Abstract
Background:
The aim of this work was to examine the risk of lymphohaematopoietic (LH) cancer according to benzene exposure among offshore workers.
Methods:
Cancer registry data were used to identify 112 cancer cases diagnosed during 1999–2011 in a cohort of 24 917 Norwegian men reporting offshore work between 1965 and 1999. Analyses were conducted according to a stratified case–cohort design with a reference subcohort of 1661 workers. Cox regression was used to estimate hazard ratios with 95% confidence intervals, adjusted for other benzene exposure and smoking.
Results:
Most workers were exposed to benzene for <15 years. The upper range values of average intensity and cumulative exposure were estimated to 0.040 p.p.m. and 0.948 p.p.m.-years, respectively. Risks were consistently elevated among exposed workers for all LH cancers combined and for most subgroups, although case numbers were small and yielded imprecise risk estimates. There was evidence of dose-related risk patterns according to cumulative exposure for acute myeloid leukaemia (AML), multiple myeloma (MM) (P trends 0.052 and 0.024, respectively), and suggestively so for chronic lymphocytic leukaemia (CLL) according to average intensity (P trend 0.094).
Conclusions:
Our results support an association between cumulative and intensity metrics of low-level benzene exposure and risk for AML, MM, and suggestively for CLL.
Similar content being viewed by others
Main
Offshore production of crude oil and natural gas has been carried out in the North Sea from movable or stationary installations since the late 1960s. As opposed to the ‘downstream’ petroleum industry, which refers to refinement and distribution of petroleum products, the offshore industry is an ‘upstream’ activity and refers to exploration, drilling, and extraction of oil and gas from underwater reservoirs.
Aromatic hydrocarbons such as benzene, toluene, ethylbenzene, and xylene (collectively labelled: BTEX) are natural components of the petroleum stream and exposure to these agents in the offshore work environment may occur during production control and maintenance of the process systems that separate crude oil, natural gas, condensate, and water (Bråtveit et al, 2007; Steinsvåg et al, 2007). The International Agency for Research on Cancer (IARC) has classified benzene as carcinogenic to humans, giving rise to acute myeloid leukaemia (AML), and possibly increasing the risk of acute lymphoblastic leukaemia (ALL), chronic lymphocytic leukaemia (CLL), multiple myeloma (MM), and non-Hodgkin lymphoma (NHL). Other BTEX-hydrocarbons are listed as possible carcinogens or considered as not classifiable (IARC, 2014).
In 1998, the Cancer Registry of Norway established a cohort of workers employed offshore between 1965 and 1999 to examine associations between cancer and potential hazards in the working environment, whereof benzene’s relation to lymphohaematopoietic (LH) cancer was of particular interest (Stenehjem et al, 2014). Less than 400 personal measurements of benzene during 12-h offshore shifts, all sampled after 1994, existed to assist exposure assessment for work before 1999, and 95% of them showed concentrations <0.16 parts per million (p.p.m.), which complies well with the occupational exposure limit for benzene of 0.6 p.p.m. (Steinsvåg et al, 2007). Because of the paucity of measurement data, experts on industrial hygiene developed semi-quantitative exposure estimates based on information on duration and frequency of job tasks that involved benzene exposure (Bråtveit et al, 2011, 2012). Benzene exposure in everyday life has been considered less important (Duarte-Davidson et al, 2001; IARC, 2012) but may have a role when effects from low occupational exposures are addressed.
Historically, NHL has been defined as all lymphomas except ALL, CLL, MM, and Hodgkins lymphoma (HL). However, the most recent WHO classification of haematopoietic and lymphoid tissues considers ALL, CLL, and MM of B-cell lineage to be major subtypes of B-cell NHL (B-NHL) together with diffuse large B-cell lymphoma (DLBCL) and follicular lymphoma (FL) (Swerdlow et al, 2008). Early studies that examined NHL as one entity (Schnatter et al, 1996; Hayes et al, 1997; Nilsson et al, 1998; Collins et al, 2003), as well as more recent studies where NHL was examined by subtypes (Wang et al, 2009; Cocco et al, 2010), have reported inconsistent findings of benzene-related risks.
For subgroups of myeloid neoplasms, such as AML, chronic myeloid leukaemia (CML), and myelodysplastic syndrome (MDS), several studies have provided evidence for benzene-related risks in both downstream and upstream segments of the petroleum industry (Rushton, 1993; Schnatter et al, 1996, 2012; Järvholm et al, 1997; Rushton and Romaniuk, 1997; Nilsson et al, 1998; Lewis et al, 2003; Glass et al, 2003, 2014; Sorahan et al, 2005; Kirkeleit et al, 2008; Rushton et al, 2014), but few studies have examined such a link for lymphoid neoplasms, among upstream petroleum workers (Gun et al, 2004, 2006; Kirkeleit et al, 2008), and only one study used quantitative estimates of benzene exposure (Glass et al, 2003). Some reports have suggested that benzene may be leukaemogenic and haematotoxic at low concentrations (<10 p.p.m. and <0.2 p.p.m., respectively) (Glass et al, 2003; Hayes et al, 1997; Lan et al, 2004, 2006; Schnatter et al, 2012; Talbott et al, 2011; Vlaanderen et al, 2010). Although the link with LH cancers has been studied thoroughly in recent years, there is limited evidence to decide which characteristics of benzene exposure are the most relevant for identification and assessment of LH cancer risk: for example, cumulative exposure, exposure duration, intensity, or peak exposures (Collins et al, 2003).
