Abstract
Aim:
Adenosine monophosphate-activated protein kinase (AMPK), a vital regulator of glucose metabolism, may affect insulin secretion in β-cells. However, the role of AMPK in β-cell lipotoxicity remains unclear. Fenofibrate has been reported to regulate lipid homeostasis and is involved in insulin secretion in pancreatic β-cells. In the present study, we aimed to investigate the effect of palmitate on AMPK expression and glucose-stimulated insulin secretion (GSIS) in rat islets and INS-1 β-cell, as well as the effect of fenofibrate on AMPK and GSIS in INS-1 cells treated with palmitate.
Methods:
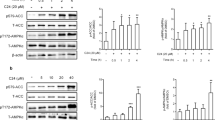
Isolated rat islets and INS-1 β-cells were treated with and without palmitate or fenofibrate for 48 h. The mRNA levels of the AMPKα isoforms were measured by real-time PCR. Western blotting was used to detect the protein expression of total AMPKα (T-AMPKα), phosphorylated AMPKα (P-AMPKα), and phosphorylated acetyl coenzyme A carboxylase (P-ACC). Insulin secretion was detected by radioimmunoassay induced by 20 mmol/L glucose as GSIS.
Results:
The results showed that chronic exposure of β-cells to palmitate for 48 h inhibited the expression of AMPKα1 mRNA and T-AMPKα protein levels, as well as P-AMPKα and P-ACC protein expressions in a dose-dependent manner. Accordingly, GSIS was inhibited by palmitate. Compared with the palmitate-treated cells, fenofibrate ameliorated these changes impaired by palmitate and exhibited a significant elevation in the expression of AMPKα and GSIS.
Conclusion:
Our findings suggest a role of AMPKα reduction in β-cell lipotoxicity and a novel role of fenofibrate in improving GSIS associated with the AMPKα activation in β-cells chronically exposed to palmitate.
Similar content being viewed by others
Article PDF
References
Bergeron R, Previs SF, Cline GW, Perret P, Russell RR, Young LH, et al. Effect of 5-aminoimidazole-4-carboxamide-1-beta-D-ribofuranoside infusion on in vivo glucose and lipid metabolism in lean and obese Zucker rats. Diabetes 2001; 50: 1076–82.
Fisher JS, Gao J, Han DH, Holloszy JO, Nolte LA . Activation of AMP kinase enhances sensitivity of muscle glucose transport to insulin. Am J Physiol Endocrinol Metab 2002; 282: E18–23.
Iglesias MA, Ye JM, Frangioudakis G, Saha AK, Tomas E, Ruderman NB, et al. AICAR administration causes an apparent enhancement of muscle and liver insulin action in insulin-resistant high-fat-fed rats. Diabetes 2002; 51: 2886–94.
Ruderman N, Prentki M . AMP kinase and malonyl-CoA: targets for therapy of the metabolic syndrome. Nat Rev Drug Discov 2004; 3: 340–51.
Fryer LG, Parbu-Patel A, Carling D . The Anti-diabetic drugs rosiglitazone and metformin stimulate AMP-activated protein kinase through distinct signaling pathways. J Biol Chem 2002; 277: 25 226–32.
Hayashi T, Hirshman MF, Kurth EJ, Winder WW, Goodyear LJ . Evidence for 5′ AMP-activated protein kinase mediation of the effect of muscle contraction on glucose transport. Diabetes 1998; 47: 1369–73.
Merrill GF, Kurth EJ, Hardie DG, Winder WW . AICA riboside increases AMP-activated protein kinase, fatty acid oxidation, and glucose uptake in rat muscle. Am J Physiol 1997; 273: E1107–12.
Bergeron R, Russell RR, Young LH, Ren JM, Marcucci M, Lee A, et al. Effect of AMPK activation on muscle glucose metabolism in conscious rats. Am J Physiol 1999; 276: E938–44.
Zhou G, Myers R, Li Y, Chen Y, Shen X, Fenyk-Melody J, et al. Role of AMP-activated protein kinase in mechanism of metformin action. J Clin Invest 2001; 108: 1167–74.
Wollen N, Bailey CJ . Inhibition of hepatic gluconeogenesis by metformin. Synergism with insulin. Biochem Pharmacol 1988; 37: 4353–8.
Salt I, Celler JW, Hawley SA, Prescott A, Woods A, Carling D, et al. AMP-activated protein kinase: greater AMP dependence, and preferential nuclear localization, of complexes containing the α2 isoform. Biochem J 1998; 334 (Pt 1): 177–87.
Akkan AG, Malaisse WJ . Insulinotropic action of AICA riboside. I. Insulin release by isolated islets and the perfused pancreas. Diabetes Res 1994; 25: 13–23.
Salt IP, Johnson G, Ashcroft SJ, Hardie DG . AMP-activated protein kinase is activated by low glucose in cell lines derived from pancreatic β cells, and may regulate insulin release. Biochem J 1998; 335 (Pt 3): 533–9.
Aoyama T, Peters JM, Iritani N, Nakajima T, Furihata K, Hashimoto T, et al. Altered constitutive expression of fatty acid-metabolizing enzymes in mice lacking the peroxisome proliferator-activated receptor α (PPARα). J Biol Chem 1998; 273: 5678–84.
Djouadi F, Weinheimer CJ, Saffitz JE, Pitchford C, Bastin J, Gonzalez FJ, et al. A gender-related defect in lipid metabolism and glucose homeostasis in peroxisome proliferator-activated receptor α-deficient mice. J Clin Invest 1998; 102: 1083–91.
