Abstract
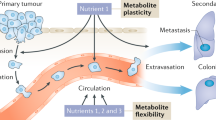
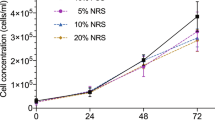
The effect of the Walker 256 rat carcinoma, growing at a site remote from the liver, on liver glycogen synthesis has been determined. Although the mechanism has not been defined it has been shown that the deposition of liver glycogen in response to a glucose load is inhibited in tumour bearing rats. It has been further demonstrated that, by comparison with the normal liver, the liver depleted of glycogen is more susceptible to the development of metases from circulating cancer cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Rights and permissions
About this article
Cite this article
Garvie, W., Grant, R. Liver Metastases—an Experimental Study. Br J Cancer 25, 166–171 (1971). https://doi.org/10.1038/bjc.1971.23
Issue Date:
DOI: https://doi.org/10.1038/bjc.1971.23
This article is cited by
-
Effect of hydrocortisone on the blood sugar and glycogenesis in rats with tumors
Bulletin of Experimental Biology and Medicine (1974)