The aim of the present study was to examine risk of LH cancers in a cohort of 24 917 male offshore workers exposed to low average concentrations of benzene.
Materials and Methods
Study population and study design
No complete and reliable employment registers of Norwegian offshore workers was available in the 1990s, and in 1998, the Cancer Registry of Norway therefore established a cohort by conducting a survey among active and former offshore workers. Postal questionnaires were distributed to a roster of 57 328 men and women compiled from personnel lists of onshore and offshore workers from oil companies, member lists from labour unions, and lists of participants at offshore safety courses. Data on work history and lifestyle factors were included in the questionnaires (appendices) for prospective analysis of cancer and cause-specific mortality. A total of 27 987 individuals confirmed having worked offshore on the Norwegian continental shelf for at least 20 days between 1965 and 1999 (inclusion criterion), and in a recent study, the survey response rate was estimated to be 69% among true offshore workers verified by comparison with the Norwegian State Register of Employers and Employees (Stenehjem et al, 2014).
For workers with more than two offshore employments, and for onshore employments, information had to be extracted manually from the questionnaires. For economic reasons, data extraction was performed on a random subsample of the cohort only, as well as for all LH cancers, according to a stratified case–cohort design (Borgan et al, 2000). We chose to stratify on birth cohorts with the aim of having a minimum of five controls per cancer case in each birth cohort. Within each stratum, the subcohort is a representative sample of the full cohort. This approach is a type of case–control design that allows fitting prospective models such as Cox-regression models to the data. A key advantage of the design is that the controls (sampled non-cases) may be used for different types of cancer cases, as they are not matched to specific cases.
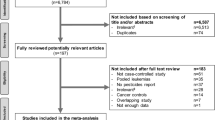
Female offshore workers were not considered for the present study due to a low number of subjects (n=2572), and because most women were employed as administrative and catering personnel assumed to have low exposure to benzene. A total of 498 subjects were excluded from the full cohort owing to irregularities in the reporting (Figure 1), and the remaining 24 917 male offshore workers were eligible for sampling of the subcohort. We drew at random 1675 workers, stratified on 5-year birth cohorts and frequency matched to the birth year distribution of all potentially occupational cancers observed through 2009 (n=556). For the present study, 112 LH cancers were identified in the full cohort.
Based on work histories, the offshore workers were grouped into four main platform activities (production, drilling, maintenance, and catering) and 29 job categories. A total of 913 employments among 615 workers (42 cases and 573 subcohort non-cases) were grouped according to main activity only (no specific job category available). Missing start dates for 289 employments among 157 workers (16 cases and 141 subcohort non-cases) were assigned the average start date within each corresponding birth cohort stratum (1918–1940, 1941–1949, or 1950–1978) and job category. Missing stop dates for 328 employments among 196 workers (18 cases and 178 subcohort non-cases) were assigned the average stop date within each corresponding stratum of start date (1965–1979, 1980–1989, or 1990–1999) and job category. A total of 457 workers (33 cases and 424 subcohort non-cases) were still employed offshore when the survey was conducted in 1998, and their jobs were considered terminated by 31 December 1998.
Participation in the survey was voluntary and based on informed consent. Necessary legal and ethical approvals were obtained from the Norwegian Data Inspectorate, the Regional Committee for Medical Research Ethics, and the Norwegian Directorate of Health.
Identification of cancer cases
The full cohort (n=24 917) was linked to the national database of incident cancer diagnoses at the Cancer Registry of Norway and to the Norwegian National Population Register for data on vital status, year of death, and year of emigration, if relevant. Cases were required to be the first LH cancer diagnosed in each individual between 30 June 1999 and 31 December 2011. The observed 112 LH cancer cases are displayed by subtype and International Classification of Diseases Tenth revision (ICD-10) code in Table 1. Reporting of incident cancers to the Cancer Registry is compulsory in Norway, and data from a number of sources ensure a high degree of completeness (Larsen et al, 2009). Information on cancer localisation was based on a modified version of the ICD-7 and ICD-10, all converted into ICD-10 codes. LH cancer subtypes were classified according to the most recent WHO classification of haematopoietic and lymphoid tissues (Swerdlow et al, 2008) by using histology codes from the ICD-Oncology (ICD-O) second revision for the time period 1999–2001 and from the third revision of ICD-O for the time period 2002–2011. In the analyses, we gradually split the case group into finer subgroups of LH cancers, in order to display the value of detailed diagnostic data. CLL was combined with one case of small lymphocytic lymphoma (SLL) in the main analysis, denoted as ‘CLL’ only. In a supplemental analysis, 11 B-cell CLL cases were analysed together with 1 CLL case of unknown lineage and 2 cases of hairy cell leukaemia and without the 1 SLL case according to the Kiel classification system for non-solid tumors, which were used with local extensions by the Cancer Registry before 2002 (Larsen et al, 2009).