Staels B, Dallongeville J, Auwerx J, Schoonjans K, Leitersdorf E, Fruchart JC . Mechanism of action of fibrates on lipid and lipoprotein metabolism. Circulation 1998; 98: 2088–93.
Koh EH, Kim MS, Park JY, Kim HS, Youn JY, Park HS, et al. Peroxisome proliferator-activated receptor (PPAR)-α activation prevents diabetes in OLETF rats: comparison with PPAR-α activation. Diabetes 2003; 52: 2331–7.
Lalloyer F, Vandewalle B, Percevault F, Torpier G, Kerr-Conte J, Oosterveer M, et al. Peroxisome proliferator-activated receptor-α improves pancreatic adaptation to insulin resistance in obese mice and reduces lipotoxicity in human islets. Diabetes 2006; 55: 1605–13.
Ravnskjaer K, Boergesen M, Rubi B, Larsen JK, Nielsen T, Fridriksson J, et al. Peroxisome proliferator-activated receptor α (PPARα) potentiates, whereas PPARα attenuates, glucose-stimulated insulin secretion in pancreatic β-cells. Endocrinology 2005; 146: 3266–76.
Zhou YT, Shimabukuro M, Wang MY, Lee Y, Higa M, Milburn JL, et al. Role of peroxisome proliferator-activated receptor-α in disease of pancreatic β cells. Proc Natl Acad Sci USA 1998; 95: 8898–903.
Tordjman K, Standley KN, Bernal-Mizrachi C, Leone TC, Coleman T, Kelly DP, et al. PPARα suppresses insulin secretion and induces UCP2 in insulinoma cells. J Lipid Res 2002; 43: 936–43.
Holness MJ, Smith ND, Greenwood GK, Sugden MC . Acute (24 h) activation of peroxisome proliferator-activated receptor-α (PPARα) reverses high-fat feeding-induced insulin hypersecretion in vivo and in perfused pancreatic islets. J Endocrinol 2003; 177: 197–205.
Gotoh M, Maki T, Satomi S, Porter J, Bonner-Weir S, O'Hara CJ, et al. Reproducible high yield of rat islets by stationary in vitro digestion following pancreatic ductal or portal venous collagenase injection. Transplantation 1987; 43: 725–30.
Busch AK, Cordery D, Denyer GS, Biden TJ . Expression profiling of palmitate- and oleate-regulated genes provides novel insights into the effects of chronic lipid exposure on pancreatic β-cell function. Diabetes 2002; 51: 977–87.
Assimacopoulos-Jeannet F, Thumelin S, Roche E, Esser V, McGarry JD, Prentki M . Fatty acids rapidly induce the carnitine palmitoyltransferase I gene in the pancreatic β-cell line INS-1. J Biol Chem 1997; 272: 1659–64.
da Silva Xavier G, Leclerc I, Varadi A, Tsuboi T, Moule SK, Rutter GA . Role for AMP-activated protein kinase in glucose-stimulated insulin secretion and preproinsulin gene expression. Biochem J 2003; 371: 761–74.
Liu Y, Wan Q, Guan Q, Gao L, Zhao J . High-fat diet feeding impairs both the expression and activity of AMPKα in rats' skeletal muscle. Biochem Biophys Res Commun 2006; 339: 701–7.
Wang X, Zhou L, Li G, Luo T, Gu Y, Qian L, et al. Palmitate activates AMP-activated protein kinase and regulates insulin secretion from β cells. Biochem Biophys Res Commun 2007; 352: 463–8.
Dubois M, Kerr-Conte J, Gmyr V, Bouckenooghe T, Muharram G, D'Herbomez M, et al. Non-esterified fatty acids are deleterious for human pancreatic islet function at physiological glucose concentration. Diabetologia 2004; 47: 463–9.
Yaney GC, Corkey BE . Fatty acid metabolism and insulin secretion in pancreatic β cells. Diabetologia 2003; 46: 1297–312.
Liang Z, Yan L, Li D, Li F, Qi Y, Cai M, et al. Impact of fenofibrate on insulin resistance and insulin secretion in individuals with hypertriglyceridemia. Chin J Endocrinol Metab 2007; 23: 8–11.
Murakami H, Murakami R, Kambe F, Cao X, Takahashi R, Asai T, et al. Fenofibrate activates AMPK and increases eNOS phosphorylation in HUVEC. Biochem Biophys Res Commun 2006; 341: 973–8.
Chen M . Lipotoxicity and AMPKα key target for its prevention and treatment. Chin J Endocrinol Metab 2007; 23: 5–7.
Diraison F, Parton L, Ferre P, Foufelle F, Briscoe CP, Lexlerc I, et al. Over-expression of sterol-regulatory-element-binding protein-1c (SREBP1c) in rat pancreatic islets induces lipogenesis and decreases glucose-stimulated insulin release: modulation by 5-aminoimidazole-4-carboxamide ribonucleoside (AICAR). Biochem J 2004; 378: 769–78.
Zhang S, Kim KH . Glucose activation of acetyl-CoA carboxylase in association with insulin secretion in a pancreatic β-cell line. J Endocrinol 1995; 147: 33–41.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by a grant from National Natural Science Foundation of China (No 30670994).
Rights and permissions
About this article
Cite this article
Sun, Y., Ren, M., Gao, Gq. et al. Chronic palmitate exposure inhibits AMPKα and decreases glucose-stimulated insulin secretion from β-cells: modulation by fenofibrate. Acta Pharmacol Sin 29, 443–450 (2008). https://doi.org/10.1111/j.1745-7254.2008.00717.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2008.00717.x