Abstract
Aim:
To investigate the effects of darapladib, a specific inhibitor of lipoprotein-associated phospholipase A2 (lp-PLA2), on inflammation and atherosclerotic formation in the low density lipoprotein receptor (LDLR)-deficient mice.
Methods:
Six-week-old LDLR-deficient mice were fed an atherogenic high-fat diet for 17 weeks and then randomly divided into two groups. One group was administered darapladib (50 mg·kg−1·d−1; po) for 6 weeks. The other group was administered saline as a control. Serum lipid levels were measured using the corresponding kits, and three inflammatory markers — interleukin-6 (IL-6), C reactive protein (hs-CRP), and platelet activating factor (PAF) — were determined using ELISA. Atherosclerotic plaque areas were stained with Sudan IV, and inflammatory gene expression at the lesions was evaluated using quantitative real-time PCR.
Results:
The body weight and serum lipid level between the two groups were similar at the end of the dietary period. The serum lp-PLA2 activity, hs-CRP and IL-6 levels, however, were significantly reduced in the darpladib group. The inhibition of lp-PLA2 did not alter the serum PAF level. Furthermore, the plaque area, from the aortic arch to the abdominal aorta, was significantly reduced in the darpladib group. Additionally, the expression of inflammatory genes monocyte chemotactic protein-1 (MCP-1) and vascular cell adhesion molecule-1 (VCAM-1) was significantly reduced at the lesions in the darapladib group.
Conclusion:
Inhibition of lp-PLA2 by darapladib decreases the inflammatory burden and atherosclerotic plaque formation in LDLR-deficient mice, which may be a new strategy for the treatment of atherosclerosis.
Similar content being viewed by others
Introduction
Phospholipases A2 (PLA2s) constitute a superfamily of enzymes that catalyze the hydrolysis of fatty acids from the sn-2 position of glycerophospholipids to produce free fatty acids and lysophospholipids, which are both involved in signaling transduction and metabolic processes, and account for a vast number of diseases1, 2. PLA2s, then, are obvious candidates for pharmacological research and intervention. Of all the PLA2s, lp-PLA2 in particular has gained increasing attention as a growing number of epidemiological and experimental studies suggest that it plays an important role in diseases such as atherosclerosis, diabetes, and asthma3, 4, 5, 6.
Lp-PLA2, also referred to as platelet-activating factor acetylhydrolase (PAF-AH; E.C. 3.1.1.47), is a Ca2+-independent, 45 kDa secreted protein that associates with lipoproteins and circulates within the plasma in active form7. Lp-PLA2 can be up-regulated by the oxidized phospholipids in oxLDL8 and in turn acts upon those oxidized phospholipids to produce two pro-inflammatory mediators, lysophosphatidylcholines (lysoPCs) and oxidized nonesterified fatty acids (oxNEFAs)9. Research suggests that the regulatory roles of these two products, especially lysoPCs, are in the promotion of atherosclerotic plaque formation. For instance, lysoPCs have the capacity to recruit leukocytes to lesions, activate leukocytes to initiate immune responses, and promote foam cell formation10, 11, 12.
The role of lp-PLA2 in the processing of oxLDL, inflammation, and atherogenesis suggests that the inhibition of this enzyme could play a positive role in the treatment of cardiovascular events. Indeed, earlier studies have reported that a specific lp-PLA2 inhibitor, darapladib, attenuates the inflammatory burden in patients with stable coronary artery disease and prevents necrotic core expansion, a key determinant of plaque rupture13, 14. In animal assays, the inhibition of lp-PLA2 by darapladib reduced complex coronary atherosclerotic plaque formation in pigs with induced diabetes and hypercholesterolemia15.
However, there has been no in vivo data about the effects of lp-PLA2 inhibitor on the development of atherosclerosis in mouse models. In our study, we evaluated the specific lp-PLA2 inhibitor, darapladib, in the low density lipoprotein receptor (LDLR)-deficient mice to further confirm its role in the development of atherosclerosis.