Exposure levels
In 2007, Steinsvåg et al (2005, 2007, 2008) published an expert-developed job time–exposure matrix (JEM) for selected carcinogens in the Norwegian offshore industry during 1970–2005, specially prepared for the present cohort. In 2011, the expert group refined the benzene estimates by using more monitoring data and information on job-specific determinants of benzene exposure (Bråtveit et al, 2011, 2012). In brief, semi-quantitative exposure estimates were derived according to a four-step procedure based on the principles described by Hopf et al, 2010: step 1, identifying relevant tasks associated with benzene exposure. Tasks were selected based on results from pooled benzene exposure measurements, most of which were conducted after year 2000; step 2, for each task, 10 determinants of benzene exposure (related to exposure source, transmission path, and individual factors) were scored 0, 1, or 2. Each task was assigned the mean score of the 10 determinants as the average intensity; step 3, estimating frequency and duration of the tasks. Task frequency was assessed as times per work week (t/w) scored 0=0 t/w, 0.5=>0–1 t/w, 1=>1–7 t/w, and 2=>7 t/w. Task duration was scored 1=<15 min., 2=15–60 min., 3=>60 min. An exposure burden score for each task was created by multiplying scores from intensity, duration, and frequency. Estimates of exposure burden scores for each job category were derived by summing the exposure burden scores for each task divided by nine (the total number of benzene-related tasks); step 4, the job-specific exposure burden scores (hereafter JEM-score) had an observed range of 0–2.4. Job categories were categorised into four groups based on the JEM-score (group I: 0, group II: >0–⩽0.5, group III: <0.5–⩽1.0, group IV: >1.0). In addition, scores were developed to reflect the frequency of exceeding the short-term exposure limit (STEL-score). The Norwegian STEL is 3 p.p.m. measured over a 15-min sampling period (NLIA, 2011). STEL-scores for each job category were calculated by assessing whether the STEL was exceeded ‘often’=2, ‘sometimes’=1, or ‘never’=0, when performing the respective tasks. Then, this number (0, 1 or 2) for each task was multiplied with the same frequency-score used for the JEM-score on how often this task was performed (0, 0.5, 1, and 2). Finally, each task’s product (exceedings times frequency) was summed for the tasks relevant for each job category, creating a STEL-score (i.e., related to benzene exposures >3 p.p.m.). The STEL-score had a range of 0–9.0, and job categories were categorised into four groups based on this score (group I: 0, group II: >0–⩽2.0, group III: <2.0–⩽5.0, group IV: >5.0). The benzene JEM and STEL-matrices are available at http://www.uib.no/filearchive/supplementary-information-to-the-jem-.pdf (Bråtveit et al, 2011, 2012). JEM and STEL-scores were assessed for 29 job categories for four 10-year time periods (1970–79, 1980–89, 1990–99, 2000–09). Work histories from 1965 to 1969 were assigned scores from the period 1970 to 1979. Employments described according to main platform activity only were assigned the average JEM and STEL-scores of the job categories within the relevant main platform activity.
In order to facilitate comparison with other studies, the JEM-scores were translated into corresponding p.p.m. values estimated on the basis of all available personal benzene measurements on Norwegian installations from 1994 to 2003. The average level of 0.036 p.p.m. from the 204 samples for process technicians (Steinsvåg et al, 2007) was judged to correspond to their assigned JEM-score of 2.1 for the time period 1990–99, giving a ratio of 0.0171 (0.036/2.1). By multiplying this ratio with average JEM-scores from 0.05 to 2.35, we estimated a proportional corresponding range of average p.p.m.-values from 0.0009 to 0.040. In order to show the distribution of individual average exposure intensities and the corresponding durations of exposure for all exposed workers, we plotted these values in Figure 2, also showing the tertiles according to cumulative exposure. The y axes shows the JEM intensity values (left side) and the translated p.p.m. values (right side).
Average benzene exposure intensity (scores derived from an expert-based job–exposure matrix (JEM) on the left y axis and estimated corresponding p.p.m. values on the right y axis) against duration of exposure among exposed cases and subcohort members in a cohort of offshore oil industry workers. Workers were divided into tertiles according to cumulative exposure (intensity times duration).
Data analysis
Five exposure metrics were derived to reflect different characteristics of benzene exposure: (1) cumulative exposure was derived by multiplying the JEM-score and duration (years) for each employment period, summarising these products for each individual from the start of employment until 31 December 1998 (range: 0–55.3 JEM-score-years, 0–0.948 p.p.m.-years); (2) cumulative peak exposure was derived following the same procedure as cumulative exposure, except using the STEL-score (range: 0–208.3 STEL-score-years); (3) exposure duration (range: 0–33.5 years); (4) average exposure intensity (cumulative exposure divided by exposure duration; range: 0–2.35 JEM-score, 0–0.040 p.p.m.); and (5) an average frequency score of peak exposures (cumulative peak exposure divided by exposure duration; range: 0–157 STEL-score). Cut-points for exposure duration were chosen as ‘>0–5.49 years’, ‘5.5–12.9 years’, and ‘13–33.5 years’ to achieve almost equally sized groups among the exposed workers. Exposure strata for the benzene metrics 1, 2, 4, and 5 above were defined according to tertiles among exposed workers (labelled T1, T2, T3, the latter representing the highest exposed). Based on the full case–cohort set (112 cases and 1661 non-cases), tertiles were labelled with the estimated range of p.p.m.-values within each tertile for cumulative exposure (metric 1): ‘T1 (<0.001–0.037 p.p.m.-years)’, ‘T2 (>0.037–0.123 p.p.m.-years)’, ‘T3 (0.124–0.948 p.p.m.-years)’, and for average exposure intensity (metric 4): ‘T1 (<0.001–0.007 p.p.m)’, ‘T2 (>0.007–0.013 p.p.m)’, and ‘T3 (>0.013–0.040 p.p.m)’. For all analyses, unexposed workers were used as reference (benzene exposure or duration=0). ‘Ever’ and ‘never’ (reference) exposed to benzene were defined as exposure duration >0 years and =0 years, respectively, and the corresponding risks were analysed by period of first employment (1965–1979 and 1980–1998).
