Abstract
Background:
We report updated overall survival (OS) data from study NO16966, which compared capecitabine plus oxaliplatin (XELOX) vs 5-fluorouracil/folinic acid plus oxaliplatin (FOLFOX4) as first-line therapy in metastatic colorectal cancer.
Methods:
NO16966 was a randomised, two-arm, non-inferiority, phase III comparison of XELOX vs FOLFOX4, which was subsequently amended to a 2 × 2 factorial design with further randomisation to bevacizumab or placebo. A planned follow-up exploratory analysis of OS was performed.
Results:
The intent-to-treat (ITT) population comprised 2034 patients (two-arm portion, n=634; 2 × 2 factorial portion, n=1400). For the whole NO16966 study population, median OS was 19.8 months in the pooled XELOX/XELOX-placebo/XELOX-bevacizumab arms vs 19.5 months in the pooled FOLFOX4/FOLFOX4-placebo/FOLFOX4-bevacizumab arms (hazard ratio 0.95 (97.5% CI 0.85–1.06)). In the pooled XELOX/XELOX-placebo arms, median OS was 19.0 vs 18.9 months in the pooled FOLFOX4/FOLFOX4-placebo arms (hazard ratio 0.95 (97.5% CI 0.83–1.09)). FOLFOX4 was associated with more grade 3/4 neutropenia/granulocytopenia and febrile neutropenia than XELOX, and XELOX with more grade 3 diarrhoea and grade 3 hand-foot syndrome than FOLFOX4.
Conclusion:
Updated survival data from study NO16966 show that XELOX is similar to FOLFOX4, confirming the primary analysis of progression-free survival. XELOX can be considered as a routine first-line treatment option for patients with metastatic colorectal cancer.
Similar content being viewed by others
Main
Capecitabine (Xeloda; F. Hoffmann–La Roche, Basel, Switzerland) is an oral fluoropyrimidine with similar efficacy to bolus 5-fluorouracil/folinic acid (5-FU/FA) in the first-line treatment of metastatic colorectal cancer (Hoff et al, 2001; van Cutsem et al, 2001) and as adjuvant therapy for stage III colon cancer (Twelves et al, 2005). The efficacy of capecitabine and a 3-weekly dose of oxaliplatin (XELOX regimen) is also non-inferior to 5-FU/FA plus oxaliplatin (FOLFOX4 or FOLFOX6) in the first- and second-line treatment of patients with metastatic colorectal cancer (Rothenberg et al, 2008; Cassidy et al, 2008a; Ducreux et al, 2011).
The largest of the recent trials evaluating XELOX, NO16966 (XELOX-1), started as a two-arm, randomised, non-inferiority, phase III study designed to compare XELOX with FOLFOX4 as first-line treatment for metastatic colorectal cancer. The protocol was subsequently amended to include the further addition of bevacizumab or placebo using a 2 × 2 factorial design. The study had two co-primary objectives: (1) to evaluate the non-inferiority of XELOX ± bevacizumab vs FOLFOX4 ± bevacizumab; and (2) to evaluate the superiority of bevacizumab vs placebo when combined with oxaliplatin-based chemotherapy (i.e., XELOX or FOLFOX4). The results pertaining to both objectives have been previously published (Saltz et al, 2008; Cassidy et al, 2008a). The present paper describes an updated analysis of overall survival (OS) for the comparison of XELOX/XELOX-placebo/XELOX-bevacizumab vs FOLFOX4/FOLFOX4-placebo/FOLFOX4-bevacizumab from study NO16966.
Patients and methods
The methods of this trial have been described in detail previously (Saltz et al, 2008; Cassidy et al, 2008a). The study was performed in accordance with the Declaration of Helsinki and Good Clinical Practice Guidelines. Written informed consent was obtained from all patients participating in the study. Approval of the protocol was obtained from an independent ethics committee or institutional review board of each site.
