Abstract
Aim:
The aim of the present study is to establish a population pharmacokinetic (PPK) model of valproate (VPA) in Chinese epileptic children to promote the reasonable use of anti-epileptic drugs.
Methods:
Sparse data of VPA serum concentrations from 417 epileptic children were collected. These patients were divided into 2 groups: the PPK model group (n=317) and the PPK valid group (n=100). The PPK parameter values of VPA were calculated by NONMEM software using the data of the PPK model group. A basic model and a final model were set up. To validate the 2 models, the concentrations of PPK valid group were predicted by each model, respectively. The mean prediction error (MPE), mean squared prediction error (MSPE), root mean squared prediction error (RMSPE), weight residues (WRES), and the 95% confidence intervals (95% CI) were also calculated. Then, the values between the 2 models were compared.
Results:
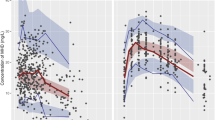
The PPK of VPA was determined by a 1-compartment model with a first-order absorption process. The basic model was: Ka=3.09 (h−1), V/F=20.4 (L), CL/F=0.296 (L/h). The final model was: Ka=0.251+2.24·(1-HS) (h−1), V/F=2.88+0.157·WT (L), CL/F=0.1060.98·CO+ 0.0157·AGE (L/h). For the basic model, the MPE, MSPE, RMSPE, WRES, and the 95% CI were −23.53 (−30.36, −16.70), 3728.96 (2872.72, 4585.20), 39.62 (34.34, 44.90), and −0.06 (−0.14, 0.02), respectively. For the final model, the MPE, MSPE, RMSPE, WRES, and the 95% CI were −1.16 (−4.85, 2.53), 1002.83 (1050.64, 1143.61), 23.04 (21.12, 24.96), and 0.08 (−0.04, 0.20), respectively. The final model was more optimal than the basic model.
Conclusion:
The PPK model of VPA in Chinese epileptic children was successfully established. It will be valuable to facilitate individualized dosage regimens.
Similar content being viewed by others
Article PDF
References
Bourgeois BFD . Antiepileptic drugs in pediatric practice. Epilepsia 1995; 36: S34–45.
Davis R, Peters D V, McTavish D . Valproic acid: a reappraisal of its pharmacological properties and clinical efficacy in epilepsy. Drugs 1994; 47: 332–72.
Bourgeois BFD . Valproic acid: clinical use. In: Levy RH, Mattson RH, Meldrum BS, editors. Antiepileptic drugs. New York: Raven Press; 1995. p 633–9.
Cloyd JC, Fisher JH, Kriel RL, Kraus DM . Valproic acid pharma-cokinetics in children. IV: Effects of age and antiepileptic drugs on protein binding and intrinsic clearance. Clin Pharmacol Ther 1993; 53: 22–9.
Yukawa E, Hokazono T, Satou M, Ohdo S, Higuchi S, Aoyama T . Pharmacokinetic interactions among phenobarbital, carbamaze-pine, and valproic acid in pediatric Japanese patients: clinical considerations on steady-state serum concentration-dose ratios. Am J Ther 2000; 7: 303–8.
Thummel KE, Shen DD . Design and optimization of dosage regimens: pharmacokinetic data. In: Hardman JG, Limbird LE, Gilman AG, editors. Goodman and Gilman's the pharmacological basis of therapeutics; 10th ed. New York: McGraw-Hill; 2001. p 1917–2023.
Llopis-Salvia P, Jimenez-Torres NV . Population pharmacokinetic parameters of vancomycin in critically ill patients. J Clin Pharm Ther 2006; 31: 447–54.
Chen Z Y, Xie HT, Zheng QS, Sun RY, Hu G . Pharmacokinetic and pharmacodynamic population modeling of orally administered rabeprazole in healthy Chinese volunteers by the NONMEM method. Eur J Drug Metab Pharmacokinet 2006; 31: 27–33.
Litalien C, Theoret Y, Faure C . Pharmacokinetics of proton pump inhibitors in children. Clin Pharmacokinet 2005; 44: 441–66.
Carlsson KC, Hoem NO, Glauser T, Vinks AA . Development of a population pharmacokinetic model for carbamazepine based on sparse therapeutic monitoring data from pediatric patients with epilepsy. Clin Ther 2005; 27: 618–26.
Panomvana NA, Ayudhya D, Suwanmanee J, Visudtibhan A . Phar-macokinetic parameters of total and unbound valproic acid and their relationships to seizure control in epileptic children. Am J Ther 2006; 13: 211–7.
El Desoky ES, Fuseau E, EL Din Amry S, Cosson V . Pharmaco-kinetic modelling of valproic acid from routine clinical data in Egyptian epileptic patients. Eur J Clin Pharmacol 2004; 59: 783–90.
Jiao Z, Zhong MK, Hu M, Shi XJ, Li ZD, Zhang JH, et al. Population pharmacokinetic modeling of valproic acid clearance. Chin J Hosp Pharm 2004; 24: 515–7.
Park HM, Kang SS, Lee YB, Shin DJ, Kim ON, Lee SB, et al. Population pharmacokinetics of intravenous valproic acid in Korean patients. J Clin Pharm Ther 2003; 27: 419–25.
Yukawa E, Nonaka T, Yukawa M, Higuchi S, Kuroda T, Goto Y . Pharmacoepidemiologic investigation of a clonazepam-valproic acid interaction by mixed effect modeling using routine clinical pharmacokinetic data in Japanese patients. J Clin Pharm Ther 2003; 28: 497–504.
