Abstract
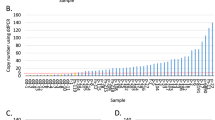
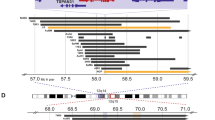
DNA amplification seems to be particularly frequent in human breast tumours and has been associated with cancer evolution and aggressiveness. Recent data indicate that new events should be added to the list, such as the amplifications at chromosome 20q13 or the MDM2 gene. The present work aimed at determining the incidence and clinicopathological signification of these amplifications in a large series of breast and ovarian tumours. We tested 1371 breast and 179 ovarian tumours by Southern blotting and observed amplification of 20q13 in 5.4% breast and 2.8% ovarian carcinomas, whereas MDM2 was found amplified in 5.3% and 3.8% of breast and ovarian tumours respectively. MDM2 RNA expression levels were analysed in a subset of 57 breast tumours and overexpression was observed in 4/57 (7%) of the tumours. Elevated expression levels coincided with amplification of the gene. In breast cancer, 20q13 and MDM2 amplifications seem to define subsets of aggressive tumours. Indeed, 20q13 was correlated to axillary nodal involvement and occurred preferentially in younger patients (< 50 years). Furthermore, 20q13 correlated, as did MDM2 amplification, to aneuploidy. In parallel, we had also tested our tumour DNAs for amplification of CCND1, ERBB-2 and MYC, which made it possible to test for correlations with 20q13 or MDM2 amplifications. Whereas 20q13 showed a very strong correlation to CCND1 amplification, that of MDM2 was prevalent in MYC-amplified tumours. Interestingly, 20q13 and MDM2 amplifications showed some degree of correlation to each other, which may possibly be owing to the fact that both events occurred preferentially in aneuploid tumours. In ovarian cancer, no statistically significant correlation was observed. However, 20q13 amplification occurred preferentially in stage 3 tumours and MDM2 was correlated to ERBB-2 amplification. This may suggest that in ovarian tumours also, 20q13 and MDM2 amplifications occur in late or aggressive cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Courjal, F., Cuny, M., Rodriguez, C. et al. DNA amplifications at 20q13 and MDM2 define distinct subsets of evolved breast and ovarian tumours. Br J Cancer 74, 1984–1989 (1996). https://doi.org/10.1038/bjc.1996.664
Issue Date:
DOI: https://doi.org/10.1038/bjc.1996.664
This article is cited by
-
Gene amplification in ductal carcinoma in situ of the breast
Breast Cancer Research and Treatment (2010)
-
Relationship and prognostic significance of phospho-(serine 166)-murine double minute 2 and Akt activation in node-negative breast cancer with regard to p53 expression
Virchows Archiv (2006)
-
Genetic variability in MCF-7 sublines: evidence of rapid genomic and RNA expression profile modifications
BMC Cancer (2003)
-
Protein elongation factor EEF1A2 is a putative oncogene in ovarian cancer
Nature Genetics (2002)
-
The expression of murine double minute 2 is a favorable prognostic marker in esophageal squamous cell carcinoma without p53 protein accumulation
Annals of Surgical Oncology (2002)