Abstract
Background:
Cancers not detected by breast screening are commonly assumed to have poorer prognosis.
Methods:
We examined the survival experience of all women aged 50–74 years diagnosed with a first breast cancer between 1998 and 2006 in British Columbia, Canada and determined their screening experience. Disease-specific survival rates were calculated and, for cases diagnosed in 2002, prognostic factors (size, nodal involvement, grade ER status and stage) were examined by time since screening.
Results:
Breast cancers diagnosed at screening had the best survival (P<0.001). Cancers detected within 12 months of a negative screen had similar survival rates (P=0.98) to those diagnosed within 12–23 and 24–47 months, with other non-screen-detected cancers having poorer survival (P<0.001). The prognostic profile of cancers diagnosed in 2002 followed a similar pattern.
Interpretation:
There was no evidence that cancers diagnosed within 12 months had poorer prognosis than those diagnosed up to 48 months following screening.
Similar content being viewed by others
Main
Mammographic screening does not detect all breast cancers and cancers arising between tests (interval cancers) typically represent 25–50% of cancers diagnosed in women undergoing biennial screening. The length-biased selection effect of screening implies that rapidly growing cancers are more likely to manifest as interval cancers than slower growing cases that have more opportunity to be screen detected. The proportion of interval cancers that are fast growing should decline with the elapsed time since last screen, as the selection effect of testing diminishes. Because of the preferential selection for rapid growth, interval cancers are frequently considered to be more aggressive (Welch and Black, 2010; Narod, 2011; Miller, 2013). However, some recent research has found screening programme interval cancers diagnosed within 24 months of a preceding negative screen to have better survival than cancers diagnosed in comparable non-screened women (Bordas et al, 2006; Lawrence et al, 2009). Earlier results from randomized trials have been inconsistent (Holmberg et al, 1986; Frisell et al, 1992).
This study set out to examine the survival and prognostic distribution of cancers arising in British Columbian women eligible to participate in breast screening and determine the relationship effect of the these outcomes to elapsed time since preceding screen.
Materials and Methods
All first cases of invasive breast cancer, excluding death certificate only registrations, diagnosed in British Columbian women aged 50–74 years between 1998 and 2006 were identified from the BC Cancer Registry and their records linked to the publically funded provincial breast screening programme (SMPBC) database (Olivotto et al, 2000). Attention was restricted to first cancers, as women with a history of breast cancer are not eligible for programme breast screening. SMPBC was established in 1988 and has provided biennial screening to women aged 50–74 years since 1997 (Wai et al, 2005). Survival information was available and complete up until 31 December 2010. Screening data were used to classify cases as screen-detected (diagnosed within 12 months of an abnormal screen), by time since previous screen, and never screened. Staging and prognostic factor data were not generally available from the BC Cancer Registry before 2010, but all breast cancer cases diagnosed in 2002 were staged as part of a quality control exercise and this midpoint year was used to represent the total cohort of 1998–2006. Prognostic factors available on the 2002 cases included stage, tumour size, nodal status, tumour grade and oestrogen receptor status. Where pathologic stage was unknown or incomplete staging was based on clinical findings.
Survival rates were calculated using the Kaplan–Meier method (Lawless, 1982) with death from breast cancer counted as events and other deaths or end of follow-up counted as censoring events. Survival differences were compared using Cox proportional hazards (Lawless, 1982), adjusted for age. Tests for differences in categorical variables were based on the χ2-test.
Results
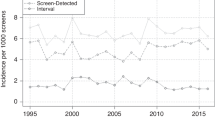
There were 13 453 cases of invasive breast cancer registered in BC in women aged 50–74 years between 1998 and 2006: 12 605 were first cancers and there were 1671 deaths from breast cancer recorded in the study period. The average age at diagnosis of the sample was 61.8 years and the 5-year breast cancer-specific survival rate was 89.9%. Breast cancers were classified into six categories based on the screening status at diagnosis: screen-detected, diagnosed within 0–11 months of a negative screen, 12–23 months, 24–47 months, 48+ months and no screening history. Breast cancer-specific survival rates (Figure 1), adjusted for age, were elevated for screen-detected cancers (P<0.001), were similar between the groups diagnosed within 1–47 months of screening (P=98) and between non-participants and those not screened for 48+ months (P=15). Survival was significantly different between cases diagnosed within 48 months and those after 48 months of a screen or never screened (P<0001).
There were 1431 first invasive breast cancers diagnosed in 2002. Table 1 provides a distribution of the following prognostic factors: age, tumour size, nodal involvement, TNM stage, tumour grade, and ER status for the six screening groups. Tests of significance showed that in the comparison of cancers diagnosed more than 4 years post screening versus never screened, only stage was significant (P=02). In the comparison of cancers occurring within 4 years post screening there were no significant differences on any prognostic factors. And for the comparison of cancers within 4 years post screening to those after 4 years or never screened, significant differences were found for size (P=005) and stage (P=003) and not for grade or ER, whereas nodal involvement was close to significant (P=07).
Discussion
As expected, screen-detected cancers had the best survival, as they have been downstaged and are subject to lead time and length biases (Miller, 2013). Cancers diagnosed in women who had never been screened or who had not been screened in four or more years had the worst survival. These findings have been seen in other analyses (Bordas et al, 2006; Lawrence et al, 2009) and are likely to be because of the effect of participant self-selection. No consistent pattern of difference was found between the screening-defined groups on the prognostic factors available. However, comparisons of prognostic factors had less power than the survival analysis because of the smaller sample size available for analysis.
