Abstract
Background:
S-1 is an oral fluoropyrimidine that mimics infusional 5-fluorouracil. The aim of this phase II trial was to explore the clinical efficacy of the triplet regimen TIROX, which consists of S-1, irinotecan and oxaliplatin.
Methods:
Forty-two chemo-naive patients with metastatic colorectal cancer (mCRC) were planned to be enrolled and be treated with irinotecan 150 mg m−2 followed by oxaliplatin 85 mg m−2 on day 1 and S-1 80 mg m−2 per day from day 1 to 14 every 3 weeks. Polymorphisms in the UGT1A1, UGT1A6, UGT1A7 and CYP2A6 genes were analysed.
Results:
Between July 2007 and February 2008, 43 patients were enrolled. An objective response was noted in 29 patients (67.4%, 95% confidence interval: 53.4–81.4), of which 2 achieved durable complete responses. The median progression-free survival was 10.0 months and the median overall survival was 19.2 months. Significant grade 3 or 4 adverse events were neutropenia (45.2%), febrile neutropenia (9.5%), diarrhoea (7.1%) and vomiting (9.5%). Increased gastrointestinal toxicities were associated with the presence of UGT1A6*2 or UGT1A7*3 and an improved tumour response was noted in those without variant alleles of CYP2A6 or UGT1A1*60.
Conclusion:
The combination of S-1, irinotecan and oxaliplatin showed favourable efficacy and tolerability in untreated patients with mCRC.
Similar content being viewed by others
Main
Colorectal cancer is the third most common cancer and the fourth most common cause of death in Korea (Jung et al, 2012). Although there have been considerable progresses in the field of palliative chemotherapy for metastatic colorectal cancer (mCRC), the focus has mainly been focused on the development of targeted agents and their biomarkers. By contrast, the development of conventional chemotherapeutics has been relatively ignored, even though these drugs are still the mainstay of front line treatment of these patients. A meta-analysis of phase III trials revealed that the survival of patients correlated with the proportion of patients who had received three active drugs (fluoropyrimidine, irinotecan and oxaliplatin; Grothey et al, 2004). This became the theoretical basis for the upfront administration of these three drugs in a triplet regimen. Subsequently, a phase III trial conducted by the Gruppo Oncologico Nord Ovest (GONO) showed that compared with the reference doublet regimen of irinotecan and infusional 5-fluorouracil (5-FU) plus leucovorin (FOLFIRI), the triplet combination chemotherapy of oxaliplatin, irinotecan and infusional 5-FU plus leucovorin (FOLFOXIRI) was superior in terms of response rate, progression-free survival (PFS) and overall survival (OS); however, there was a higher incidence of adverse events, which was a cause of concern (Falcone et al, 2007).
S-1 is an oral fluoropyrimidine that has theoretical advantages compared with 5-FU in that it contains 5-chloro-2,4-dihydroxypyridine, an inhibitor of an enzyme that degrades 5-FU. Several phase III trials on mCRC (Muro et al, 2010; Hong et al, 2012) have shown that S-1 can substitute for infusional 5-FU. Our group has also reported the feasibility of the triplet regimen of S-1, irinotecan and oxaliplatin (TIROX), through a phase I trial for gastric and colorectal cancer patients (Park et al, 2008), and a phase II trial for heavily treated patients with mCRC (Kim et al, 2009).
Several polymorphisms have been reported to affect the metabolism of irinotecan and S-1. Uridine diphosphate glucuronosyltranferase (UGT) is an enzyme that inactivates the toxic metabolite of irinotecan, SN-38 and several genetic variants in UGT1A1, UGT1A6 and UGT1A7 associate with the decreased enzymatic activity of UGT1A isoforms, which causes irinotecan to become more toxic (Innocenti et al, 2004; Han et al, 2006; Cecchin et al, 2009). On the other hand, tegafur, an active element of S-1, is transformed to 5-FU by cytochrome P450 2A6 (CYP2A6). It is known that tegafur-based regimens induce less gastrointestinal toxicity in Asian patients than in Caucasians (Shirao et al, 2004), and that the activity of CYP2A6 is relatively lower in Asians; this has been largely attributed to defective alleles of the CYP2A6 gene that result in attenuated enzymatic activity (Chuah et al, 2011). Exploring such genetic variations concerning drug metabolism might aid the selection of patients who can be safely treated with triplet regimen.
The results of a phase II trial of TIROX for previously untreated patients with mCRC are described here. A collateral pharmacogenetic study to test how genetic variations in UGT1A1, UGT1A6, UGT1A7 and CYP2A6 relate to TIROX efficacy and safety was also performed.