Patient population
Patients ⩾18 years of age with histologically confirmed, unresectable metastatic colorectal cancer (⩾1 unidimensionally measurable lesion), an ECOG performance status of ⩽1 and a life expectancy of >3 months were eligible. No previous systemic therapy for metastatic disease or previous oxaliplatin or bevacizumab were allowed. Patients were required to have adequate haematologic/clotting, hepatic and renal function. Patients were excluded if they had any of the following: clinically significant cardiovascular disease; clinically detectable ascites; use of full-dose anticoagulants or thrombolytics; known CNS metastases; serious non-healing wound, ulcer or bone fracture; known bleeding diathesis or coagulopathy; and proteinuria ⩾500 mg per 24 h.
Treatment plan
Patients were randomly assigned to treatment using an interactive voice response system. Randomisation was stratified by region, ECOG performance status, liver as a metastatic site, number of metastatic sites (organs) and alkaline phosphatase level.
XELOX consisted of a 2-h intravenous infusion of oxaliplatin 130 mg m–2 on day 1 plus oral capecitabine 1000 mg m–2 twice daily for 2 weeks as a 3-week cycle. The first dose of capecitabine was given in the evening of day 1 and the last dose on the morning of day 15. The FOLFOX4 regimen has been previously described (de Gramont et al, 2000). After amendment of the study protocol, bevacizumab (7.5 mg kg–1 every third week) or placebo was added to XELOX, and bevacizumab (5 mg kg–1 every second week) or placebo to FOLFOX4. Bevacizumab or placebo was given as a 30- to 90-min intravenous infusion on day 1 of each cycle before oxaliplatin.
Treatment was continued until disease progression or for 48 weeks, whichever came first (study treatment phase). Patients who completed the 48-week treatment phase without disease progression were eligible to continue treatment until progression (post-study treatment phase). Patients whose tumour became operable, and for whom resection was performed, were allowed to enter the post-study treatment phase.
Assessments
Tumour assessments (CT scan, MRI) were performed within 28 days before starting study treatment and repeated after every two XELOX cycles and every three FOLFOX4 cycles (i.e., every sixth week in both arms), and at the end of treatment. After completion of study treatment, patients were followed every 3 months until disease progression and/or death.
Patients were evaluated for adverse events during therapy and until 28 days after the last study drug dose. Adverse events were graded according to National Cancer Institute Common Toxicity Criteria (NCI-CTC), version 3. Predefined adverse events of special interest for chemotherapy were: grade 3/4 neutropenia/granulocytopenia; grade 3/4 neurosensory toxicity; grade 3/4 diarrhoea; grade 3/4 vomiting/nausea; grade 3/4 stomatitis and grade 3 hand-foot syndrome.
Statistical analysis
The intent-to-treat (ITT) patient population included all patients who underwent randomisation and signed the informed consent form. The eligible patient population (EPP) was the ITT population minus patients who did not receive at least one dose of study drug, and those patients who violated major protocol inclusion/exclusion criteria. As the results for the EPP population were the same as for the ITT population, ITT data only will be presented in this paper. The safety population included all patients receiving at least one dose of study drug.
Overall survival was defined as the time from the date of randomisation to the date of death. Patients who were not reported as having died at the time of the analysis were censored using the date they were last known to be alive. Overall survival was analysed using a Cox model and presented as Kaplan–Meier estimates with hazard ratios (HRs) and 97.5% confidence intervals (CIs).
The primary analysis of NO16966 was event driven and was performed on 31 January 2006 when 1200 progression-free survival events had occurred in the EPP; this approach ensured 90% power at an α level of 2.5 (Saltz et al, 2008). A further planned follow-up analysis of OS was performed at the time of the 4-month safety update.
As the study was not powered for formal testing of non-inferiority for OS, the OS analysis is exploratory and the results described by Kaplan–Meier estimates with HRs and 97.5% CIs. An additional exploratory analysis of OS was performed to control for any possible crossover effects of FOLFOX in patients who received XELOX as their first-line regimen. In this analysis, patients in the XELOX arms who received FOLFOX4 or similar regimen as second-line therapy were censored.
