGK1 mRNA in cancer tissues (3.542±3.737, normalised by GAPDH gene expression) was significantly higher than the value (1.995±1.711) for the corresponding normal tissues (P=0.010). We then defined >1-fold changes as upregulation of PGK1 mRNA between the cancer tissues and matched normal tissues. The results revealed that 61% (28/46) of breast cancer tissues expressed a higher level of PGK1 compared with the matched normal tissues (Figure 1A).
PGK1 elevated expression in fresh breast cancer tissues. (A) Histogram of PGK1 mRNA expression in breast cancer.The PGK1 mRNA expression was calculated by the 2−ΔΔCt method, the relative expression in each patient was presented as the ratio of T (tumour tissue)/N (normal tissue). (B) Representative western blotting analysis of PGK1 expression in breast tissues. The levels of GAPDH were used as an internal control.
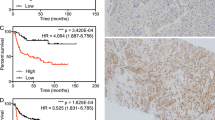
The difference in PGK1 expression between the cancer and normal tissues reflected at the protein level was investigated using western blotting and IHC. Overall, breast cancer tissues exhibited dramatically higher levels of PGK1 protein expression compared with normal tissues. Western blotting assays demonstrated that PGK1 exhibited high expression in cancer tissues compared with the normal tissues (Figure 1B). Immunohistochemistry (IHC) analysis showed high expression of PGK1 in 136 of the cancer specimens (33.9%). However, of the 123 normal specimens, we found high PGK1 expression only in 10 (8.1%) samples (P<0.001). Figures 2A and B show representative images of PGK1 staining in cancer tissue from a cancer patient and matched normal breast tissue, respectively.
Relationship between immunoreactivity of PGK1 and clinicopathologic features
As shown in Table 1, PGK1 expression was significantly associated with histologic grade (P=0.009) and the status of ER (P=0.004), Her-2 (P=0.026) and P53 (P=0.012). A total of 102 of 266 patients (38.3%) who were grade III had significantly higher incidences of high PGK1 expression than those patients of grade I and II (25.2%, 34 of 135, P=0.009). We detected high PGK1 expression in 66 of 234 (28.2%) ER-negative patients and 70 of the 167(41.9%) ER-positive patients (P=0.004). PGK1 was present in 29.1% (74 of 254) and 42.0% (37 of 88) of patients in the Her-2-negative group and the Her-2-positive group, respectively (P=0.026). High PGK1 expression was more frequently observed in P53-positive patients (P=0.012). However, no significant association was observed between PGK1 overexpression and age (P=0.430), tumour size (P=0.733), LNM (P=0.200), TNM stage (P=0.309), molecular subtypes (P=0.275) and the status of PR (P=0.883) and Ki67 (P=0.192).
Predictive significance of PGK1 expression in invasive breast cancer patients
Univariate and multivariate analyses were conducted to determine the predictors for OS (Table 2). In the univariate analysis, tumour size (P=0.023), LNM (P<0.001), TNM stage (P<0.001), PGK1 expression (P=0.022) and the status of PR (P=0.005), Her-2 (P=0.015) and Ki67 (P<0.001) were significantly associated with OS. Lymph node metastasis (P<0.001, risk ratio=4.242, 95% confidence interval: 2.077–8.664) and positive Ki67 (P<0.001, risk ratio=3.748, 95% confidence interval: 1.977–7.105) expression were independent factors confirmed by multivariate analysis.
Figure 3A shows that the OS was significantly higher in patients expressing elevated levels of PGK1 (P=0.02). The data indicated that high PGK1 expression was associated with worse OS; however, high PGK1 expression was not an independent prognostic factor.
Prognostic significance of PGK1 expression in breast cancer patients treated with paclitaxel
Univariate and multivariate survival analyses were also used to evaluate the effects of PGK1 expression and the clinicopathologic characteristics on prognosis in patients treated with paclitaxel. Univariate analysis showed that the following factors were significantly related to OS: Ki67 (P=0.030) and PGK1 expression (P=0.001). Multivariate analysis indicated that PGK1 (P=0.001, risk ratio=3.127, 95% confidence interval: 1.616–6.049) was an independent predictor of OS (Table 3).