Materials and Methods
Patient selection
Patients who met the following eligibility criteria were enrolled: adenocarcinoma of the colon or rectum; unresectable or metastatic disease, age above 18 years; previously untreated for metastatic disease (adjuvant or neo-adjuvant treatment with fluoropyrimidines for non-metastatic disease was allowed if it was completed at least 6 months before the initiation of study treatment); Eastern Cooperative Oncology Group (ECOG) performance status 0-1; measurable disease per Response Evaluation Criteria in Solid Tumours (RECIST, version 1.0); and adequate haematological, renal and hepatic function. Patients with any prior treatment by oxaliplatin or irinotecan were excluded. Patients were also excluded if they had coexisting malignancy, peripheral neuropathy of which severity exceeded grade 2 by National Cancer Institute Common Terminology Criteria for Adverse Events (version 3.0) or brain metastases. The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. All patients provided their informed consent before registration into the study. The protocol was approved by the Institutional Review Board of National Cancer Center, Korea (protocol number NCCNCS-07-261).
Study treatment
Patients were planned to be treated with irinotecan 150 mg m−2 followed by oxaliplatin 85 mg m−2 on day 1 and S-1 80 mg m−2 per day (40 mg m−2 twice a day) from day 1 to 14 every 3 weeks. This regimen was based on the results from a dose-finding phase I study (Park et al, 2008). Treatment was continued until disease progression, occurrence of unacceptable toxicity or patient refusal.
Evaluation criteria
The primary end point was objective response rate, and the secondary end points were PFS, OS, toxicity and pharmacogenetic analysis. The objective response was evaluated after every two cycles of chemotherapy or sooner if needed for documentation of disease progression. A partial response had to be confirmed at 4 weeks or later. For the efficacy analysis, all enrolled patients (intention-to-treat population) were assessed. Patients who received at least one dose of study treatments were assessed as the safety population. Clinical and laboratory toxicities were monitored according to National Cancer Institute Common Terminology Criteria for Adverse Events (version 3.0).
Pharmacogenetic study
DNA was extracted from the peripheral blood buffy-coat cells by using QIAamp Blood Kit (Qiagen, Hilden, Germany). UGT1A1, UGT1A6, UGT1A7 and CYP2A6 genotypes were determined as described elsewhere (Kong et al, 2009; Park et al, 2011b). Direct sequencing was used to examine the following polymorphic sites and to determine the presence of variant alleles that associate with reduced enzymatic activity; UGT1A1*6 (221G>A), UGT1A1*28 ((TA)6>(TA)7), UGT1A1*60 (-3279T>G), UGT1A6 (19T>G, 315A>G, 541A>G, and 552A>C), UGT1A7 (387T>G, 391C>A, 392G>A, and 622T>C), CYP2A6*4 (whole deletion of CYP2A6), CYP2A6*7(6558T>G) and CYP2A6*9 (−48T>G).
Appropriate primers were designed and PCR was performed by using a GeneAmp PCR system 9700 thermal cycler (Applied Biosystems, Foster City, CA, USA). Sequencing was carried out with an Automated ABI Prism 3100 Genetic Analyser (Applied Biosystems). The presence of the CYP2A6 deletion allele(*4) was determined by restriction fragment length polymorphism as described in a previous study (Kong et al, 2009).
Statistical analyses
The sample size was calculated according to Fleming’s single-stage design. A targeted objective response rate of 55% vs an objective response rate of no interest of 35% with a power of 0.80 at a one-sided significance level of 0.05 was chosen, and accrual of 37 assessable patients was projected. Assuming that the data of 10% of the patients would not be evaluable, it was planned to accrue at least 42 patients for this study. Descriptive statistics were reported as proportions and medians. OS and PFS were assessed by the Kaplan–Meier method and the 95% confidence interval (95% CI) for the median time to event was computed. The association of a particular genotype with toxicity or tumour response was analysed by using χ2 test or Fisher’s exact test. Differences between genotypes in PFS or OS were assessed by using log-rank test. In genotype analysis, a P-value less than 0.05 was considered to be potentially significant. Haplotypes for the UGT1A gene were estimated by using the Bayesian method with PHASE version 2.1 (Stephens et al, 2001). Linkage disequilibrium (LD) plot and pairwise LD values were presented by using Haploview (Barrett et al, 2005). Data analyses were performed by using STATA version 10.1 (StataCorp, College Station, TX, USA).
Results
Patient characteristics
Between July 2007 and February 2008, 43 patients were enrolled (Table 1). Eighteen had single organ metastases, which had been decided to be unresectable by the multidisciplinary team. Of 36 patients with synchronous metastases, 21 underwent palliative resection of primary tumour before the enrollment.