Cox regression, adapted to a stratified case–cohort design (Borgan et al, 2000), was used to estimate hazard ratios (HRs) of cancer and corresponding 95% confidence intervals (CIs) associated with the benzene metrics. Cases were assigned a weight of 1 and subcohort non-cases were given weights according to the inverse sampling fraction from their corresponding 5-year birth cohort stratum. Robust variances were used to compute s.es. of the HRs. Age was used as timescale with entry at age by start of follow-up. Subjects were censored at the date of cancer diagnosis, date of emigration, date of death, or end of study, whichever came first. A total of 14 cases were identified as members of the randomly drawn subcohort and were analysed as cases only (weight=1) (Borgan et al, 2000). Thus a sample of 112 LH cases and 1661 subcohort non-cases was used in the analyses (Figure 1).
The robustness of the results was examined by conducting two sensitivity analyses. First, we analysed the data after excluding workers who were categorised according to main platform activity only and excluding workers with replaced start or stop dates (56 cases and 713 non-cases). Second, we conducted an analysis where the 457 workers still employed in 1998 were assumed to be exposed at the same intensity level until censoring or end of observation time (methodological details in Supplementary Material).
In all analyses, HRs were adjusted for potential confounding effects from age (as the time scale), benzene exposure from other work (yes, no), and ever being a daily smoker (yes, no, unknown). Assessment of ‘benzene exposure from other work’ was based on self-reported job titles and industry sector (question 35 in questionnaire, appendices). Workers reporting job titles that included one or more of the keywords: ‘tank’, ‘mechanic’, ‘lab’, ‘painter’, ‘machinist’, ‘refinery’, and ‘chemi’ were assumed to be exposed to benzene. Similarly, benzene exposure was assumed to have taken place for workers reporting experience from the following industry sectors: ‘shipping (bridge, deck, machinist)’, ‘chemical industry’, ‘painting and surface treatment’, ‘farming and forestry’, and ‘other industry’. Tests for linear trend across categories were performed by using the median within each exposure category as a quantitative score. The proportional hazards assumption was evaluated by Schoenfeld residuals. Tests for significance were two-sided, and P-values of ≤0.05 were considered to represent statistical significance. Data analyses were performed using Stata version 13.1 (StataCorp, College Station, TX, USA).
Results
The distribution of exposed workers according to average exposure intensity and exposure duration is displayed in Figure 2. Most workers were exposed to benzene for <15 years, and the median and interquartile range of estimated average intensity, for a 12-h work shift, were 0.008 p.p.m. and 0.006–0.014 p.p.m., respectively. Estimated cumulative exposure values varied from <0.001 to 0.948 p.p.m.-years.
Background characteristics of the case group and non-case subcohort are shown in Table 2. The distributions of first year of employment offshore, benzene exposure from other work, and history of ever being a daily smoker were fairly similar between cases and non-cases.
Table 3 shows the risk estimates (HRs) for LH cancers according to ever exposure to benzene. Risk estimates were consistently elevated among the exposed workers for most LH cancer groups, except for DLBCL, but none were statistically significant. Risks among those with first employment offshore before 1980 were generally higher compared with those with later employment start, although the number of cases was low and the risk estimates correspondingly unstable.
Tables 4 and 5 show the effects of different benzene exposure metrics (cumulative, cumulative peak, duration, average intensity, and average peak) for all LH cancers combined, for all myeloid cancers combined, and for AML (Table 4), and for B-NHL and its major subtypes, including DLBCL, FL, CLL, and MM (Table 5).
For cumulative exposure, risk estimates were elevated for most cancer outcomes. For all myeloid cancers combined, for AML, and for MM, risk estimates in the third tertile of exposure was substantially higher than those in the first and second tertiles. A statistically significant test for trend was observed for MM (P trend 0.024), and one of borderline statistical significance for AML (P trend 0.052).
The risk estimates according to cumulative peak exposure followed a similar pattern as cumulative exposure, although there were suggestions of a weaker response. The risk of AML increased with increasing cumulative peak exposure, but the trend test was not statistically significant (P trend 0.166).
For most outcomes, exposure duration showed a less convincing dose–response pattern.
For both average intensity and average peak, risk estimates were somewhat fluctuating across tertiles of exposure and few clear patterns were observed. However, the risk of AML increased with increasing average intensity (P trend 0.092) and STEL exceedings (P trend 0.056). A similar pattern was found for MM, where the risk increased with increasing average peak (P trend 0.130).
For all exposure metrics, the risk of CLL was consistently elevated among exposed workers, although risk estimates rarely reached statistical significance. In an additional analysis, CLL was analysed with other cancers according to the Kiel classification (Supplementary Table S1, Supplementary Material), and the results were similar to the main analyses. A dose-related pattern was suggested for average intensity in both the main and the supplementary analysis of CLL with P trends of 0.094 and 0.067, respectively.
No association was found for DLBCL in any of the benzene exposure metrics.
The analyses shown in Tables 3 and 4 for all LH cancers combined were repeated with a case–cohort set restricted to subjects with reported job category and without missing start and stop dates. The results were largely similar to those presented in Tables 2, 3, 4 (Supplementary Tables S2–S4, Supplementary Material). The analyses for all LH cancers combined in Table 4 were also repeated where the 457 workers still employed in 1998 were assumed to be exposed until censoring. The results were quite similar to those presented in Table 4 (Supplementary Table S5, Supplementary Material).