Results
Patient population
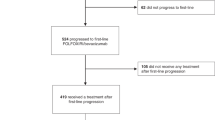
Between July 2003 and May 2004, 634 patients were randomised in the two-arm portion of the study. Between February 2004 and February 2005, a further 1400 patients were randomised in the 2 × 2 factorial part of the study. Overall, 2034 patients made up the ITT population (Figure 1). The baseline demographic and clinical characteristics were well balanced between treatment arms (Table 1).
Treatment exposure and second-line therapy
The median dose intensities (ratio of dose received to dose planned) of 5-FU, capecitabine, oxaliplatin and bevacizumab were ⩾0.89 in all treatment arms. The median number of cycles administered was 11 (range 1–24) in the FOLFOX4/FOLFOX4-placebo group, 12 (range 1–25) in the FOLFOX4-bevacizumab group, 7 (range 1–18) in the XELOX/XELOX-placebo group and 8 (range 1–17) in the XELOX-bevacizumab group.
There were no major imbalances between the treatment groups with respect to the use of second-line therapy: XELOX-containing arms (65%) and FOLFOX4-containing arms (70%). The agents most commonly used were: irinotecan (56% with FOLFOX4 vs 53% with XELOX); 5-FU (41 vs 34%); capecitabine (19 vs 14%); cetuximab (20 vs 18%); and bevacizumab (10 vs 10%).
Overall survival
The OS data as at 31 July 2008 in the ITT population are shown in Table 2. The corresponding Kaplan–Meier curves for OS are shown in Figure 2.
For the whole NO16966 study population, median OS was 19.8 months in the pooled XELOX/XELOX-placebo/XELOX-bevacizumab arms vs 19.5 months in the pooled FOLFOX4/FOLFOX4-placebo/FOLFOX4-bevacizumab arms, with a corresponding HR of 0.95 (97.5% CI 0.85–1.06).
In the pooled XELOX/XELOX-placebo arms, median OS was 19.0 vs 18.9 months in the pooled FOLFOX4/FOLFOX4-placebo arms, with a corresponding HR of 0.95 (97.5% CI 0.83–1.09).
In the XELOX-bevacizumab arm, median OS was 21.6 vs 21.0 months in the FOLFOX4-bevacizumab arm, with a corresponding HR of 0.95 (97.5% CI 0.78–1.15).
In the XELOX arm, median OS was 18.8 vs 17.7 months in the FOLFOX4 arm, with a corresponding HR of 0.87 (97.5% CI 0.72–1.05). FOLFOX4 or a similar regimen was given to 8% of patients in the pooled XELOX arms as second-line therapy (XELOX, n=15, XELOX-placebo, n=38, XELOX-bevacizumab, n=29). After censoring these patients, the median OS was 18.9 months in the pooled XELOX/XELOX-placebo arms and 18.9 months in the pooled FOLFOX4/FOLFOX4-placebo arms, with a corresponding HR of 0.94 (97.5% CI 0.82–1.08), and 21.6 months in the XELOX-bevacizumab arm and 21.0 months in the FOLFOX4-bevacizumab arm (HR=0.93; 97.5% CI 0.76–1.13).
Safety
For the updated safety assessment of XELOX vs FOLFOX4, patients in the pooled XELOX/XELOX-placebo (n=655) and pooled FOLFOX-4/FOLFOX4-placebo (n=648) arms were compared. The updated safety analysis showed that little had changed since the previous analysis (Cassidy et al, 2008a). Predefined adverse events of special interest and key events pooled by body system are presented in Table 3.