Treatment delivery
In total, 368 cycles of chemotherapy were administered to 42 patients; 1 patient withdrew consent before treatment. The median number of cycles was 7 (range, 0–12). The most common cause for discontinuing treatment was disease progression (17, 40%). Other causes included chemotherapy-free interval (15, 36%), metastasectomy (4, 10%), adverse events (3, 7%) and consent withdrawal (3, 7%). There were treatment delays in 170 cycles (46.2%) for 40 patients (95%), and oxaliplatin, irinotecan or S-1 treatment was modified in 19, 23 and 23 patients, respectively. The median relative dose intensity of S-1, irinotecan and oxaliplatin was 83.1% (range: 42–109%), 80.5% (range: 38–105%) and 91.4% (range: 48–105%), respectively.
Efficacy
An objective response was not evaluable in two patients as one withdrew consent before commencement of treatment and another refused to be treated after the first cycle was given. A complete response was noted in 2 patients and a partial response was observed in 27 patients. The confirmed objective response rate was 67.4% (29 out of 43; 95% CI: 53.4–81.4). All 41 patients whose response could be evaluated showed at least stable disease as their best overall response. Moreover, in 22 (51.2%) patients, the reduction in the sum of target lesions exceeded 50% (Figure 1).
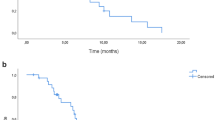
As of April 2012, the median follow-up duration was 46.7 months (range 29.8–56.4). The median PFS was 10.0 months (95% CI: 7.7–12.1) and the median OS was 19.2 month (95% CI: 14.8–28.6; Figure 2). Three patients remained disease free at the time of last follow-up. Two had achieved complete responses to the study treatment and had sustained remissions for 56.4 and 52 months, respectively. The third patient had undergone resection of the single pulmonary nodule that remained after the other lesions disappeared after 12 cycles of study treatment.
Adverse events
The safety population included the 42 patients who were treated with at least one cycle of chemotherapy (Table 2). Overall, 33 patients (78.6%) experienced at least one episode of grade 3 or 4 toxicity. Grade 3 or 4 neutropenia was noted in 19 patients (45.2%). Febrile neutropenia occurred in four patients (9.5%), including two patients with bacteremia. There were no treatment-related deaths because of acute toxicity of chemotherapy.
Metastasectomy and post-study treatment
Five patients eventually underwent hepatic (n=3) or pulmonary (n=2) metastasectomy, although their tumours had been regarded initially to be unresectable. R0 resection was performed in all five patients including three with extensive metastases, namely, at least eight nodules in liver or lung at the time of operation. The consequences of surgery are as follows: one patient with a single liver metastasis, a hepatitis B viral carrier, underwent hepatic metastasectomy but died of postoperative hepatic insufficiency in 1 month after surgery; another patient who had initially more than 10 hepatic nodules showed a complete pathological response in the hepatic resection specimen, but died of postoperative infectious complications; two patients developed disease progression within 6 and 8 months after metastasectomy. Only one patient who underwent pulmonary metastasectomy has been disease free; he is one of the long-term survivors mentioned above.
Second-line or more lines of chemotherapy was given to 35 patients (83.3%) after completion or discontinuation of the study treatment. Of these, 33 patient received chemotherapy in which either irinotecan- or oxaliplatin-containing regimen was reintroduced. Seven patients were treated with a cetuximab-containing regimen and three patients were given a bevacizumab-containing chemotherapy.
Pharmacogenetic analysis
All 42 patients who received the study treatment were genotyped. The allelic frequencies were 0.17 for UGT1A1*6, 0.2 for UGT1A1*28, 0.31 for UGT1A1*60, 0.23-0.35 for single-nucleotide polymorphisms in UGT1A6 and UGT1A7, 0.06 for CYP2A6*4, 0.04 for CYP2A6*7 and 0.15 for CYP2A6*9. When the associations of these genotypes with grade 2 or more toxicity were analysed (Table 3), both UGT1A6*2 or UGT1A7*3 associated significantly with vomiting (both P=0.014) and had marginal relationships with diarrhoea (both P=0.063). UGT1A1*6 also associated marginally with vomiting (P=0.060). There were no significant relationships between haematologic toxicities and genotypes.
When the associations between genotype and efficacy outcomes were analysed, the patients with the UGT1A1*60 or CYP2A6 variant alleles had significantly poorer response rates (Table 4). The adjusted odds ratio of UGT1A1*60 and CYP2A6 variant alleles for tumour response was 0.16 (95% confidence interval 0.03–0.94, P=0.043) and 0.20 (95% confidence interval 0.04–0.99, P=0.049), respectively, when adjusted for ECOG performance status and M1 stage (M1a vs M1b, according to American Joint Committee on Cancer Staging 7th edition; Edge and American Joint Committee on Cancer, 2010).