Discussion
This study was designed to examine risk of LH cancers according to different metrics of benzene exposure in a cohort of male Norwegian offshore workers with their first offshore employment between 1965 and 1999. Our primary expectations were to identify elevated risks of the specific diseases listed by IARC as known or suspected to be caused by benzene exposure: AML, ALL, CLL, and MM (IARC, 2012). Although the exposure levels were low (<0.040 avg. p.p.m. and <1 p.p.m.-yrs.), the overall picture was one of an increased risk of LH cancer among the exposed, who constituted two-thirds of the cohort. There were evidence of dose-related patterns for cumulative exposure, exposure intensity, and peak exposures for AML, for MM, and suggestively for CLL.
Our findings are generally in line with other studies conducted in petroleum workers or other benzene-exposed workers. Kirkeleit et al (2008) reported a three-fold increased risk of AML in Norwegian upstream operators employed before 1985 compared with an economically active and geographically matched reference group. An Australian study reported a seven-fold increased risk of acute non-lymphocytic leukaemia among petroleum workers exposed to >8 p.p.m.-years (Glass et al, 2003). Further, a study in UK petroleum marketing and distribution workers reported increased risks of AML or monocytic leukaemia in relation to cumulative, duration, and intensity metrics of benzene (Rushton and Romaniuk, 1997). A Canadian study did not report any association between risk of leukaemia (all subtypes combined) and cumulative benzene exposure (Schnatter et al, 1996), but the chosen reference category included 10 out of the 14 cases with exposure up to 0.45 p.p.m-years, which may have hampered the identification of risks at any level. A recent pooled analysis of the Canadian, the Australian, and the UK data, comprising a total of 60 AML cases, showed an elevated risks of AML according to cumulative, intensity, duration, and peak metrics of benzene exposure (Schnatter et al, 2012; Rushton et al, 2014), although there was little evidence of a dose-related pattern. In contrast to the pooled analysis, and in line with our results, a study of leukaemia risk in relation to gasoline spill in Pennsylvania, USA suggested a dose–response relationship between atmospheric benzene levels <1 p.p.m. and AML (Talbott et al, 2011). Moreover, recent studies have detected genotoxic effects and altered gene expression linked to leukaemia among workers exposed to low levels of benzene (i.e., <1 p.p.m.), which supports a biological plausability for a dose–response relation between average benzene levels <1 p.p.m and AML risk (Angelini et al, 2011; McHale et al, 2011; Li et al, 2014).
The elevated risks observed for CLL in our study are consistent with the findings in the Australian and UK studies (Rushton and Romaniuk, 1997; Glass et al, 2003). A recent review and meta-analysis addressed the association between lymphoma subtypes and benzene exposure by stratifying occupational studies according to three quality dimensions: classification of lymphomas, known to be better after 1970; quality of exposure assessment; and strength of the association between AML risk and benzene exposure (Vlaanderen et al, 2011). The latter criterion served as a quality measure for the exposure estimates, and a similar evaluation would add support to the present study. The meta-analysis provided evidence that CLL is associated with occupational benzene exposure, in line with our results. Another large case–control study also suggested a link between benzene exposure and CLL (Cocco et al, 2010).
Our data suggested increased risks of MM for all exposure metrics, with a statistically significant trend test for cumulative exposure. These results accord with those published by Kirkeleit et al (2008) of increased risk of MM in upstream operators employed offshore before 1985 and with similar findings in two meta-analyses (Infante, 2006; Vlaanderen et al, 2011). The Australian study, however, reported no association between benzene exposure and risk of MM, although a weakness was that MM was combined with NHL (Glass et al, 2003).
In their most recent evaluation of benzene as a carcinogen, the IARC pointed out that NHL is a heterogeneous group of histological subtypes and that few cohort studies have reported benzene-related risks of NHL by histological subtype according to the present day understanding (IARC, 2012). When we examined B-NHL as one entity, cumulative exposure and exposure duration suggested dose-related patterns. Even the lack of an association between exposure metrics for benzene and DLBCL and the opposite for CLL correspond with the findings of Cocco et al (2010), while another study suggested a closer relationship between benzene and DLBCL compared with CLL (Wang et al, 2009).
Interestingly, the observed risk patterns for MM and CLL correspond to the finding of benzene toxicity to B-cells in the bone marrow at occupational benzene exposure <0.2 p.p.m. (Lan et al, 2004, 2006). Also, immunosuppression of T-cells mediated by benzene reduces the immunosurveillance and thus the ability to prevent B-cell neoplasia (IARC, 2012).
It is an ongoing discussion what metric is most closely associated with benzene-related risks (Collins et al, 2003). Our data suggested a stronger link between LH cancer and cumulative, intensity, and peak metrics, compared with exposure duration. This may, however, depend on study-specific contrasts within the exposure metrics. Also, time period of exposure seemed to have a role on LH cancer risk. The observed elevated risks among those with first exposure before 1980, compared with those with a later employment start, are probably explained by reduced exposure levels as a result of technological improvements during the 1980s and 1990s (e.g., closing open fluid flow lines, more efficient ventilation systems, etc.) (Steinsvåg et al, 2006, Stenehjem et al, 2015).
