Abstract
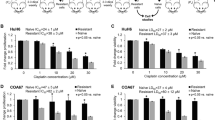
NIH3T3 cells transfected with c-H-ras and/or c-myc genes were examined for differences in drug sensitivity. The five transfectants used were N8, NIH3T3-nm-1, pT22-3-nm-2, pP1-4 and pT22-3. They were transfected with pKOneo alone, pKOneo and c-myc, pKOneo and c-myc plus activated c-H-ras, normal c-H-ras and activated c-H-ras genes, respectively. The IC50s of cisplatin, 4-hydroperoxycyclophosphamide, adriamycin, melphalan, and CPT-11 were significantly higher for NIH3T3-nm-1 abd pT22-3-nm-2 than for the parental NIH3T3 and N8 cells. Transfection with normal and activated C-H-ras oncogenes only led to increases in the IC50s of alkylating agents. There was no significant difference between the IC50s of N8 and those of NIH3T3 parental cells to any of these anticancer agents. These results strongly suggest that the expression of the c-myc gene plays a role in the acquisition of drug resistance. The c-myc gene may therefore provide us with an important clue in determining the mechanism of drug resistance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Niimi, S., Nakagawa, K., Yokota, J. et al. Resistance to anticancer drugs in NIH3T3 cells transfected with c-myc and/or c-H-ras genes. Br J Cancer 63, 237–241 (1991). https://doi.org/10.1038/bjc.1991.56
Issue Date:
DOI: https://doi.org/10.1038/bjc.1991.56
This article is cited by
-
Cisplatin exposure causes c-Myc-dependent resistance to CDK4/6 inhibition in HPV-negative head and neck squamous cell carcinoma
Cell Death & Disease (2019)
-
Are RAS mutations predictive markers of resistance to standard chemotherapy?
Nature Reviews Clinical Oncology (2009)
-
Prognostic significance of K- ras and TP53 mutations in the role of adjuvant chemotherapy on survival in patients with dukes C colon cancer
Diseases of the Colon & Rectum (2001)
-
MycN sensitizes neuroblastoma cells for drug-induced apoptosis
Oncogene (1999)
-
Topographic genotyping of colorectal carcinoma: From a molecular carcinogenesis model to clinical relevance
Annals of Surgical Oncology (1997)