None of the genotypes associated significantly with PFS. Although UGT1A1*60 or UGT1A1*28 associated with reduced OS in univariate analysis, the relationship became insignificant after adjustment for performance status and M1 stage (data not shown).
As shown in Figure 3, there was high LD with D′>0.9 and r2>0.5 among single-nucleotide polymorphisms in UGT1A6 (19T>G, 315A>G, 541A>G and 552A>C) and UGT1A7 (387T>G, 391C>A, 392G>A and 622T>C). In particular, there was a perfect LD between UGT1A6 541A>G and UGT1A7 622T>C. In other words, patients with UGT1A6*2 were exactly same as those with UGT1A7*3.
Discussion
The present study suggested that first-line treatment with the triplet combination chemotherapy is an excellent option for patients with mCRC who are fit and require maximum tumour shrinkage. The response rate was 67.4% and our primary end point was met by exceeding the initial target response rate of 55%. The degree of tumour shrinkage was satisfactory: more than half of the participants (51.2%) recorded size reductions of more than 50%. The PFS and OS, the secondary end points of this study, were also promising, and the toxicity profiles were acceptable.
The triplet chemotherapy regimen, FOLFOXIRI, is one of the treatment options for mCRC, and is recommended in National Comprehensive Cancer Network guidelines with an evidence level of category 2B. FOLFOXIRI treatment resulted in an objective response rate as high as 66% and a PFS of 9.8 months (Falcone et al, 2007). XELOXIRI, which is composed of capecitabine, oxaliplatin and irinotecan, is another triplet regimen. It was developed by the GONO group and generated an objective response in 67% of the patients and a PFS of 10.1 months (Vasile et al, 2009). In present study, S-1 was incorporated as the fluoropyrimidine component of the triplet regimen, which was called TIROX. Its efficacy was comparable to that of XELOXIRI and FOLFOXIRI: the objective response rate was 67.4% and thus the study met its primary objective, namely, the response rate was higher than the target response rate of 55%. The PFS was 10.0 months, which was also consistent with the PFSs of other triplet regimens or doublets plus biological agents (Hurwitz et al, 2004; Van Cutsem et al, 2009).
TIROX also had a favourable toxicity profile. In this study, irinotecan 150 mg m−2 and oxaliplatin 85 mg m−2 were administered every 3 weeks. Thus, the dose intensities of these drugs were lower than those in FOLFOXIRI or XELOXIRI, where similar doses were given on a biweekly basis (Falcone et al, 2007; Vasile et al, 2009). Because of the reduced dose intensity, non-haematological toxicities, such as neuropathy and diarrhoea, occurred less frequently in this study compared with the studies on FOLFOXIRI and XELOXIRI: grade 3/4 diarrhoea was observed in 7.1% (20.5% in FOLFOXIRI and 30% in XELOXIRI) and there were no cases of grade 3 or more peripheral neuropathy (2% in FOLFOXIRI and 6% in XELOXIRI). However, haematological toxicities occurred with a similar frequency as with other triplet regimens: the incidences of grade 3 or 4 neutropenia (45.2%) and febrile neutropenia (9.5%) were comparable to those observed in studies on FOLFOXIRI and XELOXIRI (Falcone et al, 2007; Vasile et al, 2009).
The OS was 19.2 months, which was less than expected. It should be noted that the proportion of patients with extensive tumour burden was relatively higher in this study: 25% of the patients had liver-only metastases, compared with 32% in the FOLFOXIRI arm of the GONO trial. Moreover, fewer patients underwent metastasectomy (12%) in the present study than in the FOLFOXIRI arm (15%). Notably, four of the five patients who underwent hepatic or pulmonary metastasectomy in the present study failed to achieve long-term survival because of recurrence or adverse events after surgery. This may partly reflect the fact that the patients had extensive tumour burdens and multiple tumour deposits that were initially unresectable at the time of presentation. It is possible that during metastasectomy, not all of the initial lesions were resected despite ‘R0 resection’ being performed, especially if the lesions had disappeared during chemotherapy. Such residual lesions, which contained ‘disappeared’ tumours, are at risk of reappearance as a radiological complete response is not a guarantee of cure (Benoist et al, 2006). Moreover, in this study, two of the three patients who underwent hepatic metastasectomy died of immediate post-operative complications. This may reflect the fact that hepatotoxicity or other peri-operative morbidities are major concerns of neoadjuvant treatment for liver metastases. In contrast, there was no peri-operative mortality in patients who underwent hepatic resection after being treated with the GONO-FOLFOXIRI regimen (Masi et al, 2009). However, the two patients who died after hepatic metastasectomy had other clinical risk factors that precipitated the post-operative complications, namely, combined systemic infection and hepatitis B virus carrier status. Nevertheless, the possibility that triplet chemotherapy may associate with a higher risk of hepatotoxicity and peri-operative complications than doublet chemotherapy cannot be ruled out. It is not clear whether substituting S-1 for 5-FU in pre-operative chemotherapy may increase the peri-operative morbidity or mortality associated with hepatic metastasectomy.
