Abstract
Aim:
To study the mechanism by which nanoparticle realgar powders (NRP) induce human histocytic lymphoma U937 cell apoptosis.
Methods:
After the U937 cells were treated with various doses of NRP, the viability of the NRP-induced U937 cells was detected by 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphen-yltetrazolium bromide (MTT) assay. Granular apoptotic bodies with membrane blebbing and condensed nuclei were observed by fluorescence microscopy. The apoptotic ratio induced by NRP was measured by lactate dehydrogenase (LDH) activity-based assay. Caspase-3 and the expressions of Akt, p-Akt, a nicotin-amide adenine dinucleotide (NAD+)-dependent histone deacetylase (SIRT1), p53, and p-p53 were detected by Western blot analysis.
Results:
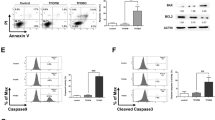
The growth-inhibitory activity of NRP for U937 cells was in a time-and dose-dependent manner. After treatment with various concentrations of NRP for 24 h, the majority of U937 cells underwent apoptosis as measured by LDH assay. In the presence of NRP, wortmannin, the inhibitor of phosphoinositide 3-kinase (PI3-K), and Akt inhibitor KP372-1 augmented the NRP-induced cell apoptosis. When the U937 cells were treated with NRP for the indicated time periods, procaspase-3 was gradually degraded and the activated caspase-3 was significantly increased. The expressions of anti-apoptotic proteins Akt and p-Akt were downregulated. Importantly, the inhibition of SIRT1 contributed to the activation of p53 and the inactivation of the PI3-K/Akt signaling pathway increased the expression of the p53 protein and downregulated the SIRT1 protein expression.
Conclusion:
The PI3-K/Akt signaling pathway plays an important role in NRP-induced U937 cell apoptosis. The reduced SIRT1 expression and activated p53 might be partially due to the inhibition of the PI3-K/Akt pathway triggered by the NRP-induced initiation of U937 cell apoptosis.
Similar content being viewed by others
Article PDF
References
Luo LY, Huang J, Gou BD, Zhang TL, Wang K . Induction of human promyelocytic leukemia HL-60 cell differentiation into monocytes by arsenic sulphide: Involvement of serine/threonine protein phosphatases. Leuk Res 2006; 30: 1399–405.
Wu JZ, Ho PC . Evaluation of the in vitro activity and in vivo bio-availability of realgar nanoparticles prepared by cryo-grinding. Eur J Pharm Sci 2006; 29: 35–44.
Ning N, Peng ZF, Yuan L, Gou BD, Zhang TL, Wang K . Realgar nano-particles induce apoptosis and necrosis in leukemia cell lines K562 and HL-60. China J Chin Mat Med 2005; 30: 136–40. Chinese.
Kim SO, Han J . Pan-caspase inhibitor zVAD enhances cell death in RAW 246.7 macrophages. J Endotoxin Res 2001; 7: 292–6.
Kawazoe N, Watabe M, Masuda Y, Nakajo S, Nakaya K . Tiamil is involved in the regulation of bufalin-induced apoptosis in human leukemia cells. Oncogene 1999; 18: 2413–21.
Hill PA, Tumber A, Meikle MC . Multiple extracellular signals promote osteoblast survival and apoptosis. Endocrinology 1997; 138: 3849–58.
Mizukami S, Kikuchi K, Higuchi T, Urano Y, Mashima T, Tsuruo T, et al. Imaging of caspase-3 activation in HeLa cells stimulation with etoposide using a novel fluorescent probe. FEBS Lett 1999; 453: 356–60.
Wu Z, Wu LJ, Li LH, Tashiro SI, Onodera S, Ikejima T . p53-mediated cell cycle arrest and apoptosis induced by shikonin via a caspase-9-dependent mechanism in human malignant melanoma A375-S2 cell death. J Pharmacol Sci 2004; 94: 166–76.
