Abstract
Aim:
This study was designed to examine the effect of scutellarein on high glucose- and hypoxia-stimulated proliferation of human retinal endothelial cells (HREC).
Methods:
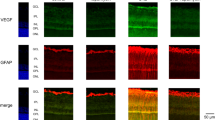
HREC were cultured under normal glucose (NG), moderate, and high glucose (NG supplemented with 10 or 25 mmol/L D-glucose) and/or hypoxic (cobalt chloride treated) conditions. Cell proliferation was evaluated by a cell counting kit. The expression of vascular endothelial growth factor (VEGF) was assessed by Western blot analysis.
Results:
The proliferation of HREC was significantly elevated in response to moderately-high glucose and hypoxic conditions. The combination of high glucose and hypoxia did not have any additive effects on cell proliferation. Consistent with the proliferation data, the expression of VEGF was also upregulated under both moderately-high glucose and hypoxic conditions. The treatment with scutellarein (1×10−11–1×10−5 mol/L) significantly inhibited high glucose- or hypoxia-induced cell proliferation and VEGF expression.
Conclusion:
Both hypoxia and moderately-high glucose were potent stimuli for cell proliferation and VEGF expression in HREC without any significant additive effects. Scutellarein is capable of inhibiting the proliferation of HREC, which is possibly related to its ability to suppress the VEGF expression.
Similar content being viewed by others
Article PDF
References
The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993; 329: 977–86.
Lu M, Kuroki M, Amano S, Tolentino M, Keough K, Kim I, et al. Advanced glycation end products increase retinal vascular endothelial growth factor expression. J Clin Invest 1998; 101: 1219–24.
Kern TS, Engerman RL . Capillary lesions develop in retina rather than cerebral cortex in diabetes and experimental galactosemia. Arch Ophthalmol 1996; 114: 306–10.
Agardh CD, Agardh E, Zhang H, Ostenson CG . Altered endothelial/pericyte ratio in Goto-Kakizaki rat retina. J Diabetes Complications 1997; 11: 158–62.
Bursell SE, Takagi C, Clermont AC, Tagaki H, Mori F, Ishii H, et al. Specific retinal diacylglycerol and protein kinase C beta isoform modulation mimics abnormal retinal hemodynamics in diabetic rats. Invest Ophthalmol Vis Sci 1997; 38: 2711–20.
Duh E, Aiello LP . Vascular endothelial growth factor and diabetes: the agonist versus antagonist paradox. Diabetes 1999; 48: 1899–906.
Miller JW, Adamis AP, Aiello LP . Vascular endothelial growth factor in ocular neovascularization and proliferative diabetic retinopathy. Diabetes Metab Rev 1997; 13: 37–50.
Hammes HP, Lin J, Bretzel RG, Brownlee M, Breier G . Upregulation of the vascular endothelial growth factor/vascular endothelial growth factor receptor system in experimental background diabetic retinopathy of the rat. Diabetes 1998; 47: 401–6.
Gilbert RE, Vranes D, Berka JL, Kelly DJ, Cox A, Wu LL, et al. Vascular endothelial growth factor and its receptors in control and diabetic rat eyes. Lab Invest 1998; 78: 1017–27.
Enaida H, Kabuyama Y, Oshima Y, Sakamoto T, Kato K, Kochi H, et al. VEGF-dependent signaling in retinal microvascular endothelial cells Fukushima. J Med Sci 1999; 45: 77–91.
Hata Y, Rook SL, Aiello LP . Basic fibroblast growth factor induces expression of VEGF receptor KDR through a protein kinase C and p44/p42 mitogen-activated protein kinase-dependent pathway. Diabetes 1999; 48: 1145–55.
Aiello LP, Northrup JM, Keyt BA, Takagi H, Iwamoto MA . Hypoxic regulation of vascular endothelial growth factor in retinal cells. Arch Ophthalmol 1995; 113: 1538–44.