In the present study, there were two long-term survivors who maintained a durable complete response. Both patients had had unresectable multiple metastases and were not eligible for secondary surgery for metastases. Their complete responses have been durable for more than 4 years and they can be stated to be ‘cured’. Such complete responses occur very infrequently. This is true even when modern chemotherapy incorporating biological agents is used: their complete response rates range from 0.5 to 4% (Hurwitz et al, 2004; Van Cutsem et al, 2009). Long-term durable complete responses appear to be even rarer: Ferrarotto et al (2011) showed that only 6 of 2541 patients with mCRC (0.24%) had complete responses after palliative chemotherapy that were sustained for more than 5 years. The two cases exhibiting a durable complete response in the present study suggest that triplet chemotherapy can ‘cure’ the metastatic disease of a certain subset of patients.
How can we identify the individual who is likely to get maximum benefit from TIROX? Biomarker study through randomised trial will be able to answer that question. Recently, BRAF mutation is emerging as a biomarker predicting benefit of triplet chemotherapy. Masi et al (2010) showed the poor prognostic impact of BRAF mutation in mCRC was diluted in patients who were treated with bevacizumab and FOLFOXIRI in a phase II trial. Furthermore, recently TRIBE trial, which compared bevacizumab plus FOLFOXIRI with bevacizumab plus FOLFIRI, showed BRAF mutant patients benefited from FOLFOXIRI rather than FOLFIRI (Falcone et al, 2013).
In this study, to identify pharmacogenetic predictive marker, the patients were genotyped for polymorphisms that are known to affect the metabolism of irinotecan and S-1. UGT1A6*2 and UGT1A7*3 both associated with grade 2/3 gastrointestinal toxicity. Both variant alleles are linked to the decreased glucuronidation activity of UGT1A isoforms (Miners et al, 2002) and increased toxicity with irinotecan-based chemotherapy (Cecchin et al, 2009; Park et al, 2011b). The CYP2A6 wild-type homozygote genotype associated with an increased rate of response to TIROX: the response rate was higher in wild-type patients (*1/*1) than those with one or more variant alleles (*4, *7 and *9). This may reflect the fact that the variant alleles are linked to decreased conversion of tegafur, the active component of S-1, to 5-FU; this might have resulted in reduced exposure to the active metabolite of 5-FU. These results agree with those of previous studies on S-1-containing chemotherapies for gastric cancer, which showed that patients with CYP2A6 wild type have improved efficacy outcomes (Kong et al, 2009; Park et al, 2011a).
In the case of UGT1A1*60, patients without the variant allele had improved response and survival outcome but the allelic distribution did not associate with changes in the toxicity profile. It was known that haplotypes including UGT1A1*60 associated with reduced glucuronidation and increased bilirubin levels in irinotecan-treated Japanese patients (Sai et al, 2004) and with haematological toxicities in Caucasians (Cecchin et al, 2009). However, significant associations between UGT1A1*60 genotypes and irinotecan treatment efficacy have not been observed previously. Theoretically, reduced glucuronidation could increase the exposure to SN-38, the active metabolite of irinotecan, and could therefore associate with improved efficacy outcomes. However, in the present study, the opposite results were observed for UGT1A1*60. Although numerous studies have shown relationships between UGT polymorphisms and toxicity in irinotecan-treated patients, the impact of UGT1A polymorphisms or glucuronidation activity on tumour response or survival is not clear. Unexpected results have also been observed with UGT1A1*6, which associates with poorer clinical outcome despite inducing lower glucuronidation ratios (Han et al, 2006).
To our knowledge, this study is the first data for Asians about efficacy and safety of triplet regimen comprising irinotecan, oxaliplatin and fluoropyrimidine for mCRC. TIROX showed equivalent response, similar haematologic toxicity, and much less gastrointestinal toxicity compared with European regimens, despite of relatively lower dose intensity of irinotecan and oxaliplatin. This trend is consistent with previous studies that showed East Asian individuals were more susceptible to the effects of chemotherapeutic agents than their Western counterparts (O'Donnell and Dolan, 2009). Approved doses of several chemotherapeutic agents have been frequently intolerable in East Asian patients, whereas clinical outcome of reduced doses in East Asian patients was similar with or even better than that of Caucasian patients (Millward et al, 2003; Watanabe et al, 2003). The exception is tegafur-based oral fluoropyrimidine; S-1 or UFT seem to induce more frequent gastrointestinal toxicities in Caucasians, where allele frequencies of CYP2A6 variant genotypes are low (Shirao et al, 2004; Chuah et al, 2011). However, pharmacogenetic analysis of our study seems to be generally insufficient to elucidate the association between clinical outcome and ethnic-specific polymorphisms because of its small sample size and a single-arm trial design.