Generally, adjustment for benzene exposure from other work and from smoking had negligible effects on the HRs, and no change of direction occurred except for estimates close to the null, in agreement with a low degree of confounding. Still, our adjustment was rather crude and residual confounding, of both directions, cannot be ruled out.
Important strengths of our study were the prospective case–cohort design, the independent outcome data from a high-quality national cancer registry, virtually complete linkage based on unique personal identity numbers, and the access to independent exposure estimates developed by industrial hygiene experts specifically for cancer studies in our cohort.
On the other hand, some of the workers reported occupations from up to three decades before the start of follow-up. The long time span may introduce recall differences, although self-reported occupational history has been found to be quite robust (Wärneryd et al, 1991). Further, we were unable to allow for individual differences in exposure within each occupational group. Average exposure intensity of benzene was estimated for typical workers within the respective job categories in three time periods but did not allow for variability in exposure across installations and oil fields. Some degree of exposure misclassification obviously exists in our data. The potential recall bias in the occupational histories would possibly be related to age, rather than to disease status. A bias towards the null therefore seems to be the most likely effect. We also had no information on work experience and exposures after 1999 for the study subjects, but the sensitivity analysis indicated no substantial bias from this shortcoming. Moreover, the observed risk pattern for AML indicated that the benzene exposure estimates for the present study were satisfactory, at least on a relative scale. We therefore consider benzene as a strong candidate to explain the observed risk pattern, and we find it unlikely that bias, confounding, or chance should create an overall picture like the one seen in our study.
Measurements of benzene exposure had mainly been conducted among process operators after year 2000 (Bråtveit et al, 2011, 2012), and a semi-quantitative expert assessment was chosen in order to take into account exposure differences on a work-task level back in time. The average exposure concentration in p.p.m. for process technicians was estimated to correspond to the process technicians' semi-quantitative JEM-score for the period 1990–1999, based on available measurements, knowledge of the actual conditions, and performed tasks during sampling. The transformation of JEM-scores to p.p.m. levels carries uncertainty as no measurement data from before 1990 existed, neither to indicate job-specific p.p.m. levels nor to describe the relationship (proportionality) between the semi-quantitative scores and actual p.p.m. levels.
In conclusion, our analyses indicated that there is an increased risk of LH cancer among benzene-exposed offshore workers, compared with unexposed, on the Norwegian continental shelf. The 12-h shift average intensity levels were estimated to be <0.040 p.p.m., and the cumulative levels to be <1 p.p.m.-years. There was evidence of a dose-related risk pattern according to exposure intensity and cumulative exposure for AML, for MM, and suggestively for CLL.
Change history
28 April 2015
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Angelini S, Kumar R, Bermejo JL, Maffei F, Barbieri A, Graziosi F, Carbone F, Cantelli-Forti G, Violante FS, Hemminki K, Hrelia P (2011) Exposure to low environmental levels of benzene: evaluation of micronucleus frequencies and S-phenylmercapturic acid excretion in relation to polymorphisms in genes encoding metabolic enzymes. Mut Res 719 (1-2): 7–13.
Borgan Ø, Langholz B, Samuelsen SO, Goldstein L, Pogoda J (2000) Exposure stratified case-cohort designs. Life Data Anal 6 (1): 39–58.
Bråtveit M, Kirkeleit J, Hollund BE, Moen BE (2007) Biological monitoring of benzene exposure for process operators during ordinary activity in the upstream petroleum industry. Ann occup Hyg 51 (5): 487–494.
Bråtveit M, Kirkeleit J, Hollund BE, Vågnes KS, Abrahamsen EH (2011) Retrospective JEM for benzene in the Norwegian oil and gas industry. Conference abstract Occup Environ Med 68 (Suppl 1): A26.
Bråtveit M, Hollund BE, Kirkeleit J, Abrahamsen EH (2012) Supplementary information to the Job Exposure Matrix for benzene, asbestos and oil mist/oil vapour among Norwegian offshore workers. Report. University of Bergen: Bergen, Norway, Available from: http://www.uib.no/filearchive/supplementary-information-to-the-jem-.pdf (accessed 20 January 2013).
Cocco P, t’Mannetje A, Fadda D, Melis M, Becker N, de Sanjosé S, Foretova L, Mareckova J, Staines A, Kleefeld S, Maynadié M, Nieters A, Brennan P, Boffetta P (2010) Occupational exposure to solvents and risk of lymphoma subtypes: results from the Epilymph case–control study. Occup Environ Med 67 (5): 341–347.
Collins JJ, Ireland B, Buckley CF, Shepperly D (2003) Lymphohaematopoeitic cancer mortality among workers with benzene exposure. Occup Environ Med 60 (9): 676–679.
Duarte-Davidson R, Courage C, Rushton L, Levy L (2001) Benzene in the environment: an assessment of the potential risks to the health of the population. Occup Environ Med 58 (1): 2–13.
Glass DC, Gray CN, Jolley DJ, Gibbons C, Sim MR, Fritschi L, Adams GG, Bisby JA, Manuell R (2003) Leukemia risk associated with low-level benzene exposure. Epidemiology 14 (5): 569–577.
Glass DC, Schnatter AR, Tang G, Irons RD, Rushton L (2014) Risk of myeloproliferative disease and chronic myeloid leukaemia following exposure to low-level benzene in a nested case-control study of petroleum workers. Occup Environ Med 71 (4): 266–274.
Gun RT, Pratt NL, Griffith EC, Adams GG, Bisby JA, Robinson KL (2004) Update of a prospective study of mortality and cancer incidence in the Australian petroleum industry. Occup Environ Med 61 (2): 150–156.