Furthermore, the Kaplan–Meier 5-year survival curves stratified for PGK1 expression in patients treated with paclitaxel are shown in Figure 3B. Among the selected patients, PGK1 expression showed significant effects on OS (P<0.001).
Discussion
Glycolysis plays a major role in the energy metabolism of cancer cells, which is characterised by increased glucose uptake and elevated energy generation (Young and Anderson, 2008). Otto Warburg first noted in the 1920s that cancer cells undergo glycolysis even in the presence of ample oxygen (Warburg, 1956). This preferential use of aerobic glycolysis, termed the Warburg effect, has emerged as a metabolic hallmark of many cancers. As a result, there is great interest in understanding the markers in the glycolysis pathway.
PGK1 is an ATP-generating enzyme in the glycolytic pathway catalysing the conversion of 1,3-diphosphoglycerate to 3-phosphoglycerate (Wang et al, 2007). In addition to the catalytic role in the glycolytic pathway, PGK1 is indeed a multifunctional molecule. PGK1 has been shown to affect DNA replication and repair in mammalian cell nuclei (Vishwanatha et al, 1992; Popanda et al, 1998) and several studies have linked its expression to tumour biology (Lay et al, 2000; Hwang et al, 2006; Zieker et al, 2010b). Elevated levels of PGK1 protein have been detected in the serum of patients with pancreatic cancer, suggesting its potential as a biomarker for cancer (Hwang et al, 2006). The increased expression of PGK1 at the protein and mRNA levels has been associated with a poor outcome in lung adenocarcinoma patients (Chen et al, 2003). However, several studies on the function of angiogenesis and metastasis have seemed to contradict the hypothesis that overexpression of PGK1 is a step in tumour development. PGK1 may play a role in inhibiting tumour angiogenesis by promoting an extracellular formation of angiostatin from plasmin (Vaupel and Mayer, 2007). PGK1 has been suggested to be a critical downstream target of CXCL12 and an important negative regulator of an angiogenic switch that is essential for tumour and metastatic growth (Wang et al, 2007). Moreover, overexpression of PGK1 has been reported to reduce the expression of uPAR in human lung cancer cells H157 and to inhibit migration in H157 cells (Shetty et al, 2005).
PGK1 has been shown to be activated in breast cancer (Zhang et al, 2005; Kabbage et al, 2008; Cortesi et al, 2009). Overexpression of PGK1 was observed in proteins secreted in tumour interstitial fluid of breast cancer (Cortesi et al, 2009). Elevated levels of PGK1 protein have been detected in breast cancer tissues compared with normal tissues (Kabbage et al, 2008). PGK1 is overexpressed in Her-2-positive tissues and cell lines (Zhang et al, 2005). In this study, high expression of PGK1 in breast cancer was confirmed by both real-time qPCR (P=0.010) and western blotting using matched cancer-normal tissues. We also found that PGK1 was highly expressed in breast cancer tissues compared with matched normal tissues as assessed by IHC using breast cancer TMA (P<0.001). Our results agreed with the data analysis from the UCSC Cancer Browser (https://genome-cancer.ucsc.edu, TCGA), which showed higher PGK1 expression levels in breast cancer tissues than normal breast tissues (P<0.001).
To our knowledge, most of the previous studies in breast cancer either reported the findings on PGK1 expression only or just performed by cancer cells of Her-2-positive and Her-2-negative breast cancers. The significance of PGK1 expression in clinical breast cancer remains uncertain. Therefore, we analysed the correlation of PGK1 expression with the clinicopathologic features and survival in breast cancer.