In general, XELOX and FOLFOX4 had a similar profile of adverse events. The most common adverse events were gastrointestinal (i.e., diarrhoea, nausea, vomiting and stomatitis) and neurosensory toxicities (i.e., paraesthesia and peripheral neuropathy). However, there were differences between the two regimens in the rates at which key events occurred. FOLFOX4/FOLFOX4-placebo was associated with more grade 3/4 neutropenia/granulocytopenia (44%) and febrile neutropenia (5%) than XELOX/XELOX-placebo (7 and <1%, respectively). Conversely, XELOX/XELOX-placebo was associated with more hand-foot syndrome (all-grade, 31 vs 11%; grade 3, 6 vs 1%) and diarrhoea (all-grade, 66 vs 61%; grade 3/4, 20 vs 11%) than FOLFOX4/FOLFOX4-placebo, although the rate of grade 4 diarrhoea was 1% with both regimens. Rates of grade 3/4 neurosensory toxicity were similar with both regimens (17%). Cardiac disorders were reported in 6 (1%) XELOX/XELOX-placebo recipients and 9 (1%) FOLFOX-4/FOLFOX4-placebo recipients. The addition of bevacizumab did not alter the similarities and differences in safety profiles between XELOX and FOLFOX4 (Table 4).
Treatment-related mortality up to 28 days after the last treatment dose was documented in 11 (1.7%) FOLFOX4/FOLFOX4-placebo patients and in 15 (2.3%) XELOX/XELOX-placebo patients. The respective 60-day all-cause mortality rates were 2.3% (n=15) and 3.4% (n=22).
Discussion
The primary efficacy analysis of study NO16966 showed that XELOX is non-inferior to FOLFOX4 in terms of progression-free survival, OS and overall response rate in the first-line treatment of patients with metastatic colorectal cancer (Cassidy et al, 2008a). This updated analysis of OS again demonstrates that XELOX and FOLFOX4 have similar efficacy and supports the primary efficacy findings. It is also notable that both XELOX and FOLFOX4 were similar in terms of OS after the addition of bevacizumab.
Overall survival is the most clinically meaningful and objective measure of efficacy in patients with cancer. However, potential differences between study treatments can be masked by second-line and later lines of chemotherapy when this end point is used (Di Leo et al, 2004). In study NO16966, there were no restrictions regarding crossover or salvage therapies after the completion of study treatment. It is therefore possible that crossover to the alternate study treatment was a confounding factor in the present analysis. To allow for this, we performed a separate analysis in which all patients randomised to XELOX and who received FOLFOX as second-line therapy were censored. The results were consistent with those obtained in the ITT population and again support the similar efficacy of XELOX vs FOLFOX4.
The question of whether or not capecitabine is non-inferior to 5-FU/FA when given in combination with oxaliplatin in metastatic colorectal cancer has now been addressed in six different randomised phase III trials (Díaz-Rubio et al, 2007; Porschen et al, 2007; Rothenberg et al, 2008; Cassidy et al, 2008a; Comella et al, 2009; Ducreux et al, 2011), of which NO16966 is the largest. The other five studies, which involved 300–600 patients each, were largely supportive of NO16966. In three of the studies, the efficacy of XELOX or OXXEL was shown to be similar to that of 5-FU/FA-oxaliplatin regimens (Rothenberg et al, 2008; Comella et al, 2009; Ducreux et al, 2011), whereas the remaining two were inconclusive with regard to non-inferiority (Díaz-Rubio et al, 2007; Porschen et al, 2007). Since the completion of the phase III trials, three separate meta-analyses of relevant studies comparing capecitabine or 5-FU/FA plus oxaliplatin in patients with metastatic colorectal cancer have been performed (Arkenau et al, 2008; Cassidy et al, 2008b; Cuppone et al, 2008). Even though each meta-analysis included a different selection of phase II and III studies, the outcomes were very similar with respect to both progression-free survival (HR/relative risk 0.98–1.04) and OS (1.02–1.04). Thus, there is now strong evidence to support the non-inferiority of capecitabine when used in combination with oxaliplatin vs infusional 5-FU-based oxaliplatin regimens in the treatment of patients with metastatic colorectal cancer, both in the first- and second-line settings.