Materials and methods
Chemicals
Darapladib was synthesized and procured from Prof Jian-hua SHEN's lab at the Shanghai Institute of Materia Medica, Chinese Academy of Sciences, Shanghai 201203, China. TRIzol reagent was purchased from Invitrogen (Carlsbad, CA, USA). PCR-related reagents were purchased from BIO-RAD (Hercules, CA, USA). Other reagents, unless otherwise specified, were obtained from Sigma-Aldrich (St Louis, MO, USA).
Animals
Male homozygous LDLR-deficient mice (C57/Bl6 genetic background) were obtained from the Jackson Laboratory. The animals were cared for in accordance with the institutional guidelines of the Animal Care and Use Committee of the Shanghai Institute of Materia Medica, Chinese Academy of Sciences.
The mice were fed a high-fat diet consisting of 18% hydrogenated cocoa butter, 0.15% cholesterol, 7% casein, 7% sucrose, and 3% maltodextrin for 17 weeks, beginning at 6 weeks of age. Forty mice were divided into two groups (n=20 per group) randomly. One group received darapladib by gavage (50 mg·kg-1·d-1) once daily, while the other group received the vehicle (saline). During the 6 weeks of treatment, all mice were housed in a room with a 12-h light/dark cycle and were allowed free access to a high-fat diet and water.
Serum lipid analysis
Blood samples were obtained from the retro-orbital plexus of the mice prior to drug administration, and 24 h after the last round of drug administration. Serum was obtained through the centrifugation of blood at 1000×g and stored at -80 °C until analysis. Total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C) and triglyceride (TG) levels were measured with an auto-analyzer (Hitachi 7100, Japan) using the corresponding kits from Wako Inc (Enid, OK, USA).
Measurement of serum Lp-PLA2 activity
Serum lp-PLA2 activity was measured using 2-thio-PAF as the substrate. Briefly, 10 μL of plasma was added to 0.1 mol/L Tris-HCl (pH 7.2) containing 1 mmol/L EGTA, 50 μmol/L 2-thio-PAF and 10 μL of 2 mmol/L 5,5′-dithiobis (2-nitrobenzoic acid) in a total volume of 200 μL. The assay was performed using a plate reader to obtain absorbance values at 414 nm every minute. The lp-PLA2 activity was calculated from the change in absorbance per minute.
Measurement of serum interleukin-6 (IL-6), high-sensitivity C-reactive protein (hs-CRP), and platelet activating factor (PAF)
Serum IL-6, hs-CRP, and PAF levels were determined by corresponding ELISA kits (Mingrui Biotech Inc, Shanghai, China).
Morphology of atherosclerotic plaques
At the end of the 6-week treatment period, the mice were anesthetized and killed. Several mice were perfusion-fixed with 4.5% formaldehyde. The aortas were dissected, from the heart to approximately 3 mm distal to the iliac bifurcation. The aortas were then preserved in fresh paraformaldehyde solution for 2 weeks and Sudan IV staining was employed to analyze plaque formation along the entire length of the aortas. Briefly, the aortas were opened longitudinally and pinned upon on a black silica gel plate after removing the surrounding adventitial fatty tissue. The aorta was rinsed in 70% ethanol after 12 h of fixation in the paraformaldehyde solution, stained with 1% Sudan IV in 50% acetone/35% ethanol for approximately 10 min, and washed in 80% ethanol for 5 min. Finally, the stained aortas were photographed and analyzed using the Image Pro-Plus 6.0 software.
Quantitative RT-PCR
After 6 weeks of treatment, the total RNA was extracted from the aortic arch and the thoracic-abdominal aortas of the two groups of mice using TRIzol reagent (Invitrogen Inc, Carlsbad, CA, USA). First-strand cDNAs were synthesized from 4 μg of total RNA using M-MLV reverse transcriptase (Promega Inc). Quantitative real-time PCR was performed using SYBR Green I as the detector dye. Relative gene expression was calculated by normalizing to the amount of mouse actin gene. The primer sequences are shown in Table 1.