Gun RT, Pratt N, Ryan P, Roder D (2006) Update of mortality and cancer incidence in the Australian petroleum industry cohort. Occup Environ Med 63 (7): 476–481.
Hayes RB, Yin S-N, Dosemeci M, Li G-L, Wacholder S, Travis LB, Li CY, Rothman N, Hoover RN, Linet MS (1997) Benzene and the dose-related incidence of hematologic neoplasms in China. J Natl Cancer Inst 89 (14): 1065–1071.
Hopf NB, Waters MA, Ruder AM, Prince MM (2010) Development of a retrospective job exposure matrix for PCB-exposed workers in capacitor manufacturing. J Occup Health 52 (4): 199–208.
Infante PF (2006) Benzene exposure and multiple myeloma: a detailed meta-analysis of benzene and cohort studies. Ann NY Acad Sci 1076: 90–109.
IARC (2012) International Agency for Research on Cancer: monograph on the evaluation of the carcinogenic risk of chemicals Vol. 100F. WHO: Lyon, France.
IARC (2014) International Agency for Research on Cancer: agents classified by the IARC monographs Vol. 1–109., IARC monographs on the evaluation of the carcinogenic risks to humans. IARC: Lyon, France, updated 14 January 2014. Available from: http://monographs.iarc.fr/ENG/Classification/ClassificationsAlphaOrder.pdf (accessed 1 March 2014).
Järvholm B, Mellblom B, Norrman R, Nilsson R, Nordlinder R (1997) Cancer incidence of workers in the Swedish petroleum industry. Occup Environ Med 54 (9): 686–691.
Kirkeleit J, Riise T, Bråtveit M, Moen BE (2008) Increased risk of acute myelogenous leukemia and multiple myeloma in a historical cohort of upstream petroleum workers exposed to crude oil. Cancer Causes Control 19 (1): 13–23.
Lan Q, Zhang L, Li G, Vermeulen R, Weinberg RS, Dosemeci M, Rappaport SM, Shen M, Alter BP, Wu Y, Kopp W, Waidyanatha S, Rabkin C, Guo W, Chanock S, Hayes RB, Linet M, Kim S, Yin S, Rothman N, Smith MT (2004) Hematotoxicity in workers exposed to low levels of benzene. Science 306 (5702): 1774–1776.
Lan Q, Vermeulen R, Zhang L, Li G, Rosenberg PS, Alter BP, Sehn M, Rappaport SM, Weinberg RS, Chanock S, Waidyanatha S, Rabkin C, Hayes RB, Linet M, Kim S, Yin S, Rothman N, Smith MT (2006) Response to the letter “benzene exposure and hematotoxicity” by Lamm SH and Grünwald HW. Science 312 (5776): 998–999.
Larsen IK, Småstuen M, Johannesen TB, Langmark F, Parkin DM, Bray F, Møller B (2009) Data quality at the Cancer Registry of Norway: an overview of comparability, completeness, validity and timeliness. Eur J Cancer 45 (7): 1218–1231.
Lewis RJ, Schnatter AR, Drummond I, Murray N, Thompson FS, Katz AM, Jorgensen G, Nicolich MJ, Dahlman D, Thériault G (2003) Mortality and cancer morbidity in a cohort of Canadian petroleum workers. Occup Environ Med 60 (12): 918–928.
Li K, Jing Y, Yang C, Liu S, Zhao Y, He X, Li F, Han J, Li G (2014) Increased leukemia-associated gene expression in benzene-exposed workers. Sci Rep 4 (5369): 1–5.
McHale CM, Zhang L, Lan Q, Vermeulen R, Li G, Hubbard AE, Porter KE, Thomas R, Portier CJ, Shen M, Rappaport SM, Yin S, Smith MT, Rothman N (2011) Global gene expression profiling of a population exposed to a range of benzene levels. Environ Health Perspect 119 (5): 628–634.
Nilsson RI, Nordlinder R, Hörte L-G, Järvholm B (1998) Leukaemia, lymphoma, and multiple myeloma in seamen on tankers. Occup Environ Med 55 (1): 517–521.
Norwegian Labour Inspection Authorities (NLIA) (2011) Veiledning om administrative normer for forurensning i arbeidsatmosfære [Guidelines on occupational exposure limits for polution in the working atmosphere]. Report no. 326 in Norwegian. NLIA: Trondheim, Norway, updated December 2011. Available from: http://www.arbeidstilsynet.no/binfil/download2.php?tid=77907 (accessed 27 January 2015).
Rushton L (1993) A 39-year follow-up of the U.K. oil refinery and distribution center studies: results for kidney cancer and leukaemia. Environ Health Persp 101 (Suppl. 6): 77–84.
Rushton L, Romaniuk H (1997) A case-control study to investigate the risk of leukaemia associated with exposure to benzene in petroleum marketing and distribution workers in the United Kingdom. Occup Environ Med 54 (3): 152–166.
Rushton L, Schnatter AR, Tang G, Glass DC (2014) Acute myeloid and chronic lymphoid leukaemias and exposure to low-level benzene among petroleum workers. Br J Cancer 110 (3): 783–787.
Schnatter AR, Armstrong TW, Nicolich MJ, Thompson FL, Katz AM, Huebner WW, Pearlman ED (1996) Lymphohaematopoietic malignancies and quantitative estimates of exposure to benzene in Canadian petroleum distribution workers. Occup Environ Med 53 (11): 773–781.