Overall, positive correlations of PGK1 expression with histologic grade (P=0.009) and the status of ER (P=0.004), Her-2 (P=0.026) and P53 (P=0.012) have been demonstrated in the current study. Schulz et al (2009) compared the protein expression pattern of Her-2-positive breast cancer vs triple-negative breast cancer, demonstrating that PGK1 showed increased expression levels in the Her-2-positive breast cancer. It was also observed that PGK1 expression was reduced by partially switching off Her-2 signalling with Herceptin treatment (Zhang et al, 2005). These findings correspond with our results indicating that PGK1 was more highly expressed in the Her-2-positive tumours. The high expression of PGK1 in breast cancer also serves as a prognostic factor for poor OS (P=0.02), although it was not an independent factor. In lung cancer, PGK1 was associated with survival in the protein and mRNA study (Chen et al, 2003). Elevated levels of PGK1 were also significantly correlated with poor outcome in 107 patients with lung adenocarcinomas using ELISA analysis and 117 adenocarcinomas and squamous lung cancers using TMA. Collectively, these data suggest that PGK1 has a significant role in the oncogenesis and progression of human breast cancer.
Chemoresistance is of paramount importance in cancer, because in the absence of an effective chemotherapy, other treatments are most often doomed to failure. Chemoresistance-associated gene discovery has shifted from the traditional empiric random screening approach to a more rational and target-based approach whereby the prognosis of patients may be predicted. Although mechanisms of chemoresistance are multiple, it has been indicated that hypoxic cells are the most chemoresistant cells (Vaupel and Mayer, 2007; Bertout et al, 2008). Additionally, the expression levels of PGK1 are controlled by oxygen tension and increased PGK1 expression may reflect more hypoxic tumours (Daly et al, 2004). Thus, as PGK1 appears to play a key role in oncogenesis and survival of breast cancer, it may also be a prognostic target for chemotherapy (Cortesi et al, 2009). PGK1 has been found to be overexpressed in cisplatin-resistant ovarian cancer (Gong et al, 2011; Lincet et al, 2012) and in adriamycin-resistant leukemic K562 cells (Peng et al, 2011). PGK1 may also induce a multidrug-resistant phenotype through a MDR-1-independent mechanism (Duan et al, 2002). However, its role is ambiguous in breast cancer.
As paclitaxel is one of the most used chemotherapy drugs in breast cancer, patients whose treatment included paclitaxel were divided into two groups according to PGK1 expression for OS analysis in this study. During the follow-up period, the survival time of patients with high expression of PGK1 was shorter than that of patients with low expression of PGK1 (P<0.001). Moreover, PGK1 was an independent predictor of survival in patients treated with paclitaxel (P=0.001). Taken together, we hypothesised that PGK1 expression might be the reason for poor prognosis in patients treated with paclitaxel. However, the exact mechanism remains unknown and requires further investigation.
Conclusion
In conclusion, the identification of molecules associated with the response to chemotherapy might be important for predicting the efficacy of specific anti-neoplastic drugs and for the development of less empiric strategies regarding therapeutic choices. Our data demonstrated a significant correlation between PGK1 expression and poor prognosis in breast cancer. On the basis of the correlation between PGK1 expression and survival in breast cancer patients treated with paclitaxel, we suggested that PGK1 overexpression might be a prognostic biomarker of chemoresistance to paclitaxel treatment in breast cancer. Therefore, more studies are required to understand the precise role of PGK1 and to determine whether PGK1 may be used as a prognostic target.
Change history
14 April 2015
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Ai J, Huang H, Lv X, Tang Z, Chen M, Chen T, Duan W, Sun H, Li Q, Tan R, Liu Y, Duan J, Yang Y, Wei Y, Li Y, Zhou Q (2011) FLNA and PGK1 are two potential markers for progression in hepatocellular carcinoma. Cell Physiol Biochem 27 (3-4): 207–216.
Berry DA, Cronin KA, Plevritis SK, Fryback DG, Clarke L, Zelen M, Mandelblatt JS, Yakovlev AY, Habbema JD, Feuer EJ (2005) Effect of screening and adjuvant therapy on mortality from breast cancer. N Engl J Med 353 (17): 1784–1792.
Bertout JA, Patel SA, Simon MC (2008) The impact of O2 availability on human cancer. Nat Rev Cancer 8 (12): 967–975.