Statistical analysis
Data are presented as mean values±SD. For serum lipid and lp-PLA2 activity, comparisons were made using the one-way analysis of variance (ANOVA), followed by the post-hoc Dunnett test for significance. Comparisons of body weight, plaque area and gene expression were analyzed by a two-tailed Student's t-test. For all tests, P<0.05 was considered statistically significant.
Results
Darapladib inhibits serum lp-PLA2 activity in vivo
Our previous unpublished research has shown that mouse lp-PLA2 is less sensitive to darapladib than human lp-PLA2. In this study, then, we tested the dosage range of darapladib to establish the dose at which lp-PLA2 in mouse serum would be significantly inhibited. As shown in Figure 1, the activity of serum lp-PLA2 was inhibited by more than 60% in LDLR-deficient mice after oral administration of 50 mg/kg once daily of darapladib for 6 weeks.
Inhibition of lp-PLA2 by darapladib has no significant effects on serum lipid profile
Lipid level and body weight were evaluated in both groups. As expected, there was no significant difference in TC, TG, LDL-C, and HDL-C levels between the two groups (Table 2). Additionally, there was no significant difference in the body weight of LDLR-deficient mice in either group (Table 2).
Inhibition of lp-PLA2 attenuates the inflammatory burden in serum
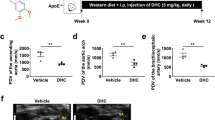
To evaluate the effects of lp-PLA2 inhibition on the inflammatory burden in vivo, we examined two typical inflammatory markers in the serum of LDLR-deficient mice by ELISA — hs-CRP and IL-6. As shown in Figure 2A and 2B, both hs-CRP and IL-6 were significantly reduced in the darapladib group as compared with those in the vehicle group. To determine whether inhibition of lp-PLA2 affects levels of PAF, we also examined the serum PAF. As shown in Figure 2C, plasma PAF levels between the two groups were similar.
Inhibition of lp-PLA2 decreases the formation of atherosclerotic lesions
To determine the effects of the lp-PLA2 inhibitor on the development of atherosclerotic lesions in the aortic vessels of both groups of mice, we analyzed the plaque size at the proximal aorta by Sudan IV staining. As shown in Figure 3, the plaque content in the darapladib group, expressed as the occupancy of aortic luminal surface by atherosclerotic lesions, had significantly decreased compared with the plaque content in the vehicle group.
Inhibition of lp-PLA2 attenuates some inflammatory gene expression at plaque lesions
We additionally examined lp-PLA2, as well as the expression of several inflammatory genes in the aortic vessels, by quantitative real-time PCR. Interestingly, there was no significant difference in the expression of lp-PLA2 between the two groups (Figure 4). However, the levels of expression from the MCP-1 and the VCAM-1 genes were remarkably reduced in the darapladib group as compared to the vehicle group (Figure 4). Surprisingly, the expression of another inflammatory gene that we analyzed, ICAM-1, was comparable in the two groups (Figure 4).
Discussion
Atherosclerosis is the most common cause of many cardiovascular diseases, such as myocardial infarction and stroke16. It is widely recognized that the development of atherosclerosis is associated with both the metabolism of lipids and inflammation17. In our study, we examined the effects of a specific lp-PLA2 inhibitor, darapladib, on inflammation and atherogenesis in well-established LDLR-deficient mice. We found that the inhibition of lp-PLA2 does not change lipid profiles. Instead, the inhibition of lp-PLA2 attenuates the in vivo inflammatory burden and decreases atherosclerotic plaque formation in LDLR-deficient mice.
Specific gene-knockout mouse models are extensively used to study the pathological processes and pharmacological interventions of atherosclerosis, among which the model of the LDLR-deficient mouse is well established18. As the uptake of lipoprotein particles is impaired, the blood lipid profiles of these mice spontaneously rise. The high-fat diet leads to increased cholesterol, as well as atherosclerotic plaque formation19. As predicted, however, the treatment of these mice with the lp-PLA2 inhibitor did not alter their plasma lipoprotein profiles. The beneficial effects of the lp-PLA2 inhibitor, then, are not dependent upon the alteration of lipoprotein levels. The inhibition of lp-PLA2 has shown no marked influence on plasma lipoprotein profiles in pigs with induced diabetes and hypercholesterolemia (DM-HC) or in cardiovascular patients13, 15, 20. These findings are consistent with the results from our study. Interestingly, the elevation of in vivo lp-PLA2 activity by adenovirus-mediated gene transfer also does not alter the plasma lipoprotein profile21.