Schnatter AR, Glass DC, Tang G, Irons RD, Rushton L (2012) Myelodysplastic syndrome and benzene exposure among petroleum workers: an international pooled analysis. J Natl Cancer Inst 104 (22): 1724–1737.
Sorahan T, Kinlen LJ, Doll R (2005) Cancer risk in a historical UK cohort of benzene exposed workers. Occup Environ Med 62 (4): 231–236.
Steinsvåg K, Bråtveit M, Moen BE (2005) Eksponering for kreftfremkallende faktorer i norsk offshore petroleumsvirksomhet 1970–2005. [Exposure to carsinogenic agents in the Norwegian offshore petroleum industry 1970–2005]. Report in Norwegian. University of Bergen: Bergen, Norway.
Steinsvåg K, Bråtveit M, Moen BE (2006) Exposure to carcinogens for defined job categories in Norway’s offshore petroleum industry, 1970 to 2005. Ann Occup Hyg 50 (2): 109–122.
Steinsvåg K, Bråtveit M, Moen BE (2007) Exposure to carcinogens for defined job categories in Norway’s offshore petroleum industry, 1970 to 2005. Occup Environ Med 64 (4): 250–258.
Steinsvåg K, Bråtveit M, Moen B, Austgulen LV, Hollund BE, Haaland IM, Naerheim J, Svendsen K, Kromhout H (2008) Expert assessment of exposure to carcinogens in Norway’s petroleum oil industry. J Expo Sci Environ Epidemiol 18 (2): 175–182.
Stenehjem JS, Kjærheim K, Rabanal KS, Grimsrud TK (2014) Cancer incidence among 41 000 offshore oil industry workers. Occup Med (Lond) 64 (7): 539–545.
Stenehjem JS, Friesen MC, Eggen T, Kjærheim K, Bråtveit M, Grimsrud TK (2015) Self-reported occupational exposures among 28,000 offshore oil industry workers employed between 1965 and 1999. J Occup Environ Hyg e-pub ahead of print 11 February 2015 doi:10.1080/15459624.2014.989358.
Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, Thiele J, Vardiman JW (2008) WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. International Agency for Research on Cancer: Lyon, France.
Talbott EO, Xu X, Youk AO, Rager JR, Malek AM (2011) Risk of leukemia as a result of community exposure to gasoline vapors: a follow-up study. Environ Res 111 (4): 597–602.
Vlaanderen J, Portengen L, Rothman N, Lan Q, Kromhout H, Vermeulen R (2010) Flexible meta-regression to assess the shape of the benzene-leukemia exposure-response curve. Environ Health Persp 118 (4): 526–532.
Vlaanderen J, Lan Q, Kromhout H, Rothman N, Vermeulen R (2011) Occupational benzene exposure and the risk of lymphoma subtypes: A meta-analysis of cohort studies incorporating three study quality dimensions. Environ Health Persp 119 (2): 159–167.
Wang R, Zhang Y, Lan Q, Holford TR, Leaderer B, Zahm SH, Boyle P, Dosemeci M, Rothman N, Zhu Y, Qin Q, Zheng T (2009) Occupational exposure to solvents and risk of non-Hodgkin lymphoma in Conneticut women. Am J Epidemiol 15 (169): 176–185.
Wärneryd B, Thorslund M, Ostlin P (1991) The quality of retrospective questions about occupational history—a comparison between survey and census data. Scand J Soc Med 19 (1): 7–13.
Acknowledgements
We wish to thank our former Head of Department, Aage Andersen, without whose efforts the establishment of the cohort and the conduct of the subsequent studies would not have been possible, Researcher Leif Åge Strand for conducting the offshore survey, Senior Consultant Tone Eggen for cleaning the work history data, Data Manager Jan Ivar Martinsen for sampling of the subcohort and linking cancer and exposure estimates to the cohort, and MD Svein Hansen for advice on grouping of LH cancer (Department of Research, Cancer Registry of Norway). We also thank Industrial Hygienists Jorunn Kirkeleit and Bjørg Eli Hollund (Department of Occupational Medicine, Haukeland University Hospital, Norway) and Research Director John Cherrie (Institute of Occupational Medicine, Edinburgh, UK) for assisting with the JEM development. This work was funded by the Research Council of Norway’s PETROMAKS program (grant no. 189612).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License.
Supplementary Information accompanies this paper on British Journal of Cancer website
Supplementary information
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 4.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Stenehjem, J., Kjærheim, K., Bråtveit, M. et al. Benzene exposure and risk of lymphohaematopoietic cancers in 25 000 offshore oil industry workers. Br J Cancer 112, 1603–1612 (2015). https://doi.org/10.1038/bjc.2015.108
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2015.108
Keywords
This article is cited by
-
Exposure to benzene and other hydrocarbons and risk of bladder cancer among male offshore petroleum workers
British Journal of Cancer (2023)
-
Crude oil exploration in Africa: socio-economic implications, environmental impacts, and mitigation strategies
Environment Systems and Decisions (2022)
-
Occupational health risk assessment of the benzene exposure industries: a comprehensive scoring method through 4 health risk assessment models
Environmental Science and Pollution Research (2022)
-
The importance of evaluating specific myeloid malignancies in epidemiological studies of environmental carcinogens
BMC Cancer (2021)
-
The disease burden attributable to 18 occupational risks in China: an analysis for the global burden of disease study 2017
Environmental Health (2020)