Abstract
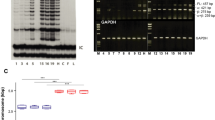
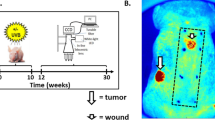
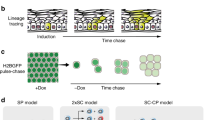
Epidermal tumourigenesis can be achieved in rodents by the application of a single subthreshold dose of a carcinogen (initiation) followed by repeated applications of a tumour promoter such as 12-0-tetradecanoyl phorbol, 13-acetate (TPA). TPA induces terminal differentiation in the majority of epidermal keratinocytes in vitro. However, transformed keratinocytes respond weakly to this terminal differentiation signal, and it is suggested that this property allows initiated cells and their progeny to obtain a selective advantage over their normal counterparts during promotion of papilloma formation by TPA. New data are reviewed which suggest that a putative wound hormone TGF-beta has similar differential effects on normal and transformed epithelial cells to those of TPA. It is proposed that the release of TGF-beta from platelets following deep skin wounding may be an explanation as to why wounding is a promoting stimulus but milder forms of epidermal injury are not. Weakly promoting hyperplasiogenic agents are also discussed within the context of a selection theory of tumour promotion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Rights and permissions
About this article
Cite this article
Parkinson, E. Defective responses of transformed keratinocytes to terminal differentiation stimuli. Their role in epidermal tumour promotion by phorbol esters and by deep skin wounding. Br J Cancer 52, 479–493 (1985). https://doi.org/10.1038/bjc.1985.219
Issue Date:
DOI: https://doi.org/10.1038/bjc.1985.219
This article is cited by
-
Integrative multicellular biological modeling: a case study of 3D epidermal development using GPU algorithms
BMC Systems Biology (2010)
-
Multi-stage chemical carcinogenesis in mouse skin: Fundamentals and applications
Nature Protocols (2009)
-
Selective clonal expansion and microenvironmental permissiveness in tobacco carcinogenesis
Oncogene (2002)
-
Regulation and clinical implications of corneal epithelial stem cells
Molecular Biology Reports (1996)