Abstract
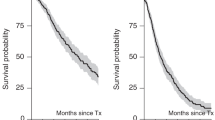
Sixty-three new untreated patients with multiple myeloma under the age of 70 years received C-VAMP induction treatment followed by high-dose intravenous melphalan (200 mg m(-2)) and autologous stem cell transplant, either with marrow [autologous bone marrow transplants (ABMT), n = 26] or with granulocyte colony-stimulating factor (G-CSF)-mobilized stem cells from the blood [peripheral blood stem cell transplants (PBSCT), n = 37]. This was a sequential study and the two groups were not significantly different for all known prognostic variables. The complete remission (CR) rate after high-dose treatment was the same for both groups [ABMT 84% and PBSCT 70%; P = not significant (NS)]. Neutrophil recovery to 0.5 x 10(9) l(-1) occurred at a median of 22 days in the ABMT patients compared with 19 days for the PBSCT patients (P = NS). Platelet recovery to 50 x 10(9) l(-1) was significantly faster in PBSCT patients (19 days vs 33 days; P = 0.0015), and the PBSCT patients spent fewer days in hospital (median 20 vs 27 days; P = 0.00001). There was no difference in the two groups with respect to starting interferon (58 days for ABMT vs 55 days for PBSCT), and tolerance to interferon was identical. The median overall survival (OS) and progression-free survival (PFS) for the PBSCT patients has not yet been reached. The OS in the ABMT patients at 3 years was 76.9% (95% CI 60-93%) compared with 85.3% (95% CI 72-99%) in the PBSCT patients (P = NS), and the PFS at 3 years in the ABMT patients was 53.8% (95% CI 34-73%) and in the PBSCT patients was 57.6% (95% CI 34-81%) (P = NS). The probability of relapse at 3 years was 42.3% in the ABMT arm compared with 40% in the PBSCT patients (P = NS). Thus, PBSCT patients had a faster engraftment and a shorter stay in hospital than ABMT; the survival outcome and probability of relapse was the same for both groups.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Raje, N., Powles, R., Horton, C. et al. Comparison of marrow vs blood-derived stem cells for autografting in previously untreated multiple myeloma. Br J Cancer 75, 1684–1689 (1997). https://doi.org/10.1038/bjc.1997.286
Issue Date:
DOI: https://doi.org/10.1038/bjc.1997.286
This article is cited by
-
Comparison of new patients with Bence-Jones, IgG and IgA myeloma receiving sequential therapy: the need to regard these immunologic subtypes as separate disease entities with specific prognostic criteria
Bone Marrow Transplantation (2001)
-
Acute lymphoblastic leukaemia-type intensive chemotherapy to eliminate minimal residual disease after high-dose melphalan and autologous transplantation in multiple myeloma – a phase I/II feasibility and tolerance study of 17 patients
Bone Marrow Transplantation (2000)
-
The role of autologous transplantation in patients with multiple myeloma aged 65 years and over
Bone Marrow Transplantation (2000)