It is therefore likely that other considerations, such as tolerability profile, convenience, patient preference and cost, will assume greater importance when selecting the fluoropyrimidine backbone of a chemotherapy regimen. With regard to tolerability, both XELOX and FOLFOX have a similar profile of adverse events, but XELOX is associated with more grade 3 diarrhoea and hand-foot syndrome, whereas FOLFOX is associated with more grade 3/4 neutropenia and febrile neutropenia (Rothenberg et al, 2008; Ducreux et al, 2011). This is supported by the updated safety data from NO16966 in the present paper.
In terms of convenience, XELOX requires fewer planned office visits than the FOLFOX regimens because oxaliplatin is administered every 3 weeks (rather than every 2 weeks) and because capecitabine is taken orally. This is supported by resource use data from NO16966, which showed that the need for drug administration visits, central venous access and patient travel and time were reduced with XELOX vs FOLFOX4 (Scheithauer et al, 2007). When costs were assigned to these data, the total direct costs of both regimens were similar, whereas the indirect costs of XELOX were considerably less than those of FOLFOX4 (Garrison et al, 2007). Similar observations were made in a cost comparison of capecitabine ± oxaliplatin vs 5-FU ± oxaliplatin based on a retrospective analysis of a US medical claims database (Chu et al, 2009). Modified FOLFOX regimens, which involve a single 46- to 48-h infusion of 5-FU, are likely to be less costly than unmodified FOLFOX regimens (Garrison et al, 2007), although a complete assessment vs XELOX has yet to be performed.
In conclusion, updated survival data from study NO16966 show that XELOX is similar to FOLFOX4, confirming the primary analysis of progression-free survival. XELOX can be considered as a routine first-line treatment option for patients with metastatic colorectal cancer.
Change history
29 March 2012
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Arkenau HT, Arnold D, Cassidy J, Diaz-Rubio E, Douillard JY, Hochster H, Martoni A, Grothey A, Hinke A, Schmiegel W, Schmoll HJ, Porschen R (2008) Efficacy of oxaliplatin plus capecitabine or infusional fluorouracil/leucovorin in patients with metastatic colorectal cancer: a pooled analysis of randomized trials. J Clin Oncol 26: 5910–5917
Cassidy J, Clarke S, Diaz Rubio E, Scheithauer W, Figer A, Wong R, Koski S, Lichinitser M, Yang TS, Rivera F, Couture F, Sirzén F, Saltz L (2008a) A randomized phase III study of capecitabine plus oxaliplatin (XELOX) versus fluorouracil/folinic acid plus oxaliplatin (FOLFOX-4) as first-line therapy for metastatic colorectal cancer. J Clin Oncol 26: 2006–2012
Cassidy J, Clarke S, Diaz-Rubio E, Scheithauer W, Figer A, Wong R, Koski S, Sirzen F, Gilberg F, Saltz L (2008b) XELOX vs FOLFOX4: update of efficacy results from XELOX-1/NO16966, a randomized phase III trial of first-line treatment for patients (pts) with metastatic colorectal cancer (MCRC). Presented at the ASCO GI Cancers Symposium: Orlando, Florida, USA, 25–27 January (abstract 341)
Chu E, Schulman KL, Zelt S (2009) Costs associated with complications are lower with capecitabine than with 5-fluorouracil in patients with colorectal cancer. Cancer 115: 1412–1423
