Abstract
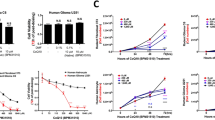
Oxygen radicals induce cytotoxicity via a variety of mechanisms, including DNA damage, lipid peroxidation and protein oxidation. Here, we explore the use of a polyethylene glycol (PEG)-stabilised enzyme capable of producing reactive oxygen species (ROS), glucose oxidase (GO), for the purpose of harnessing the cytotoxic potential of ROS for treating solid tumours. PEG-GO (200 U), administered by two intratumoral injections 3 h apart, produced a significant growth delay in subcutaneous rat 9L gliomas as compared with control animals receiving heat-denatured PEG-GO. Rats were protected from systemic toxicity by subsequent i.v. administration of PEG-superoxide dismutase (PEG-SOD) and PEG-catalase. In vivo tumour metabolic changes, monitored using 31P magnetic resonance spectroscopy (31P-MRS) 6 h following initial administration of PEG-GO, revealed a 96 +/- 2% reduction in the ATP/Pi ratio and a 0.72 +/- 0.10 unit decline in intracellular pH. A 3-fold sensitisation of 9L glioma cells in vitro to hydrogen peroxide could be achieved by a 24 h preincubation with buthionine sulphoximine (BSO). This study suggests that oxidation therapy, the use of an intratumoral ROS-generating enzyme system for the treatment of solid tumours, is a promising area which warrants further exploration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ben-Yoseph, O., Ross, B. Oxidation therapy: the use of a reactive oxygen species-generating enzyme system for tumour treatment. Br J Cancer 70, 1131–1135 (1994). https://doi.org/10.1038/bjc.1994.460
Issue Date:
DOI: https://doi.org/10.1038/bjc.1994.460
This article is cited by
-
Pharmaceutical nanoformulation strategies to spatiotemporally manipulate oxidative stress for improving cancer therapies — exemplified by polyunsaturated fatty acids and other ROS-modulating agents
Drug Delivery and Translational Research (2022)
-
Recent topics and advanced therapies in chronic granulomatous disease
Human Cell (2022)
-
Nanocomposites as biomolecules delivery agents in nanomedicine
Journal of Nanobiotechnology (2019)
-
Research Advances on Apoptosis Caused by Quantum Dots
Biological Trace Element Research (2014)
-
The E6AP E3 ubiquitin ligase regulates the cellular response to oxidative stress
Oncogene (2013)