Qiao AM, Ikejima T, Tashiro S, Onodera S, Zhang WG, Wu YL . Involvement of mitochondria and caspase pathways in N-demethylclarithromycin-induced apoptosis in human cervical cancer HeLa cell. Acta Pharmacol Sin 2006; 27: 1622–9.
Crow MT . Revisiting p53 and its effectors in ischemic heart injury. Cardiovasc Res 2006; 70: 401–3.
Oren M . Decision making by p53: life, death and cancer. Cell Death Differ 2003; 10: 431–42.
Vousden KH, Lu X . Live or let die: the cell's response to p53. Nat Rev Cancer 2002; 2: 594–604.
Ciciarello M, Mangiacasale R, Casenghi M, Limongi MZ, D'Angelo M, Soddu S, et al. p53 displacement from centrosomes and p53-mediated G1 arrest following transient inhibition of the mitotic spindle. J Biol Chem 2001; 276: 19205–13.
Karpinich NO, Tafani M, Rothman RJ, Russo MA, Farber JL . The course of etoposide-induced apoptosis from damage to DNA and p53 activation to mitochondrial release of cytochrome c. J Biol Chem 2002; 277: 16547–52.
Shieh SY, Ikeda M, Taya Y, Prives C . DNA damage induced phosphorylation of p53 alleviates inhibition by MDM2. Cell 1997; 91: 325–34.
Canman CE, Lim DS, Cimprich KA, Taya Y, Tamai K, Sakaguchi K, et al. Activation of the ATM kinase by ionizing radiation and phosphorylation of p53. Science 1998; 281: 1677–9.
Hirao A, Kong YY, Matsuoka S, Wakeham A, Ruland J, Yoshida H, et al. DNA damage-induced activation of p53 by the checkpoint kinase Chk2. Science 2000; 287: 1824–7.
Lee TK, Man K, Ho JW, Sun CK, Ng KT, Wang XH, et al. FTY720 induces apoptosis of human hepatoma cell lines through PI3-K-mediated Akt dephosphorylation. Carcinogenesis 2004; 25: 2397–405.
McCubrey JA, Steelman LS, Abrams SL, Lee JT, Chang F, Bertrand FE, et al. Roles of the RAF/MEK/ERK and PI3K/PTEN/AKT pathways in malignant transformation and drug resistance. Advan Enzyme Regul 2006; 46: 249–79.
Kalkman HO . The role of the phosphatidylinositide 3-kinase—protein kinase B pathway in schizophrenia. Pharmacol Therapeut 2006; 110: 117–34.
Trtoman LC, Alimonti A, Scaglioni PP, Koutcher JA, Cordon-Cardo C, Pandolf PP . Identification of a tumor suppressor network opposing nuclear Akt function. Nature 2006; 441: 523–7.
Zhang CL, Wu LJ, Zuo HJ, Tashiro SI, Onodera S, Ikejima T . Cytochrome c release from oridonin-treated apoptosis A375-S2 cells is dependent on p53 and extracellular signal-regulated kinase activation. J Pharmacol Sci 2004; 96: 155–63.
Cohen HY, Miller C, Bitterman KJ, Wall NR, Hekking B, Kessler B . Calorie restriction promotes mammalian cell survival by inducing the SIRT1 deacetylase. Science 2004; 305: 390–2.
Minamino T, Komuro I . Hyperglycemia-induced endothelial senescence mediated by the p53 deacetylase SIRT1 contributes to diabetic vasculopathy. Vascul Pharmacol 2006; 45: e93.
Mao YE, Liu JK, Lu ZX, Yan Z, Liu SF, Li LL, et al. Grifolin, a potential antitumor natural product from the mushroom Albatrellus confluens, inhibits tumor cell growth by inducing apoptosis in vitro. FEBS Lett 2005; 579: 3437–43.
