Abstract
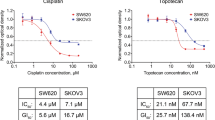
The cytotoxicity of paclitaxel against eight human tumour cell lines has been studied with in vitro clonogenic assays. The fraction of surviving cells fell sharply after exposure for 24 h to paclitaxel concentrations ranging from 2 to 20 nM; the paclitaxel IC50 was found to range between 2.5 and 7.5 nM. Increasing the paclitaxel concentration above 50 nM, however, resulted in no additional cytotoxicity after a 24 h drug exposure. Cells incubated in very high concentrations of paclitaxel (10,000 nM) had an increase in survival compared with cells treated with lower concentrations of the drug. Prolonging the time of exposure of cells to paclitaxel from 24 to 72 h increased cytotoxicity from 5 to 200 fold in different cell lines. Exponentially growing cells were more sensitive to paclitaxel than were cells in the plateau phase of growth. Cremophor EL, the diluent in which the clinical preparation of paclitaxel is formulated, antagonised paclitaxel at concentrations of 0.135% (v/v). These data suggest that paclitaxel will be most effective clinically when there is prolonged exposure of tumour to the drug. Further, it appears that modest concentrations (i.e., 50 nM) should be as effective as higher concentrations of paclitaxel. Finally, we have noted that Cremophor EL is a biologically active diluent and, at high concentrations (0.135% v/v), can antagonise paclitaxel cytotoxicity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Liebmann, J., Cook, J., Lipschultz, C. et al. Cytotoxic studies of paclitaxel (Taxol®) in human tumour cell lines. Br J Cancer 68, 1104–1109 (1993). https://doi.org/10.1038/bjc.1993.488
Issue Date:
DOI: https://doi.org/10.1038/bjc.1993.488
This article is cited by
-
Nanocomposite formulation for a sustained release of free drug and drug-loaded responsive nanoparticles: an approach for a local therapy of glioblastoma multiforme
Scientific Reports (2023)
-
Co-axial electrospraying of injectable multi-cancer drugs nanocapsules with polymer shells for targeting aggressive breast cancers
Cancer Nanotechnology (2022)
-
The deubiquitinating enzyme complex BRISC regulates Aurora B activation via lysine-63-linked ubiquitination in mitosis
Communications Biology (2022)
-
Analysis of clonogenic growth in vitro
Nature Protocols (2021)
-
Surface functionalisation of poly-APO-b-polyol ester cross-linked copolymers as core–shell nanoparticles for targeted breast cancer therapy
Scientific Reports (2020)