Many epidemiological studies have suggested that lp-PLA2 is a marker for cardiovascular risk, and that the plasma levels and activity of lp-PLA2 increase during the development of atherosclerosis22, 23, 24, 25, 26, 27, 28, 29, 30. In our study, we also found that serum lp-PLA2 activity increased in the vehicle group after the mice were fed a high-fat diet for 6 weeks; such activity was significantly inhibited in the darapladib group.
Lp-PLA2 is thought to play an important regulatory role in the development of atherosclerosis due to the role of its enzyme activity in hydrolyzing bioactive lipids, such as PAF and oxidized phosphocholines (oxPCs). However, what researchers believe concerning the precise role of lp-PLA2 remains controversial. Lp-PLA2 may hydrolyze and inactivate PAF, a well-known and typical pro-inflammatory factor that contributes to tissue damage and thrombosis formation31. On the other hand, lp-PLA2 may generate pro-inflammatory lipid mediators, such as oxNEFAs and lysoPCs, by hydrolyzing oxidized phospholipids in oxLDL. However, there is no evidence that lp-PLA2 hydrolyzes PAF in vivo. In our study, the inhibition of lp-PLA2 by darapladib did not change serum PAF levels. In addition, intravenous administration of recombinant lp-PLA2 failed to alter the PAF-mediated responses in patients with asthma or septic shock32, 33. Furthermore, a recent report suggested that circulating PAF is primarily cleared by transport and not by the hydrolysis of lp-PLA2 in vivo34.
Conversely, there has been much in vitro or in vivo research that supports the concept of the pro-atherogenic roles of lp-PLA2. By hydrolyzing the oxidized phospholipids in oxLDL, lp-PLA2 produces two kinds of inflammatory mediators, lysoPCs and oxNEFA. These mediators initiate a high level of inflammatory response, such as cell adhesion, inflammatory gene expression, and cell death10, 35. In addition, in vivo studies have suggested that the inhibition of lp-PLA2 by darapladib decreases the inflammatory burden in humans and pigs13, 14, 15. In our study, we also examined the inflammatory factors in the serum and in the gene expression of some inflammatory cytokines in atherosclerotic lesions. Consistent with other in vivo studies, our research shows that the inflammatory burden decreased in the darapladib group compared with that in the vehicle group. Interestingly, previous research, along with our study, has suggested that the inhibition of lp-PLA2 did not affect lp-PLA2 expression. Theoretically, the inhibition of lp-PLA2 may increase lp-PLA2 expression at the vessels as with the oxPCs, which can up-regulate lp-PLA2 expression8. Thus, the expression of lp-PLA2 at atherosclerotic plaques needs further study. MCP-1, VCAM-1, and ICAM-1 are typical inflammatory cytokines mediating cell adhesion, a crucial step for monocyte migration into lesions7. LysoPCs, the product of lp-PAL2, can up-regulate MCP-1 and ICAM-1/VCAM-1 expression in the endothelial cells or vascular smooth muscle cells (VSMCs)36. Surprisingly, in our study, we detected the reduction of MCP-1 and VCAM-1, but not ICAM-1, expression. Regardless, our research, and the previous research of others, supports the pro-atherogenic roles of lp-PLA2 in vivo.
Indeed, the lp-PLA2 inhibitor has been evaluated in induced diabetes and hypercholesterolemia (DM-HC) in pigs15. Treatment with darapladib resulted in a considerable decrease in plaque area and a markedly reduced necrotic core area. Clinical research has also shown that the inhibition of lp-PLA2 with darapladib has arrested the expansion of the necrotic core, a key determinant of plaque vulnerability, despite not preventing plaque formation13.