In conclusion, TIROX was an effective triplet regimen for patients with mCRC as a durable response could be induced in some patients. It was generally tolerable due to relatively lower dose intensities compared with other triplet regimens. Further studies examining the efficacy of this regimen when it is combined with targeted agents may be warranted. Some allelic variants in UGT1A or CYP2A6 associated with the efficacy or toxicity in this triplet regimen. However, the data are not enough yet to allow the identification of good candidates for intensive treatment.
Change history
17 September 2013
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Barrett JC, Fry B, Maller J, Daly MJ (2005) Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21 (2): 263–265.
Benoist S, Brouquet A, Penna C, Julie C, El Hajjam M, Chagnon S, Mitry E, Rougier P, Nordlinger B (2006) Complete response of colorectal liver metastases after chemotherapy: does it mean cure? J Clin Oncol 24 (24): 3939–3945.
Cecchin E, Innocenti F, D'Andrea M, Corona G, De Mattia E, Biason P, Buonadonna A, Toffoli G (2009) Predictive role of the UGT1A1, UGT1A7, and UGT1A9 genetic variants and their haplotypes on the outcome of metastatic colorectal cancer patients treated with fluorouracil, leucovorin, and irinotecan. J Clin Oncol 27 (15): 2457–2465.
Chuah B, Goh BC, Lee SC, Soong R, Lau F, Mulay M, Dinolfo M, Lim SE, Soo R, Furuie T, Saito K, Zergebel C, Rosen LS (2011) Comparison of the pharmacokinetics and pharmacodynamics of S-1 between Caucasian and East Asian patients. Cancer Sci 102 (2): 478–483.
Edge SB . American Joint Committee on Cancer (2010) AJCC Cancer Staging Manual. 7th edn. Springes: New York.
Falcone A, Cremolini C, Masi G, Lonardi S, Zagonel V, Salvatore L, Trenta P, Tomasello G, Ronzoni M, Ciuffreda L, Zaniboni A, Tonini G, Buonadonna A, Valsuani C, Chiara S, Carlomagno C, Boni C, Marcucci L, Boni L, Loupakis F (2013) FOLFOXIRI/bevacizumab (bev) versus FOLFIRI/bev as first-line treatment in unresectable metastatic colorectal cancer (mCRC) patients (pts): Results of the phase III TRIBE trial by GONO group. ASCO Meeting Abstracts 31 (15_suppl): 3505.
Falcone A, Ricci S, Brunetti I, Pfanner E, Allegrini G, Barbara C, Crino L, Benedetti G, Evangelista W, Fanchini L, Cortesi E, Picone V, Vitello S, Chiara S, Granetto C, Porcile G, Fioretto L, Orlandini C, Andreuccetti M, Masi G (2007) Phase III trial of infusional fluorouracil, leucovorin, oxaliplatin, and irinotecan (FOLFOXIRI) compared with infusional fluorouracil, leucovorin, and irinotecan (FOLFIRI) as first-line treatment for metastatic colorectal cancer: the Gruppo Oncologico Nord Ovest. J Clin Oncol 25 (13): 1670–1676.
Ferrarotto R, Pathak P, Maru D, Agarwal A, Overman M, Hoff PM, Kopetz S (2011) Durable complete responses in metastatic colorectal cancer treated with chemotherapy alone. Clin Colorectal Cancer 10 (3): 178–182.
Grothey A, Sargent D, Goldberg RM, Schmoll HJ (2004) Survival of patients with advanced colorectal cancer improves with the availability of fluorouracil-leucovorin, irinotecan, and oxaliplatin in the course of treatment. J Clin Oncol 22 (7): 1209–1214.
Han JY, Lim HS, Shin ES, Yoo YK, Park YH, Lee JE, Jang IJ, Lee DH, Lee JS (2006) Comprehensive analysis of UGT1A polymorphisms predictive for pharmacokinetics and treatment outcome in patients with non-small-cell lung cancer treated with irinotecan and cisplatin. J Clin Oncol 24 (15): 2237–2244.