Suzuma K, Naruse K, Suzuma I, Takahara N, Ueki K, Aiello LP, et al. Vascular endothelial growth factor induces expression of connective tissue growth factor via KDR, Flt1, and phosphatidylinositol 3-kinase-akt-dependent pathways in retinal vascular cells. J Biol Chem 2000; 275: 40725–31.
Chen XQ, Jin YY . The new edition of medicaments. 14th ed. Beijing: People's Medical Publishing House; 1997.
Wang GX . Pharmacologic action of breviscapine and clinic application. Lishizhen. Med Mater Med Res 1999; 10: 639–40.
Zhu BH, Guan YY, He H, Lin MJ . Erigeron breviscapus prevents defective endothelium-dependent relaxation in diabetic rat aorta. Life Sci 1999; 65: 1553–9.
Williams B, Gallacher B, Patel H, Orme C . Glucose-induced protein kinase C activation regulates vascular permeability factor mRNA expression and peptide production by human vascular smooth muscle cells in vitro. Diabetes 1997; 46: 1497–503.
Xia P, Aiello LP, Ishii H, Jiang ZY, Park DJ, Robinson GS, et al. Characterization of vascular endothelial growth factor's effect on the activation of protein kinase C, its isoforms, and endothelial cell growth. J Clin Invest 1996; 98: 2018–26.
Li B, Tang SB, Zhang G, Chen JH, Li BJ . Culture and characterization of human retinal capillary endothelial cell. Chin Ophthal Res 2005; 23: 20–2.
Premanand C, Rema M, Sameer MZ, Sujatha M, Balasubramanyam M . Effect of curcumin on proliferation of human retinal endothelial cells under in vitro conditions. Invest Ophthalmol Vis Sci 2006; 47: 2179–84.
Berra E, Pages G, Pouyssegur J . MAP kinases and hypoxia in the control of VEGF expression. Cancer Metastasis Rev 2000; 19: 139–45.
de Vries C, Escobedo JA, Ueno H, Houck K, Ferrara N, Williams LT . The fms-like tyrosine kinase, a receptor for vascular endothelial growth factor. Science 1992; 255: 989–91.
Ishii H, Koya D, King GL . Protein kinase C activation and its role in the development of vascular complications in diabetes mellitus. J Mol Med 1998; 76: 21–31.
Lee JW, Park JA, Kim SH, Seo JH, Lim KJ, Jeong JW, et al. Protein kinase C-δ regulates the stability of hypoxia-inducible factor-1α under hypoxia. Cancer Sci 2007; 98: 1476–81.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by a joint project of the National Education Ministry and Guangdong Province (No 2007B090400089).
Rights and permissions
About this article
Cite this article
Gao, R., Zhu, Bh., Tang, Sb. et al. Scutellarein inhibits hypoxia- and moderately-high glucose-induced proliferation and VEGF expression in human retinal endothelial cells. Acta Pharmacol Sin 29, 707–712 (2008). https://doi.org/10.1111/j.1745-7254.2008.00797.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2008.00797.x
Keywords
This article is cited by
-
The potential effects and mechanisms of hispidulin in the treatment of diabetic retinopathy based on network pharmacology
BMC Complementary Medicine and Therapies (2022)
-
Scutellarein protects against cardiac hypertrophy via suppressing TRAF2/NF-κB signaling pathway
Molecular Biology Reports (2022)
-
Preparation of nanoparticles of β-cyclodextrin-loaded scutellarein anti-tumor activity research by targeting integrin αvβ3
Cancer Nanotechnology (2021)
-
TPTEP1 suppresses high glucose-induced dysfunction in retinal vascular endothelial cells by interacting with STAT3 and targeting VEGFA
Acta Diabetologica (2021)
-
Inhibition of hsa_circ_0002570 suppresses high-glucose–induced angiogenesis and inflammation in retinal microvascular endothelial cells through miR-1243/angiomotin axis
Cell Stress and Chaperones (2020)