In summary, these in vivo studies demonstrate that the inhibition of lp-PLA2 by darapladib does not ameliorate dyslipidemia; instead, darapladib attenuates the inflammatory burden, resulting in the prevention of atherosclerosis in LDLR-deficient mice on a high-fat diet. Our study once again highlights that anti-inflammation therapy is a feasible strategy for the treatment of cardiovascular disease and lp-PLA2 is a promising target against atherosclerosis.
Author contribution
Yi-ping WANG and Wen-yi WANG designed the research project; Yi-ping WANG supervised the project; Jie ZHANG, Miao-miao HU performed the animal assays; Wen-yu WU, Yan-ling MA, and Wei-hai CHEN performed the in vitro assays; Jie ZHANG, Miao-miao HU, and Wen-yi WANG analyzed the data and wrote the manuscript; Jie ZHANG, Miao-miao HU, and Yi-ping WANG revised the manuscript.
References
Burke JE, Dennis EA . Phospholipase A2 biochemistry. Cardiovasc Drugs Ther 2009; 23: 49–59.
Prescott SM, Zimmerman GA, Stafforini DM, McIntyre TM . Platelet-activating factor and related lipid mediators. Annu Rev Biochem 2000; 69: 419–45.
Stafforini DM . Biology of platelet activating factor acetyhydrolase (PAF-AH, lipoprotein associated phospholipase A2). Cardiovasc Drugs Ther 2009; 23: 73–83.
Kasperska-Zajac A, Brzoza Z, Rogala B . Platelet-activating factor (PAF): a review of its role in asthma and clinical efficacy of PAF antagonists in the disease therapy. Recent Pat Inflamm Allergy Drug Discov 2008; 2: 72–6.
Miller RG, Costacou T, Orchard TJ . Lipoprotein-associated phospholipase A2, C-reactive protein, and coronary artery disease in individuals with type 1 diabetes and macroalbuminuria. Diab Vasc Dis Res 2010; 7: 47–55.
Wegner M, Araszkiewicz A, Piorunska-Mikolajczak A, Zozulinska-Ziolkiewicz D, Wierusz-Wysocka B, Piorunska-Stolzmann M . The evaluation of IL-12 concentration, PAF-AH, and PLA(2) activity in patients with type 1 diabetes treated with intensive insulin therapy. Clin Biochem 2009; 42: 1621–7.
Zalewski A, Macphee C . Role of lipoprotein-associated phospholipase A2 in atherosclerosis: biology, epidemiology, and possible therapeutic target. Arterioscler Thromb Vasc Biol 2005; 25: 923–31.
Wang WY, Li J, Yang D, Xu W, Zha RP, Wang YP . OxLDL stimulates lipoprotein-associated phospholipase A2 expression in THP-1 monocytes via PI3K and p38 MAPK pathways. Cardiovasc Res 2010; 85: 845–52.
MacPhee CH, Moores KE, Boyd HF, Dhanak D, Ife RJ, Leach CA, et al. Lipoprotein-associated phospholipase A2, platelet-activating factor acetylhydrolase, generates two bioactive products during the oxidation of low-density lipoprotein: use of a novel inhibitor. Biochem J 1999; 338: 479–87.
Leitinger N . Oxidized phospholipids as modulators of inflammation in atherosclerosis. Curr Opin Lipidol 2003; 14: 421–30.
Tselepis AD, John Chapman M . Inflammation, bioactive lipids and atherosclerosis: potential roles of a lipoprotein-associated phospholipase A2, platelet activating factor-acetylhydrolase. Atheroscler Suppl 2002; 3: 57–68.
Rong JX, Berman JW, Taubman MB, Fisher EA . Lysophosphatidylcholine stimulates monocyte chemoattractant protein-1 gene expression in rat aortic smooth muscle cells. Arterioscler Thromb Vasc Biol 2002; 22: 1617–23.
Serruys PW, Garcia-Garcia HM, Buszman P, Erne P, Verheye S, Aschermann M, et al. Effects of the direct lipoprotein-associated phospholipase A(2) inhibitor darapladib on human coronary atherosclerotic plaque. Circulation 2008; 118: 1172–82.