Cheang MC, Chia SK, Voduc D, Gao D, Leung S, Snider J, Watson M, Davies S, Bernard PS, Parker JS, Perou CM, Ellis MJ, Nielsen TO (2009) Ki67 index, HER2 status, and prognosis of patients with luminal B breast cancer. J Natl Cancer Inst 101 (10): 736–750.
Chen G, Gharib TG, Wang H, Huang CC, Kuick R, Thomas DG, Shedden KA, Misek DE, Taylor JM, Giordano TJ, Kardia SL, Iannettoni MD, Yee J, Hogg PJ, Orringer MB, Hanash SM, Beer DG (2003) Protein profiles associated with survival in lung adenocarcinoma. Proc Natl Acad Sci USA 100 (23): 13537–13542.
Cortesi L, Barchetti A, De Matteis E, Rossi E, Della Casa L, Marcheselli L, Tazzioli G, Lazzaretti MG, Ficarra G, Federico M, Iannone A (2009) Identification of protein clusters predictive of response to chemotherapy in breast cancer patients. J Proteome Res 8 (11): 4916–4933.
Daly EB, Wind T, Jiang XM, Sun L, Hogg PJ (2004) Secretion of phosphoglycerate kinase from tumour cells is controlled by oxygen-sensing hydroxylases. Biochim Biophys Acta 1691 (1): 17–22.
Dayan F, Roux D, Brahimi-Horn MC, Pouyssegur J, Mazure NM (2006) The oxygen sensor factor-inhibiting hypoxia-inducible factor-1 controls expression of distinct genes through the bifunctional transcriptional character of hypoxia-inducible factor-1alpha. Cancer Res 66 (7): 3688–3698.
Duan Z, Lamendola DE, Yusuf RZ, Penson RT, Preffer FI, Seiden MV (2002) Overexpression of human phosphoglycerate kinase 1 (PGK1) induces a multidrug resistance phenotype. Anticancer Res 22 (4): 1933–1941.
Edwards BK, Ward E, Kohler BA, Eheman C, Zauber AG, Anderson RN, Jemal A, Schymura MJ, Lansdorp-Vogelaar I, Seeff LC, van Ballegooijen M, Goede SL, Ries LA (2010) Annual report to the nation on the status of cancer, 1975-2006, featuring colorectal cancer trends and impact of interventions (risk factors, screening, and treatment) to reduce future rates. Cancer 116 (3): 544–573.
Etzioni R, Tsodikov A, Mariotto A, Szabo A, Falcon S, Wegelin J, DiTommaso D, Karnofski K, Gulati R, Penson DF, Feuer E (2008) Quantifying the role of PSA screening in the US prostate cancer mortality decline. Cancer Causes Control 19 (2): 175–181.
Goldhirsch A, Winer EP, Coates AS, Gelber RD, Piccart-Gebhart M, Thurlimann B, Senn HJ (2013) Personalizing the treatment of women with early breast cancer: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann Oncol 24 (9): 2206–2223.
Gong F, Peng X, Zeng Z, Yu M, Zhao Y, Tong A (2011) Proteomic analysis of cisplatin resistance in human ovarian cancer using 2-DE method. Mol Cell Biochem 348 (1-2): 141–147.
Hammond ME, Hayes DF, Dowsett M, Allred DC, Hagerty KL, Badve S, Fitzgibbons PL, Francis G, Goldstein NS, Hayes M, Hicks DG, Lester S, Love R, Mangu PB, McShane L, Miller K, Osborne CK, Paik S, Perlmutter J, Rhodes A, Sasano H, Schwartz JN, Sweep FC, Taube S, Torlakovic EE, Valenstein P, Viale G, Visscher D, Wheeler T, Williams RB, Wittliff JL, Wolff AC (2010) American Society of Clinical Oncology/College Of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Clin Oncol 28 (16): 2784–2795.
Hsu CY, Ho DM, Yang CF, Lai CR, Yu IT, Chiang H (2002) Interobserver reproducibility of Her-2/neu protein overexpression in invasive breast carcinoma using the DAKO HercepTest. Am J Clin Pathol 118 (5): 693–698.