Cancers diagnosed within 12, 12–23, and 24–47 months of a negative screen had similar survival rates in this study. Neither survival nor the distribution of prognostic factors was worse among women whose cancer was diagnosed within 1 year of a screen, for which the proportion of fast growing cancers would be anticipated to be the greatest. Kalager et al (2012) also found that interval cancer survival did not vary with elapsed time since previous screen and that survival for interval cases and for a comparable unscreened population were similar.
The reason that early interval cancers have non-inferior survival is not clear. Treatment may reduce or eliminate any relationship between growth rate and survival. Classical models of chemotherapy effectiveness predict that responsiveness is generally higher where more tumour cells are actively cycling, which is correlated with faster growth (Chu and DeVita, 2001). Bordas et al (2006) hypothesised that fast growing tumours may give rise to more readily perceived signs and symptoms resulting in earlier clinical detection. Kalager et al (2012) also discuss the potential influence of cancers resulting from screening-interpretation misses that would tend to reduce the effect of length-biased sampling.
Another possible explanation is provided by considering the dynamics of the diagnosis of cancers not detected at screening. The large variation seen in the size and extent of disease in unscreened women indicates that factors, other than tumour size, influence when a cancer will be diagnosed and its prognostic profile. Because screening preferentially harvests larger cancers present at the time of testing the distribution of cancers undetected will be enriched for smaller tumours when compared with an unscreened population. Consequently the population of potential interval cancers is skewed towards less advanced cancers. Those diagnosed soon after a screen will tend to be enriched with cases among the post-screen population that were destined for early clinical detection. This effect will tend to counter the effect of growth rate mediated length-biased sampling, and the net effect on survival and prognosis will depend on the relative strengths of these opposing influences.
We would conclude that breast cancers with poorer prognosis are not more likely to appear as early interval cancers and that other factors counterbalance the potential effect of length-biased sampling by screening on survival. A further consequence of this observation is that those screen-detected cancers, that are not overdiagnosed, have average prognosis.
Change history
04 February 2014
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Bordas P, Jonsson H, Nystrom L, Lenner P (2006) Survival from invasive breast cancer among interval cases in the mammography screening programmes of northern Sweden. Breast 16: 47–54.
Chu E, DeVita V (2001) Principles of cancer management: chemotherapy. In Cancer - Principles and Practice of Oncology DeVita V, Hellman S, Rosenberg S (eds) 6th edn pp 289–292. Lippincott, Williams and Wilkins: Philedelphia.
Frisell J, von Rosen A, Wiege M, Nilsson B, Goldman S (1992) Interval cancer and survival in a randomized breast cancer screening trial in Stockholm. Breast Cancer Res Treat 24: 11–16.
Holmberg L, Adami HO, Tabar L, Bergstrom R (1986) Survival in breast cancer diagnosed between mammographic screening examinations. Lancet 2: 27–30.
Kalager M, Tamimi RM, Bretthauer M, Adami HO (2012) Prognosis in women with interval breast cancer: population based observational cohort study. BMJ 345: e7536.
Lawless J (1982) Statistical Models and Methods for Lifetime Data 1st edn John Wiley: New York.
Lawrence G, O'Sullivan E, Kearins O, Tappenden N, Martin K, Wallis M (2009) Screening histories of invasive breast cancers diagnosed 1989-2006 in the West Midlands, UK: variation with time and impact on 10-year survival. J Med Screen 16 (4): 186–192.
Miller AB (2013) III.10 Screening. In Handbook of Epidemiology Ahrens W, Pigeot I (eds) 2nd edn pp 1267. Springer: Heidleberg.
Narod SA (2011) Age of diagnosis, tumor size, and survival after breast cancer: implications for mammographic screening. Breast Cancer Res Treat 128: 259–266.
Olivotto IA, Kan L, d'Yachkova Y, Burhenne LJ, Hayes M, Hislop TG, Worth AJ, Basco VE, King S (2000) Ten years of breast screening in the Screening Mammography Program of British Columbia, 1988-1997. J Med Screen 7 (3): 152–159.
Wai ES, D'yachkova Y, Olivotto IA, Tyldesley S, Phillips N, Warren LJ, Coldman AJ (2005) Comparison of 1- and 2-year screening intervals for women undergoing screening mammography. Br J Cancer 92 (5): 961–966.
Welch HG, Black WC (2010) Overdiagnosis in Cancer. J Nat Cancer Inst 102 (9): 605–613.
Acknowledgements
The authors wish to acknowledge the assistance of the Screening Mammography Program of BC and the BC Cancer Registry for data provided.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Both authors are employed by the BC Cancer Agency that manages the Screening Mammography Program of BC and the BC Cancer Registry.
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Coldman, A., Phillips, N. Breast cancer survival and prognosis by screening history. Br J Cancer 110, 556–559 (2014). https://doi.org/10.1038/bjc.2013.732
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2013.732
Keywords
This article is cited by
-
Dramatic increase in volume versus length of invasive ductal carcinoma mimicking intramammary lymph node in a small nodular lesion
Bulletin of the National Research Centre (2022)
-
Cost-Effectiveness Models in Breast Cancer Screening in the General Population: A Systematic Review
Applied Health Economics and Health Policy (2017)