Hong YS, Park YS, Lim HY, Lee J, Kim TW, Kim KP, Kim SY, Baek JY, Kim JH, Lee KW, Chung IJ, Cho SH, Lee KH, Shin SJ, Kang HJ, Shin DB, Jo SJ, Lee JW (2012) S-1 plus oxaliplatin versus capecitabine plus oxaliplatin for first-line treatment of patients with metastatic colorectal cancer: a randomised, non-inferiority phase 3 trial. Lancet Oncol 13 (11): 1125–1132.
Hurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W, Berlin J, Baron A, Griffing S, Holmgren E, Ferrara N, Fyfe G, Rogers B, Ross R, Kabbinavar F (2004) Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med 350 (23): 2335–2342.
Innocenti F, Undevia SD, Iyer L, Chen PX, Das S, Kocherginsky M, Karrison T, Janisch L, Ramirez J, Rudin CM, Vokes EE, Ratain MJ (2004) Genetic variants in the UDP-glucuronosyltransferase 1A1 gene predict the risk of severe neutropenia of irinotecan. J Clin Oncol 22 (8): 1382–1388.
Jung K, Park S, Kong H, Won Y, Lee J, Seo H, Lee J (2012) Cancer statistics in Korea: incidence, mortality, survival and prevalence in 2009. Cancer Res Treat 44 (1): 11–24.
Kim SY, Hong YS, Kim BC, Park JW, Choi HS, Jeong SY, Kim DY, Hong CW, Sohn DK, Jung KH (2009) A phase II study of S-1 plus irinotecan and oxaliplatin in heavily-treated patients with metastatic colorectal cancer. Invest New Drugs 27 (3): 269–274.
Kong SY, Lim HS, Nam BH, Kook MC, Kim YW, Ryu KW, Lee JH, Choi IJ, Lee JS, Park YI, Kim NK, Park SR (2009) Association of CYP2A6 polymorphisms with S-1 plus docetaxel therapy outcomes in metastatic gastric cancer. Pharmacogenomics 10 (7): 1147–1155.
Masi G, Loupakis F, Pollina L, Vasile E, Cupini S, Ricci S, Brunetti IM, Ferraldeschi R, Naso G, Filipponi F, Pietrabissa A, Goletti O, Baldi G, Fornaro L, Andreuccetti M, Falcone A (2009) Long-term outcome of initially unresectable metastatic colorectal cancer patients treated with 5-fluorouracil/leucovorin, oxaliplatin, and irinotecan (FOLFOXIRI) followed by radical surgery of metastases. Ann Surg 249 (3): 420–425.
Masi G, Loupakis F, Salvatore L, Fornaro L, Cremolini C, Cupini S, Ciarlo A, Del Monte F, Cortesi E, Amoroso D, Granetto C, Fontanini G, Sensi E, Lupi C, Andreuccetti M, Falcone A (2010) Bevacizumab with FOLFOXIRI (irinotecan, oxaliplatin, fluorouracil, and folinate) as first-line treatment for metastatic colorectal cancer: a phase 2 trial. Lancet Oncol 11 (9): 845–852.
Millward MJ, Boyer MJ, Lehnert M, Clarke S, Rischin D, Goh BC, Wong J, McNeil E, Bishop JF (2003) Docetaxel and carboplatin is an active regimen in advanced non-small-cell lung cancer: a phase II study in Caucasian and Asian patients. Ann Oncol 14 (3): 449–454.
Miners JO, McKinnon RA, Mackenzie PI (2002) Genetic polymorphisms of UDP-glucuronosyltransferases and their functional significance. Toxicology 181-182: 453–456.
Muro K, Boku N, Shimada Y, Tsuji A, Sameshima S, Baba H, Satoh T, Denda T, Ina K, Nishina T, Yamaguchi K, Takiuchi H, Esaki T, Tokunaga S, Kuwano H, Komatsu Y, Watanabe M, Hyodo I, Morita S, Sugihara K (2010) Irinotecan plus S-1 (IRIS) versus fluorouracil and folinic acid plus irinotecan (FOLFIRI) as second-line chemotherapy for metastatic colorectal cancer: a randomised phase 2/3 non-inferiority study (FIRIS study). Lancet Oncol 11 (9): 853–860.
O'Donnell PH, Dolan ME (2009) Cancer pharmacoethnicity: ethnic differences in susceptibility to the effects of chemotherapy. Clin Cancer Res 15 (15): 4806–4814.
Park SR, Hong YS, Park Y, Kim NK, Park YL, Park SY, Jung KH (2008) A phase I/II study of S-1 combined with irinotecan and oxaliplatin in patients with metastatic gastric cancer. ASCO Meeting Abstracts 26 (15_suppl): ) 4558.