Zhang CL, Wu LJ, Tashiro SI, Onodera S, Ikejima T . Oridonin induces apoptosis of HeLa cells via altering expression of Bcl-2/Bax and activating caspase-3/ICAD pathway. Acta Pharmacol Sin 2004; 25: 691–8.
Wood JG, Rogina B, Lavu S, Howitz K, Helfand SL, Tatar M, et al. Sirtuin activators mimic caloric restriction and delay ageing in metazoans. Nature 2004; 430: 686–9.
Vaziri H, Dessain SK, Ng EE, Imai SI, Frye RA, Pandita TK, et al. Hsir2 (SIRT1) functions as an NAD-dependent p53 deacetylase. Cell 2001; 107: 149–59.
Vercammen D, Beyaet R, Denecker G, Goossens V, Van Loo G, Declercq W, et al. Inhibition of caspases increases the sensitivity of L929 cells to necrosis mediated by tumor necrosis factor. J Exp Med 1998; 187: 1477–85.
Zhang CL, Wu LJ, Tashiro SI, Onodera S, Ikejima T . Oridonin induces a caspase-independent but mitochondria-and MAPK dependent cell death in the murine fibrosarcoma cell line L929. Biol Pharm Bull 2004; 27: 1527–31.
Kalkman HO . The role of the phosphatidylinositide 3-kinase—protein kinase B pathway in schizophrenia. Pharmacol Therapeut 2006; 110: 117–34.
Liu YQ, You S, Tashiro SI, Onodera S, Ikejima T . Activation of phosphoinositide 3-kinase, protein kinase C, and extracellular signal-regulated kinase is required for oridonin-enhanced phagocytosis of apoptotic bodies in human macrophage-like U937 cells. J Pharmacol Sci 2005; 98: 361–71.
Wang XW, Zhan Q, Coursen JD, Khan MA, Kontny HU, Yu LJ, et al. GADD45 induction of a G2/M cell cycle checkpoint. Proc Natl Acad Sci USA 1999; 96: 3706–11.
Ford J, Jiang M, Milner J . Cancer-specific functions of SIRT1 enable human epithelial cancer cell growth and survival. Cancer Res 2005; 65: 10457–63.
Honda R, Tanaka H, Yasuda H . Oncoprotein MDM2 is an ubiquitin ligase E3 for tumor suppressor p53. FEBS Lett 1997; 420: 25–7.
Nakamura S, Roth JA, Mukhopadhyay T . Multiple lysine mutations in the C-terminal domain of p53 interfere with MDM2- dependent protein degradation and ubiquitination. Mol Cell Biol 2000; 20: 9391–8.
Rodriguez MS, Desterro JM, Lain S, Lane DP, Hay RT . Multiple C-terminal lysine residues target p53 for ubiquitinproteasome-mediated degradation. Mol Cell Biol 2000; 20: 8458–67.
Mayo LD, Dixon JE, Durden DL, Tonks NK, Donner DB . PTEN protects p53 from Mdm2 and sensitizes cancer cells to chemotherapy. J Biol Chem 2002; 277: 5484–9.
Mayo LD, Donner DB . The PTEN, Mdm2, p53 tumor suppressoroncoprotein network. Trends Biochem Sci 2002; 27: 462–7.
Ogawara Y, Kishishita S, Obata T, Isazawa Y, Suzuki T, Tanaka K, et al. Akt enhances Mdm2-mediated ubiquitination and degradation of p53. J Biol Chem 2002; 277: 21843–50.
Cui Q, Tashiro SI, Onodera S, Ikejima T . Augmentation of oridonininduced apoptosis observed with reduced autophagy. J Pharmacol Sci 2006; 101: 230–9.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xi, Rg., Huang, J., Li, D. et al. Roles of PI3-K/Akt pathways in nanoparticle realgar powders-induced apoptosis in U937 cells. Acta Pharmacol Sin 29, 355–363 (2008). https://doi.org/10.1111/j.1745-7254.2008.00759.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2008.00759.x