Hu M, Zhang JH, Sun H, Shi XJ, Yu LY, Zhong MK, et al. Construction and clinical application of the population pharmacoki-netic model of valproic acid. Evaluat Anal Drug-use Hosp China 2001; 1: 95–7.
Serrano BB, Garcia Sanchez MJ, Otero MJ, Buelga DS, Serrano J, Dominguez-Gil A . Valproate population pharmacokinetics in children. J Clin Pharm Ther 1999; 24: 73–80.
Sanchez-Alcaraz A, Quintana MB, Lopez E, Rodriguez I . Valproic acid clearance in children with epilepsy. J Clin Pharm Ther 1998; 23: 31–4.
Yukawa EJ, To H, Ohdo S, Higuchi S, Aoyama T . Population-based investigation of valproic acid relative clearance using nonlinear mixed effects modeling: influence of drug-drug interaction and patient characteristics. J Clin Pharmacol 1997; 37: 1160–7.
Botha JH, Gray AL, Miller R . A model for estimating individualized Valproate clearance values in children. J Clin Pharmacol 1995; 35: 1020–4.
Jiang DC, Wang L . Population pharmacokinetic model of valproate and prediction of valproate serum concentrations in children with epilepsy. Acta Pharmacol Sin 2004; 25: 1576–83.
Wang G, Gu R, Liu B, Jia YT . Population pharmacokinetics of valproic acid in children. Chin J Hosp Pharm 2004; 24: 735–8. Chinese.
Breant V, Charpiat B, Sab JM, Maire P, Jelliffe RW . How many patients and blood levels are necessary for population pharma-cokinetic analysis?. A study of a one compartment model applied to cyclosporine. Eur J Clin Pharmacol 1996; 51: 283–8.
Meibohm B, Laer S, Panetta JC, Barrett JS . Population pharma-cokinetic studies in pediatrics: issues in design and analysis. AAPS J 2005; 7: E475–87.
Duffull S, Waterhouse T, Eccleston J . Some considerations on the design of population pharmacokinetic studies. J Pharmaco-kinet Pharmacodyn 2005; 32: 441–57.
Dodds MG, Hooker AC, Vicini P . Robust population pharmaco-kinetic experiment design. J Pharmacokinet Pharmacodyn 2005; 32: 33–64.
Wang L . Drugs on central nervous system. In: Wang L . Pediatric pharmacology and drug therapeutics. Beijing: Beijing Medical University Press; 2002. p 210–2.
Huang SP, He GZ, Chen ZQ . Research of pharmacokinetics and steady state concentration of valproate in epileptic children. Chin J Pediat 1994; 32: 89–91. Chinese.
Mihaly GW, Vajda FJ, Miles JL . Single and chronic dose pharma-cokinetics of sodium valproate in epileptic patients. Eur J Clin Pharmacol 1979; 16: 23–9.
Bruni J, Wilder BJ, Willmore LJ, Perchalski RJ, Villarreal HJ . Steady-state kinetics of valproic acid in epileptic patients. Clin Pharmacol Ther 1978; 24: 324–32.
Klotz U, Antonin KH . Pharmacokinetics and bioavailability of sodium valproate. Clin Pharmacol Ther 1977; 21: 736–43.
Simpson JA, Hughes D, Manyando C, Bojang K, Aarons L, Winstanley P, et al. Population pharmacokinetic and pharma-codynamic modelling of the antimalarial chemotherapy chlorproguanil/dapsone. Br J Clin Pharmacol 2006; 61: 289–300.
Roy A, Ette EI . A pragmatic approach to the design of population pharmacokinetic studies. AAPS J 2005; 7: E408–20.
Wade JR, Edholm M, Salmonson T . A guide for reporting the results of population pharmacokinetic analyses: a Swedish perspective. AAPS J 2005; 7: 45.
Duffull SB, Kirkpatrick CM, Green B, Holford NH . Analysis of population pharmacokinetic data using NONMEM and WinBUGS. J Biopharm Stat 2005; 15: 53–73.
Kappelhoff BS, Huitema AD, Crommentuyn KM, Mulder JW, Meenhorst PL, van Gorp EC, et al. Development and validation of a population pharmacokinetic model for ritonavir used as a booster or as an antiviral agent in HIV-1-infected patients. Br J Clin Pharmacol 2005; 59: 174–82.
Author information
Authors and Affiliations
Corresponding author
Additional information
This project was supported by the National Natural Science Foundation of China (No 30672504).
Rights and permissions
About this article
Cite this article
Jiang, Dc., Wang, L., Wang, Yq. et al. Population pharmacokinetics of valproate in Chinese children with epilepsy. Acta Pharmacol Sin 28, 1677–1684 (2007). https://doi.org/10.1111/j.1745-7254.2007.00704.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2007.00704.x
Keywords
This article is cited by
-
Effect of CYP2C19, UGT1A8, and UGT2B7 on valproic acid clearance in children with epilepsy: a population pharmacokinetic model
European Journal of Clinical Pharmacology (2018)
-
A population pharmacokinetic model taking into account protein binding for the sustained-release granule formulation of valproic acid in children with epilepsy
European Journal of Clinical Pharmacology (2018)
-
A Population Pharmacokinetic Model of Valproic Acid in Pediatric Patients with Epilepsy: A Non-Linear Pharmacokinetic Model Based on Protein-Binding Saturation
Clinical Pharmacokinetics (2015)
-
Population pharmacokinetics modeling of levetiracetam in Chinese children with epilepsy
Acta Pharmacologica Sinica (2012)
-
Effects of CYP2C19 and CYP2C9 genotypes on pharmacokinetic variability of valproic acid in Chinese epileptic patients: nonlinear mixed-effect modeling
European Journal of Clinical Pharmacology (2009)