Abstract
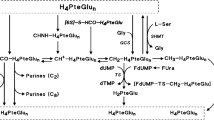
Inhibition of clonogenic potential by the glycinamideribonucleosyl transformylase inhibitor 5,10-dideazatetrahydrofolic acid (DDATHF, Lometrexol) was evaluated in vitro in a human ovarian carcinoma cell line, SW626. Drug-induced inhibition of clonogenic potential is a function of the dose and time of exposure and is independent of the formation of DNA single-strand breaks or de novo synthesis of protein. Simultaneous treatment with 100 microM hypoxanthine completely prevented the inhibition of clonogenic potential caused by 0.5 microM DDATHF. DDATHF blocked cells in the early-middle S-phases of the cell cycle, and there was a corresponding marked reduction in the rate of DNA synthesis after drug withdrawal. The cytotoxic potential of DDATHF was modulated by the folic acid concentration present in the medium. In a medium containing 0.22 microM folic acid, DDATHF cytotoxicity was at least 100 times that in a regular medium containing 2.22 microM folic acid, levels which, however, are about 100 times those found in human plasma. DDATHF cytotoxicity differed moderately when folic acid concentrations varied between 0.22 and 0 microM, suggesting that folic acid does not necessarily antagonise DDATHF anti-tumour activity. Folinic acid at a concentration as low as 0.1 microM can completely rescue cells when given simultaneously with 0.5 microM DDATHF. When folinic acid was given 24 h after DDATHF, a reversal of cytotoxicity was observed at 0.5 and 1 microM, but to a much lesser extent than simultaneous treatment. When folinic acid was added after 48 or 72 h of DDATHF washout, even at a high concentration and for a long time, no reduction in DDATHF cytotoxicity was found. In conclusion, the study highlights the modulation of DDATHF cytotoxicity by folic acid or by folinic acid and provides further rationale for in vivo clinical investigation with these combinations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Erba, E., Sen, S., Sessa, C. et al. Mechanism of cytotoxicity of 5,10-dideazatetrahydrofolic acid in human ovarian carcinoma cells in vitro and modulation of the drug activity by folic or folinic acid. Br J Cancer 69, 205–211 (1994). https://doi.org/10.1038/bjc.1994.40
Issue Date:
DOI: https://doi.org/10.1038/bjc.1994.40
This article is cited by
-
Mode of action of thiocoraline, a natural marine compound with anti-tumour activity
British Journal of Cancer (1999)
-
Automatic quantitation of cell colonies on petri dishes by computerized image analysis
Cytotechnology (1995)