Abstract
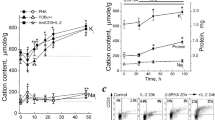
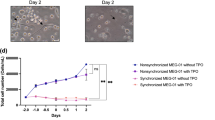
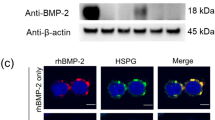
Sodium polyanethol sulphonate (SPS) when incorporated into rat erythrocyte lysate (REL) containing semi-solid agar medium at 1 mg ml-1. markedly enhanced colony formation by a number of anchorage-independent cell lines. REL usually needed to be included for the expression of SPS induced potentiation as in its absence SPS was generally cytotoxic. Studies suggested that SPS reduced the lag prior to colony initiation resulting in the earlier appearance of colonies and in a higher cloning efficiency. The effectiveness of SPS in potentiating colony formation by responsive cell lines was markedly influenced by the species of serum and to a lesser extent by differences between individual batches. Enhancement by SPS was greater with poorer foetal calf serum (FCS) batches than with better. This effect may have been partly due to SPS interfering with the action of a growth inhibiting serum component, possibly a lipoprotein. Studies in which delipidated FCS was substituted for normal FCS suggested that SPS was also able to compensate for the lack of a growth-promoting lipid component. Binding studies showed that initially 125I-SPS bound equally well at 4 degrees C and 37 degrees C with continued labelling occurring only at 37 degrees C. Autoradiography of cells labelled at 37 degrees C for 24 h revealed the presence of intracytoplasmic 125I-SPS.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Rights and permissions
About this article
Cite this article
Sheridan, J., Bishop, C., Simmons, R. et al. Potentiation of anchorage-independent colony formation by sodium polyanethol sulphonate. Br J Cancer 50, 633–645 (1984). https://doi.org/10.1038/bjc.1984.230
Issue Date:
DOI: https://doi.org/10.1038/bjc.1984.230