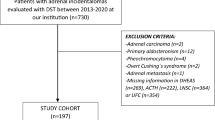
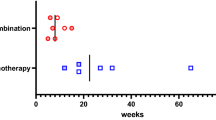
Abstract
Lower pre-chemotherapy night time cortisol excretion predicted more severe cisplatin induced nausea and vomiting in 42 ovarian cancer patients receiving ondansetron as a single antiemetic agent. Dexamethasone administration added to the antiemetic effect of ondansetron principally in patients who had low excretion of cortisol.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hursti, T., Fredrikson, M., Steineck, G. et al. Endogenous cortisol exerts antiemetic effect similar to that of exogenous corticosteroids. Br J Cancer 68, 112–114 (1993). https://doi.org/10.1038/bjc.1993.295
Issue Date:
DOI: https://doi.org/10.1038/bjc.1993.295
This article is cited by
-
Glucocorticoid resistance of migration and gene expression in a daughter MDA-MB-231 breast tumour cell line selected for high metastatic potential
Scientific Reports (2017)
-
Palonosetron plus single-dose dexamethasone for the prevention of nausea and vomiting in women receiving anthracycline/cyclophosphamide-containing chemotherapy: meta-analysis of individual patient data examining the effect of age on outcome in two phase III trials
Supportive Care in Cancer (2013)
-
Role of dexamethasone dosage in combination with 5-HT3 antagonists for prophylaxis of acute chemotherapy-induced nausea and vomiting
British Journal of Cancer (1999)
-
Single high-dose dexamethasone improves the effect of ondansetron on acute chemotherapy-induced nausea and vomiting but impairs the control of delayed symptoms
Supportive Care in Cancer (1996)