Comella P, Massidda B, Filippelli G, Farris A, Natale D, Barberis G, Maiorino L, Palmeri S, Condemi G, Southern Italy Cooperative Oncology Group (2009) Randomised trial comparing biweekly oxaliplatin plus oral capecitabine versus oxaliplatin plus i.v. bolus fluorouracil/leucovorin in metastatic colorectal cancer patients: results of the Southern Italy Cooperative Oncology study 0401. J Cancer Res Clin Oncol 135: 217–226
Cuppone F, Bria E, Sperduti I, Di Maio M, Carlini P, Milella M, Cognetti F, Terzoli E, Giannarelli D (2008) Capecitabine (CAP) versus 5-fluorouracil (FU) in combination with oxaliplatin (OX) as 1st-line chemotherapy (CT) for advanced colorectal cancer (ACRC): meta-analysis of randomized clinical trials (RCT). J Clin Oncol 26: 192s (Suppl; abstract 4056)
de Gramont A, Figer A, Seymour M, Homerin M, Hmissi A, Cassidy J, Boni C, Cortes-Funes H, Cervantes A, Freyer G, Papamichael D, Le Bail N, Louvet C, Hendler D, de Braud F, Wilson C, Morvan F, Bonetti A (2000) Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol 18: 2938–2947
Díaz-Rubio E, Tabernero J, Gómez-España A, Massutí B, Sastre J, Chaves M, Abad A, Carrato A, Queralt B, Reina JJ, Maurel J, González-Flores E, Aparicio J, Rivera F, Losa F, Aranda E, Spanish Cooperative Group for the Treatment of Digestive Tumors Trial (2007) Phase III study of capecitabine plus oxaliplatin versus continuous infusion fluorouracil plus oxaliplatin as first-line therapy in metastatic colorectal cancer: final report of the Spanish Cooperative Group for the Treatment of Digestive Tumors trial. J Clin Oncol 25: 4224–4230
Di Leo A, Buyse M, Bleiberg H (2004) Is overall survival a realistic primary end point in advanced colorectal cancer studies? A critical assessment based on four clinical trials comparing fluorouracil plus leucovorin with the same treatment combined either with oxaliplatin or with CPT-11. Ann Oncol 15: 545–549
Ducreux M, Bennouna J, Hebbar M, Ychou M, Lledo G, Conroy T, Adenis A, Faroux R, Rebischung C, Bergougnoux L, Kockler L, Douillard JY, GI Group of the French Anti-Cancer Centers (2011) Capecitabine plus oxaliplatin (XELOX) versus 5-fluorouracil/leucovorin plus oxaliplatin (FOLFOX-6) as first-line treatment for metastatic colorectal cancer. Int J Cancer 128: 682–690
Garrison L, Cassidy J, Saleh M, Lee F, Mena R, Fuloria J, Chang V, Ervin T, Stella P, Saltz L (2007) Cost comparison of XELOX compared to FOLFOX4 with or without bevacizumab (bev) in metastatic colorectal cancer. J Clin Oncol 25: 18S (Suppl, abstract 4074)
Hoff PM, Ansari R, Batist G, Cox J, Kocha W, Kuperminc M, Maroun J, Walde D, Weaver C, Harrison E, Burger HU, Osterwalder B, Wong AO, Wong R (2001) Comparison of oral capecitabine versus intravenous fluorouracil plus leucovorin as first-line treatment in 605 patients with metastatic colorectal cancer: results of a randomized phase III study. J Clin Oncol 19: 2282–2292
Porschen R, Arkenau H-T, Kubicka S, Greil R, Seufferlein T, Freier W, Kretzschmar A, Graeven U, Grothey A, Hinke A, Schmiegel W, Schmoll HJ, AIO Colorectal Study Group (2007) Phase III study of capecitabine plus oxaliplatin compared with fluorouracil and leucovorin plus oxaliplatin in metastatic colorectal cancer: a final report of the AIO Colorectal Study Group. J Clin Oncol 25: 4217–4223
