Abstract
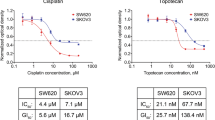
The cytotoxic activity and cross-resistance pattern of the novel topoisomerase I inhibitor topotecan (Topo) were investigated in ten cell lines, representing different mechanisms of cytotoxic drug resistance, and in 218 fresh human tumour samples using the fluorometric microculture cytotoxicity assay (FMCA). Resistance to Topo in the cell lines was associated with expression of the multidrug resistance-associated protein (MRP), whereas the cell lines with P-glycoprotein (P-gp), topoisomerase II and glutathione-associated resistance did not show decreased sensitivity to the drug. Topo was more active in haematological than in solid tumour samples, but substantial activity was observed in carcinomas of the ovary and breast, sarcoma and childhood solid tumours. Cross-resistance to standard drugs representing different mechanisms of action was generally low in patient cells. The effect of Topo was better after longer exposure, but this time-dependent effect was largely abolished when adjustment for in vitro exposure was made. Topo showed activity both in proliferative and non-proliferative cell systems. The results indicate that Topo is insensitive to major mechanisms of resistance except for MRP. Proliferation does not seem to be necessary for the effect of Topo, and no superiority for protracted dosing schedules was observed. The results also suggest that, for example, leukaemias, lymphomas, sarcomas and childhood solid tumours may be suitable targets for future phase II trials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jonsson, E., Fridborg, H., Csóka, K. et al. Cytotoxic activity of topotecan in human tumour cell lines and primary cultures of human tumour cells from patients. Br J Cancer 76, 211–219 (1997). https://doi.org/10.1038/bjc.1997.364
Issue Date:
DOI: https://doi.org/10.1038/bjc.1997.364
This article is cited by
-
Quantification of Unencapsulated Drug in Target Tissues Demonstrates Pharmacological Properties and Therapeutic Effects of Liposomal Topotecan (FF-10850)
Pharmaceutical Research (2024)
-
Transcription regulation of CDKN1A (p21/CIP1/WAF1) by TRF2 is epigenetically controlled through the REST repressor complex
Scientific Reports (2017)
-
Pharmacokinetics, pharmacodynamics and adherence to oral topotecan in myelodysplastic syndromes: a Cancer and Leukemia Group B study
Cancer Chemotherapy and Pharmacology (2006)
-
Reversal of MDR1-associated resistance to topotecan by PAK-200S, a new dihydropyridine analogue, in human cancer cell lines
British Journal of Cancer (1999)