Hwang TL, Liang Y, Chien KY, Yu JS (2006) Overexpression and elevated serum levels of phosphoglycerate kinase 1 in pancreatic ductal adenocarcinoma. Proteomics 6 (7): 2259–2272.
Kabbage M, Chahed K, Hamrita B, Guillier CL, Trimeche M, Remadi S, Hoebeke J, Chouchane L (2008) Protein alterations in infiltrating ductal carcinomas of the breast as detected by nonequilibrium pH gradient electrophoresis and mass spectrometry. J Biomed Biotechnol 2008: 564127.
Kessler J, Hahnel A, Wichmann H, Rot S, Kappler M, Bache M, Vordermark D (2010) HIF-1alpha inhibition by siRNA or chetomin in human malignant glioma cells: effects on hypoxic radioresistance and monitoring via CA9 expression. BMC Cancer 10: 605.
Lay AJ, Jiang XM, Kisker O, Flynn E, Underwood A, Condron R, Hogg PJ (2000) Phosphoglycerate kinase acts in tumour angiogenesis as a disulphide reductase. Nature 408 (6814): 869–873.
Lincet H, Guevel B, Pineau C, Allouche S, Lemoisson E, Poulain L, Gauduchon P (2012) Comparative 2D-DIGE proteomic analysis of ovarian carcinoma cells: toward a reorientation of biosynthesis pathways associated with acquired platinum resistance. J Proteomics 75 (4): 1157–1169.
McShane LM, Altman DG, Sauerbrei W, Taube SE, Gion M, Clark GM (2005) REporting recommendations for tumour MARKer prognostic studies (REMARK). Br J Cancer 93 (4): 387–391.
Millar EK, Graham PH, McNeil CM, Browne L, O'Toole SA, Boulghourjian A, Kearsley JH, Papadatos G, Delaney G, Fox C, Nasser E, Capp A, Sutherland RL (2011) Prediction of outcome of early ER+ breast cancer is improved using a biomarker panel, which includes Ki-67 and p53. Br J Cancer 105 (2): 272–280.
Monti E, Gariboldi MB (2011) HIF-1 as a target for cancer chemotherapy, chemosensitization and chemoprevention. Curr Mol Pharmacol 4 (1): 62–77.
Parker RL, Huntsman DG, Lesack DW, Cupples JB, Grant DR, Akbari M, Gilks CB (2002) Assessment of interlaboratory variation in the immunohistochemical determination of estrogen receptor status using a breast cancer tissue microarray. Am J Clin Pathol 117 (5): 723–728.
Peng X, Gong F, Xie G, Zhao Y, Tang M, Yu L, Tong A (2011) A proteomic investigation into adriamycin chemo-resistance of human leukemia K562 cells. Mol Cell Biochem 351 (1-2): 233–241.
Pommerenke E, Mattern J, Volm M (1994) Modulation of doxorubicin-toxicity by tamoxifen in multidrug-resistant tumor cells in vitro and in vivo. J Cancer Res Clin Oncol 120 (7): 422–426.
Popanda O, Fox G, Thielmann HW (1998) Modulation of DNA polymerases alpha, delta and epsilon by lactate dehydrogenase and 3-phosphoglycerate kinase. Biochim Biophys Acta 1397 (1): 102–117.
Remmele W, Stegner HE (1987) [Recommendation for uniform definition of an immunoreactive score (IRS) for immunohistochemical estrogen receptor detection (ER-ICA) in breast cancer tissue]. Pathologe 8 (3): 138–140.
Schulz DM, Bollner C, Thomas G, Atkinson M, Esposito I, Hofler H, Aubele M (2009) Identification of differentially expressed proteins in triple-negative breast carcinomas using DIGE and mass spectrometry. J Proteome Res 8 (7): 3430–3438.
Shetty S, Ganachari M, Liu MC, Azghani A, Muniyappa H, Idell S (2005) Regulation of urokinase receptor expression by phosphoglycerate kinase is independent of its catalytic activity. Am J Physiol Lung Cell Mol Physiol 289 (4): L591–L598.