Rothenberg ML, Cox JV, Butts C, Navarro M, Bang YJ, Goel R, Gollins S, Siu LL, Laguerre S, Cunningham D (2008) Capecitabine plus oxaliplatin (XELOX) versus 5-fluorouracil/folinic acid plus oxaliplatin (FOLFOX-4) as second-line therapy in metastatic colorectal cancer: a randomized phase III noninferiority study. Ann Oncol 19: 1720–1726
Saltz L, Clarke S, Díaz-Rubio E, Scheithauer W, Figer A, Wong R, Koski S, Lichinitser M, Yang TS, Rivera F, Couture F, Sirzén F, Cassidy J (2008) Efficacy and safety of bevacizumab in combination with oxaliplatin-based chemotherapy as first-line therapy in metastatic colorectal cancer: a randomized phase III study. J Clin Oncol 26: 2013–2019
Scheithauer W, Cassidy J, Figer A, Wong R, Koski S, Lichinitser M, Yang T, Clarke S, Diaz-Rubio E, Garrison L (2007) A comparison of medical resource use for 4 chemotherapy regimens as first-line treatment for metastatic colorectal cancer (MCRC): XELOX vs. FOLFOX4 ± bevacizumab (A). J Clin Oncol 25: 18S (Suppl, abstract 4098)
Twelves C, Wong A, Nowacki MP, Abt M, Burris III H, Carrato A, Cassidy J, Cervantes A, Fagerberg J, Georgoulias V, Husseini F, Jodrell D, Koralewski P, Kröning H, Maroun J, Marschner N, McKendrick J, Pawlicki M, Rosso R, Schüller J, Seitz JF, Stabuc B, Tujakowski J, Van Hazel G, Zaluski J, Scheithauer W (2005) Capecitabine as adjuvant treatment for stage III colon cancer. N Engl J Med 352: 2696–2704
Van Cutsem E, Twelves C, Cassidy J, Allman D, Bajetta E, Boyer M, Bugat R, Findlay M, Frings S, Jahn M, McKendrick J, Osterwalder B, Perez-Manga G, Rosso R, Rougier P, Schmiegel WH, Seitz JF, Thompson P, Vieitez JM, Weitzel C, Harper P, Xeloda Colorectal Cancer Study Group (2001) Oral capecitabine compared with intravenous fluorouracil plus leucovorin in patients with metastatic colorectal cancer: results of a large phase III study. J Clin Oncol 19: 4097–4106
Acknowledgements
Financial support for this research was provided by Roche. In addition to the investigators in the author list, we acknowledge the following investigators who also participated in this trial: Australia: S Begbie, I Burns, P Gibbs, D Goldstein, P Mainwaring, J McKendrick, M Michael, N Pavlakis, T Price, M Schwartz, J Shapiro, B Stein, G Van Hazel; Austria: J Thaler; Brazil: C Andrade, G Ismael, A Malzyner; Canada: J-P Ayoub, S Berry, R Burkes, P Dube, B Findlay, C Fitzgerald, A Gurjal, D Jonker, L Kaizer, P Klimo, B Lesperance, P Major, B Melosky, L Siu, M Taylor, K Virik, R Wierzbicki, J Wilson, A Wong; China: F Feng-Yi, Y He, S Jiao, J Li, R Luo, L Pan, S Song, H Wang, J Xiong, B Yu, S-Y Yu; Czech Republic: D Feltl, I Kocakova, M Kuta; Denmark: BV Jenson, P Pfeiffer; Finland: P Bono, P Kellokumpu-Lehtinen, S Pyrhoenen; France: F-X Caroli-Bosc, B Coudert, C Debrigode, J-P Delord, G Des Guetz, J-Y Douillard, E Francois, G Freyer, C Garnier, M Gil Delgado, F Goldwasser, F Husseini, P Michel, S Negrier, X Pivot, P Rougier; Germany: M Clemens, A Hochhaus, T Hoehler, S Kanzler, S Kubicka, F Kullmann, L Mantovani, W-H Schmiegel, H-J Schmoll, R Voigtmann; Guatemala: CE Hernandez-Monroy, LM