Mohler ER 3rd, Ballantyne CM, Davidson MH, Hanefeld M, Ruilope LM, Johnson JL, et al. The effect of darapladib on plasma lipoprotein-associated phospholipase A2 activity and cardiovascular biomarkers in patients with stable coronary heart disease or coronary heart disease risk equivalent: the results of a multicenter, randomized, double-blind, placebo-controlled study. J Am Coll Cardiol 2008; 51: 1632–41.
Wilensky RL, Shi Y, Mohler ER 3rd, Hamamdzic D, Burgert ME, Li J, et al. Inhibition of lipoprotein-associated phospholipase A2 reduces complex coronary atherosclerotic plaque development. Nat Med 2008; 14: 1059–66.
Pinon P, Kaski JC . Inflammation, atherosclerosis and cardiovascular disease risk: PAPP-A, Lp-PLA2, and cystatin C. New insights or redundant information? Rev Esp Cardiol 2006; 59: 247–58.
Steinberg D . Atherogenesis in perspective: hypercholesterolemia and inflammation as partners in crime. Nat Med 2002; 8: 1211–7.
Zadelaar S, Kleemann R, Verschuren L, de Vries-Van der Weij J, van der Hoorn J, Princen HM, et al. Mouse models for atherosclerosis and pharmaceutical modifiers. Arterioscler Thromb Vasc Biol 2007; 27: 1706–21.
Ishibashi S, Brown MS, Goldstein JL, Gerard RD, Hammer RE, Herz J . Hypercholesterolemia in low density lipoprotein receptor knockout mice and its reversal by adenovirus-mediated gene delivery. J Clin Invest 1993; 92: 883–93.
Rosenson RS . Fenofibrate reduces lipoprotein associated phospholipase A2 mass and oxidative lipids in hypertriglyceridemic subjects with the metabolic syndrome. Am Heart J 2008; 155: 499.e9–16.
Quarck R, De Geest B, Stengel D, Mertens A, Lox M, Theilmeier G, et al. Adenovirus-mediated gene transfer of human platelet-activating factor-acetylhydrolase prevents injury-induced neointima formation and reduces spontaneous atherosclerosis in apolipoprotein E-deficient mice. Circulation 2001; 103: 2495–500.
Packard CJ, O'Reilly DS, Caslake MJ, McMahon AD, Ford I, Cooney J, et al. Lipoprotein-associated phospholipase A2 as an independent predictor of coronary heart disease. West of Scotland Coronary Prevention Study Group. N Engl J Med 2000; 343: 1148–55.
Koenig W, Khuseyinova N, Lowel H, Trischler G, Meisinger C . Lipoprotein-associated phospholipase A2 adds to risk prediction of incident coronary events by C-reactive protein in apparently healthy middle-aged men from the general population: results from the 14-year follow-up of a large cohort from southern Germany. Circulation 2004; 110: 1903–8.
Ballantyne CM, Hoogeveen RC, Bang H, Coresh J, Folsom AR, Heiss G, et al. Lipoprotein-associated phospholipase A2, high-sensitivity C-reactive protein, and risk for incident coronary heart disease in middle-aged men and women in the Atherosclerosis Risk in Communities (ARIC) study. Circulation 2004; 109: 837–42.
Persson M, Hedblad B, Nelson JJ, Berglund G . Elevated Lp-PLA2 levels add prognostic information to the metabolic syndrome on incidence of cardiovascular events among middle-aged nondiabetic subjects. Arterioscler Thromb Vasc Biol 2007; 27: 1411–6.
May HT, Horne BD, Anderson JL, Wolfert RL, Muhlestein JB, Renlund DG, et al. Lipoprotein-associated phospholipase A2 independently predicts the angiographic diagnosis of coronary artery disease and coronary death. Am Heart J 2006; 152: 997–1003.
Koenig W, Twardella D, Brenner H, Rothenbacher D . Lipoprotein-associated phospholipase A2 predicts future cardiovascular events in patients with coronary heart disease independently of traditional risk factors, markers of inflammation, renal function, and hemodynamic stress. Arterioscler Thromb Vasc Biol 2006; 26: 1586–93.