Siegel R, Ma J, Zou Z, Jemal A (2014) Cancer statistics, 2014. CA Cancer J Clin 64 (1): 9–29.
Vaupel P, Mayer A (2007) Hypoxia in cancer: significance and impact on clinical outcome. Cancer Metastasis Rev 26 (2): 225–239.
Vishwanatha JK, Jindal HK, Davis RG (1992) The role of primer recognition proteins in DNA replication: association with nuclear matrix in HeLa cells. J Cell Sci 101 (Pt 1): 25–34.
Wang J, Wang J, Dai J, Jung Y, Wei CL, Wang Y, Havens AM, Hogg PJ, Keller ET, Pienta KJ, Nor JE, Wang CY, Taichman RS (2007) A glycolytic mechanism regulating an angiogenic switch in prostate cancer. Cancer Res 67 (1): 149–159.
Warburg O (1956) On the origin of cancer cells. Science 123 (3191): 309–314.
Young CD, Anderson SM (2008) Sugar and fat—that's where it's at: metabolic changes in tumors. Breast Cancer Res 10 (1): 202.
Zhang D, Tai LK, Wong LL, Chiu LL, Sethi SK, Koay ES (2005) Proteomic study reveals that proteins involved in metabolic and detoxification pathways are highly expressed in HER-2/neu-positive breast cancer. Mol Cell Proteomics 4 (11): 1686–1696.
Zieker D, Konigsrainer I, Traub F, Nieselt K, Knapp B, Schillinger C, Stirnkorb C, Fend F, Northoff H, Kupka S, Brücher BL, Königsrainer A (2008) PGK1 a potential marker for peritoneal dissemination in gastric cancer. Cell Physiol Biochem 21 (5-6): 429–436.
Zieker D, Konigsrainer I, Tritschler I, Loffler M, Beckert S, Traub F, Nieselt K, Bühler S, Weller M, Gaedcke J, Taichman RS, Northoff H, Brücher BL, Königsrainer A (2010a) Phosphoglycerate kinase 1 a promoting enzyme for peritoneal dissemination in gastric cancer. Int J Cancer 126 (6): 1513–1520.
Zieker D, Konigsrainer I, Weinreich J, Beckert S, Glatzle J, Nieselt K, Bühler S, Löffler M, Gaedcke J, Northoff H, Mannheim JG, Wiehr S, Pichler BJ, von Weyhern C, Brücher BL, Königsrainer A (2010)b Phosphoglycerate kinase 1 promoting tumor progression and metastasis in gastric cancer - detected in a tumor mouse model using positron emission tomography/magnetic resonance imaging. Cell Physiol Biochem 26 (2): 147–154.
Acknowledgements
This study was supported by grants from the National Natural Science Foundation of China(81172498/H1622) and the Project of Heilongjiang province applied technology research and development(GA13C201).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License.
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Sun, S., Liang, X., Zhang, X. et al. Phosphoglycerate kinase-1 is a predictor of poor survival and a novel prognostic biomarker of chemoresistance to paclitaxel treatment in breast cancer. Br J Cancer 112, 1332–1339 (2015). https://doi.org/10.1038/bjc.2015.114
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2015.114
Keywords
This article is cited by
-
Identification of a novel glycolysis-related signature to predict the prognosis of patients with breast cancer
World Journal of Surgical Oncology (2021)
-
Detecting prognostic biomarkers of breast cancer by regularized Cox proportional hazards models
Journal of Translational Medicine (2021)
-
Metabolic protein phosphoglycerate kinase 1 confers lung cancer migration by directly binding HIV Tat specific factor 1
Cell Death Discovery (2021)
-
ETV4 promotes breast cancer cell stemness by activating glycolysis and CXCR4-mediated sonic Hedgehog signaling
Cell Death Discovery (2021)
-
Metabolic classification of circulating tumor cells as a biomarker for metastasis and prognosis in breast cancer
Journal of Translational Medicine (2020)