Zetina Toache; Hong Kong: A Chan; Hungary: M Dank, I Lang, T Pinter, M Szucs; Ireland: D Fennelly, M Keane, J Kennedy, S O’Reilly; Israel: A Beny, A Hubert, A Sella, S Stemmer; Italy: C Boni, S Brugnatelli, S Cascinu, PF Conte, A Contu, S Monfardini, R Rosso, S Salvagni, A Sobrero; Republic of Korea: YS Park; Mexico: G Calderillo, E León; New Zealand: B Robinson; Norway: L Balteskard, T Guren, H Soerbye; Panama: E Diaz-Correa; Portugal: S Barroso, P Cortes; Russian Federation: VP Kharchenko, M Lichinitser, GM Manikhas, V Moiseenko; South Africa: G Cohen, C Kukard, J Raats, C Slabber, D Vorobiof; Spain: JE Ales-Martinez, A Anton Torres, J Aparicio, E Aranda, A Cervantes, P Escudero, J Feliu, C Fernandez-Martos, R Garcia-Carbonero, P Garcia-Alfonso, E Gonzalez Flores, C Gravalos, J Maurel, M Navarro-Garcia, F Rivera, R Salazar, I Sevilla, J Tabernero; Sweden: B Glimelius, H Letocha, U Loenn, D Pedersen; Switzerland: M Borner, A Roth; Taiwan: T-Y Chao, P-M Chen, AL Cheng, T-S Yang; Thailand: S Chakrapee-Sirisuk, AN Kiatikajornthada, A Sookprasert; Turkey: G Demir, E Goker; United Kingdom: E Bessell, P Chakraborti, F Coxon, D Cunningham, S Falk, F Daniel, R Glynne-Jones, M Hill, T Iveson, A Maraveyas, T Maughan, D Rea, L Samuel, C Topham; United States: N Abramson, B Amin, B Bhaskar, L Campos, R Castillo, V Chang, P DeSimone, P Eisenberg, JA Ellerton, T Ervin, G Frenette, J Fuloria, H Gomolin, MGreenblatt, G Gross, WJM Hrushesky, D Irwin, M Kane, D Kapur, B Kasimis, KS Kumar, F-C Lee, MW Lee, L Martin, R Mena, J Neidhart, J Pennacchi, E Poplin, C Redfern, R Reiling, M Saleh, L Schwartzberg, W Sikov, A Solky, P Stella, S Thomas, R Vivacqua, R Yanagihara. Support for third-party writing assistance for this manuscript, furnished by Miller Medical Communications, was provided by F. Hoffmann–La Roche Ltd.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Cassidy, J., Clarke, S., Díaz-Rubio, E. et al. XELOX vs FOLFOX-4 as first-line therapy for metastatic colorectal cancer: NO16966 updated results. Br J Cancer 105, 58–64 (2011). https://doi.org/10.1038/bjc.2011.201
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2011.201
Keywords
This article is cited by
-
In silico analysis to identify miR-1271-5p/PLCB4 (phospholipase C Beta 4) axis mediated oxaliplatin resistance in metastatic colorectal cancer
Scientific Reports (2023)
-
Aflibercept with FOLFIRI in Japanese patients with metastatic colorectal cancer: results of a post-marketing surveillance
International Journal of Clinical Oncology (2023)
-
Intraperitoneal paclitaxel combined with FOLFOX/CAPOX plus bevacizumab for colorectal cancer with peritoneal carcinomatosis (the iPac-02 trial): study protocol of a single arm, multicenter, phase 2 study
International Journal of Colorectal Disease (2023)
-
Effects of vonoprazan and proton pump inhibitors on the efficacy of bevacizumab: a multicentre retrospective study
Clinical and Experimental Medicine (2023)
-
Effectiveness of protocol-based pharmacotherapy management collaboration between hospital and community pharmacists to address capecitabine-related hand–foot syndrome in cancer patients: a retrospective study
Journal of Pharmaceutical Health Care and Sciences (2021)