Abstract
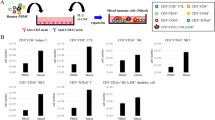
A simple method for the rapid expansion of human CD4+ T cells with both helper and killer functions was established. CD4+ T cells separated from peripheral blood mononuclear cells using immunomagnetic beads were stimulated with immobilised OKT-3 monoclonal antibody (mAb) plus recombinant interleukin 2 (rIL-2) in 96 well culture plates. After 6 day-culture, the CD4+ T cells were restimulated by immobilised OKT-3 mAb for an additional 24 h, then inoculated into concentrated rotary-tissue culture bag and cultured for further 9 days. This procedure yielded a 3000-fold increase in cell number (about 3-5 x 10(9) per bag). Most of the cells (over 96%) continued to express CD4+ antigen and retained their capacity to produce IL-2. The activated CD4+ T cells showed marked cytotoxicity against Fc receptor positive tumour cells in the presence of OKT-3 mAb. Moreover, we succeeded in a specific targeting of the expanded CD4+ helper/killer T cells to c-erb B-2 positive tumour cells by means of anti-CD3 x anti-c-erb B-2 bispecific antibody. These results suggested that our established simple system will be available for the expansion of large number of CD4+ helper/killer T cells which may provide an efficient strategy for adoptive tumour immunotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nakamura, Y., Tokuda, Y., Iwasawa, M. et al. Large-scale culture system of human CD4+ helper/killer T cells for the application to adoptive tumour immunotherapy. Br J Cancer 66, 20–26 (1992). https://doi.org/10.1038/bjc.1992.210
Issue Date:
DOI: https://doi.org/10.1038/bjc.1992.210
This article is cited by
-
Artificial capillary culture: expansion and retroviral transduction of CD4+ T-lymphocytes for clinical application
Gene Therapy (1999)
-
Primary cytotoxicity of SEB-activated human PBMC and separated CD8+and CD4+T lymphocytes elicited by two different stimulation protocols
Immunology and Cell Biology (1996)