Park SR, Kong SY, Nam BH, Choi IJ, Kim CG, Lee JY, Cho SJ, Kim YW, Ryu KW, Lee JH, Rhee J, Park YI, Kim NK (2011a) CYP2A6 and ERCC1 polymorphisms correlate with efficacy of S-1 plus cisplatin in metastatic gastric cancer patients. Br J Cancer 104 (7): 1126–1134.
Park SR, Kong SY, Rhee J, Park YI, Ryu KW, Lee JH, Kim YW, Choi IJ, Kim CG, Lee JY, Cho SJ, Kim NK (2011b) Phase II study of a triplet regimen of S-1 combined with irinotecan and oxaliplatin in patients with metastatic gastric cancer: clinical and pharmacogenetic results. Ann Oncol 22 (4): 890–896.
Sai K, Saeki M, Saito Y, Ozawa S, Katori N, Jinno H, Hasegawa R, Kaniwa N, Sawada J, Komamura K, Ueno K, Kamakura S, Kitakaze M, Kitamura Y, Kamatani N, Minami H, Ohtsu A, Shirao K, Yoshida T, Saijo N (2004) UGT1A1 haplotypes associated with reduced glucuronidation and increased serum bilirubin in irinotecan-administered Japanese patients with cancer. Clin Pharmacol Ther 75 (6): 501–515.
Shirao K, Hoff PM, Ohtsu A, Loehrer PJ, Hyodo I, Wadler S, Wadleigh RG, O'Dwyer PJ, Muro K, Yamada Y, Boku N, Nagashima F, Abbruzzese JL (2004) Comparison of the efficacy, toxicity, and pharmacokinetics of a uracil/tegafur (UFT) plus oral leucovorin (LV) regimen between Japanese and American patients with advanced colorectal cancer: joint United States and Japan study of UFT/LV. J Clin Oncol 22 (17): 3466–3474.
Stephens M, Smith NJ, Donnelly P (2001) A new statistical method for haplotype reconstruction from population data. Am J Hum Genetics 68 (4): 978–989.
Van Cutsem E, Kohne CH, Hitre E, Zaluski J, Chang Chien CR, Makhson A, D'Haens G, Pinter T, Lim R, Bodoky G, Roh JK, Folprecht G, Ruff P, Stroh C, Tejpar S, Schlichting M, Nippgen J, Rougier P (2009) Cetuximab and chemotherapy as initial treatment for metastatic colorectal cancer. N Engl J Med 360 (14): 1408–1417.
Vasile E, Masi G, Fornaro L, Cupini S, Loupakis F, Bursi S, Petrini I, Di Donato S, Brunetti IM, Ricci S, Antonuzzo A, Chiara S, Amoroso D, Andreuccetti M, Falcone A (2009) A multicenter phase II study of the combination of oxaliplatin, irinotecan and capecitabine in the first-line treatment of metastatic colorectal cancer. Br J Cancer 100 (11): 1720–1724.
Watanabe A, Taniguchi M, Sasaki S (2003) Induction chemotherapy with docetaxel, cisplatin, fluorouracil and l-leucovorin for locally advanced head and neck cancers: a modified regimen for Japanese patients. Anti-cancer Drugs 14 (10): 801–807.
Acknowledgements
S-1, irinotecan and oxaliplatin were kindly provided by Boryung Co., Ltd, Pfizer Korea/CJ Corp and Sanofi-Aventis Korea, respectively. This study was supported in part by National Cancer Center Grant (NCC-0910010).
Author information
Authors and Affiliations
Corresponding author
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License.
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Kim, S., S Hong, Y., K Shim, E. et al. S-1 plus irinotecan and oxaliplatin for the first-line treatment of patients with metastatic colorectal cancer: a prospective phase II study and pharmacogenetic analysis. Br J Cancer 109, 1420–1427 (2013). https://doi.org/10.1038/bjc.2013.479
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2013.479
Keywords
This article is cited by
-
Effect of UGT1A1, CYP3A and CES Activities on the Pharmacokinetics of Irinotecan and its Metabolites in Patients with UGT1A1 Gene Polymorphisms
European Journal of Drug Metabolism and Pharmacokinetics (2021)
-
The Roles of Common Variation and Somatic Mutation in Cancer Pharmacogenomics
Oncology and Therapy (2019)
-
Individualization of Irinotecan Treatment: A Review of Pharmacokinetics, Pharmacodynamics, and Pharmacogenetics
Clinical Pharmacokinetics (2018)
-
A phase II study of preoperative chemoradiation with tegafur-uracil plus leucovorin for locally advanced rectal cancer with pharmacogenetic analysis
Radiation Oncology (2017)
-
Phase II trial of S-1 plus leucovorin in patients with advanced gastric cancer and clinical prediction by S-1 pharmacogenetic pathway
Cancer Chemotherapy and Pharmacology (2017)