Gerber Y, McConnell JP, Jaffe AS, Weston SA, Killian JM, Roger VL . Lipoprotein-associated phospholipase A2 and prognosis after myocardial infarction in the community. Arterioscler Thromb Vasc Biol 2006; 26: 2517–22.
Caslake MJ, Packard CJ . Lipoprotein-associated phospholipase A2 as a biomarker for coronary disease and stroke. Nat Clin Pract Cardiovasc Med 2005; 2: 529–35.
Brilakis ES, McConnell JP, Lennon RJ, Elesber AA, Meyer JG, Berger PB . Association of lipoprotein-associated phospholipase A2 levels with coronary artery disease risk factors, angiographic coronary artery disease, and major adverse events at follow-up. Eur Heart J 2005; 26: 137–44.
Zalewski A, Macphee C, Nelson JJ . Lipoprotein-associated phospholipase A2: a potential therapeutic target for atherosclerosis. Curr Drug Targets Cardiovasc Haematol Disord 2005; 5: 527–32.
Henig NR, Aitken ML, Liu MC, Yu AS, Henderson WR Jr . Effect of recombinant human platelet-activating factor-acetylhydrolase on allergen-induced asthmatic responses. Am J Respir Crit Care Med 2000; 162: 523–7.
Opal S, Laterre PF, Abraham E, Francois B, Wittebole X, Lowry S, et al. Recombinant human platelet-activating factor acetylhydrolase for treatment of severe sepsis: results of a phase III, multicenter, randomized, double-blind, placebo-controlled, clinical trial. Crit Care Med 2004; 32: 332–41.
Liu J, Chen R, Marathe GK, Febbraio M, Zou W, McIntyre TM . Circulating platelet-activating factor is primarily cleared by transport, not intravascular hydrolysis by lipoprotein-associated phospholipase A2/ PAF acetylhydrolase. Circ Res 2011; 108: 469–77.
Nonas S, Birukova AA, Fu P, Xing J, Chatchavalvanich S, Bochkov VN, et al. Oxidized phospholipids reduce ventilator-induced vascular leak and inflammation in vivo. Crit Care 2008; 12: R27.
Kume N, Cybulsky MI, Gimbrone MA Jr . Lysophosphatidylcholine, a component of atherogenic lipoproteins, induces mononuclear leukocyte adhesion molecules in cultured human and rabbit arterial endothelial cells. J Clin Invest 1992; 90: 1138–44.
Acknowledgements
This study was financially supported by a grant from the Shanghai Committee of Science and Technology, China (No 11ZR1444800), and the National Basic Research Program of China (No 2009CB930300).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hu, Mm., Zhang, J., Wang, Wy. et al. The inhibition of lipoprotein-associated phospholipase A2 exerts beneficial effects against atherosclerosis in LDLR-deficient mice. Acta Pharmacol Sin 32, 1253–1258 (2011). https://doi.org/10.1038/aps.2011.127
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/aps.2011.127
Keywords
This article is cited by
-
Research Advance of Chinese Medicine in Treating Atherosclerosis: Focus on Lipoprotein-Associated Phospholipase A2
Chinese Journal of Integrative Medicine (2024)
-
Lp-PLA2 inhibition prevents Ang II-induced cardiac inflammation and fibrosis by blocking macrophage NLRP3 inflammasome activation
Acta Pharmacologica Sinica (2021)
-
Thyroxine therapy ameliorates serum levels of eicosanoids in Chinese subclinical hypothyroidism patients
Acta Pharmacologica Sinica (2016)
-
Increased serum level of Lp-PLA2 is independently associated with the severity of coronary artery diseases: a cross-sectional study of Chinese population
BMC Cardiovascular Disorders (2015)
-
Predictors of change in carotid atherosclerotic plaque inflammation and burden as measured by 18-FDG-PET and MRI, respectively, in the dal-PLAQUE study
The International Journal of Cardiovascular Imaging (2014)