Abstract
Aim:
To clarify the cause of poor oral absorption of ginsenoside Rg1 (Rg1), the active ingredient in Panax notoginseng saponins (PNS) used for treating hemorrhage.
Methods:
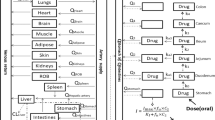
Caco-2 cell monolayers were used as an in vitro model to study the transport mechanism of Rg1 across the intestinal mucosa. Moreover, the serum concentration-time profiles after peroral (po), intraduodenal (id), portal venous (pv) and tail venous (iv) administration of Rg1 in rats were compared to evaluate the first-pass effects in the stomach, intestine, and liver.
Results:
Up take of Rg1 by Caco-2 cell monolayers was temperature-dependent, but was not influenced by cyclosporin A. The change in the apical pH produced no obvious effect on the uptake of Rg1. The uptake and transport of Rg1 was non-saturable; whereas the flux from the apical compartment to the basolateral compartment (A-B) increased in a linear manner with the increase in concentration, indicating passive transport. An apparent permeability coefficient of (2.5 9±0.17) × 10−7 cm/s (C0=1 mg/mL) predicted incomplete absorption. A significant difference was observed between the po (Fpo was 3.29% at a dose of 1500 mg/kg), id Fid was 6.60% at a dose of 1200 mg/kg) and pv (FPV was 50.56%) administration methods, and the barrier function of the intestine was more significant than those of the stomach and liver in the absorption process.
Conclusion:
Elimination in the stomach, large intestine and liver contributed to the low oral bioavailability of Rg1, but low membrane permeability might be a more important factor in determining the extent of absorption.
Similar content being viewed by others
Article PDF
References
Liu M, Zhang JT . Protective effects of ginsenoside Rb1 and Rg1 on cultured hippocampal neurons. Acta Pharm Sin 1995; 30: 674–8.
Radad K, Gille G, Moldzio R, Saito H, Rausch WD . Ginsenosides Rb1 and Rg1 effects on mesencephalic dopaminergic cells stressed with glutamate. Brain Res 2004; 1021: 41–53.
Yamaguchi Y, Higashi M, Kobayashi H . Effects of ginsenosides on impaired performance caused by scopolamine in rats. Eur J Pharmacol 1996; 312: 149–51.
Li X, Chen JX, Sun JJ . Protective effects of Panax notoginseng saponins on experimental myocardial injury induced by ischemia and reperfusion in rat. Acta Pharmacol Sin 1990; 11: 26–9.
Takino Y . Studies on the pharmacodynamics of ginsenoside-Rg1,-Rb1 and-Rb2 in rats. Yakugaku-Zasshi 1994; 114: 550–64.
Kanaoks M, Akao T, Kobashi K . Metabolism of ginseng saponins, ginsenoside, by human intestinal flora. Wakan Ryakugaku Zasshi 1994; 11: 241–6.
Hasegawa H, Sung JH, Matsumiya S, Uchiyama M . Main ginseng saponin metabolites formed by intestinal bacteria. Planta Med 1996; 62: 453–7.
Wang Yi, Liu TH, Wang W, Wang BX . Studies on the metabolism of ginsenoside Rg1 by intestinal bacteria and its absorbed metabolites in rat and human serum. Acta Pharm Sin 2000; 35: 284–8.
Wang Yi, Liu TH, Wang W, Wang BX . Research on the transformation of ginsenoside Rg1 by intestinal flora. China J Chin Materia Med 2001; 26: 188–90.
Odani T, Tanizawa H, Takino Y . Studies on the absorption, distribution, excretion and metabolism of ginseng saponins. II. The absorption, distribution and excretion of ginsenoside Rg1 in the rat. Chem Pharm Bull 1983; 31: 292–8.
Artursson P, Palm K, Luthman K . Caco-2 monolayers in experimental and theoretical predictions of drug transport. Adv Drug Deliv Rev 2001; 46: 27–43.
Azuma R, Hirota T, Manabe H, Komuro M, Kiwada H . First-pass of GTS-21 on canine gut wall and liver determined by portal-systemic concentration difference. Eur J Pharm Sci 2001; 14: 159–65.
Kim SH, Lee MG . Pharmacokinetics of ipriflavone, an isoflavone derivative, after intravenous and oral administration to rats: hepatic and intestinal first-pass effects. Life Sci 2002; 70: 1299–315.
Yamaoka K, Nakagava J, Uno T . Statistical moments in pharmacokinetics. J Pharmacokinet Biopharm 1978; 6: 547–57.
Yamashita S, Furubayashi T, Kataoka M, Sakane T, Sezaki H, Tokuda H . Optimized conditions for prediction of intestinal drug permeability using Caco-2 cells. Pharm Sci 2000; 10: 195–204.
Brunet JL, Maresca M, Fantini J, Belzunces LP . Human intestinal absorption of imidacloprid with Caco-2 cells as enterocyte model. Toxicol Appl Pharm 2004; 192: 1–9.
Rubas W, Zezyk N, Grass GM . Comparison of the permeability characterization of human colonic epithelium (Caco-2) cell line to colon of rabbit, monkey and dog intestine and human drug absorption. Pharm Res 1993; 10: 113–8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Han, M., Fang, Xl. Difference in oral absorption of ginsenoside Rg1 between in vitro and in vivo models. Acta Pharmacol Sin 27, 499–505 (2006). https://doi.org/10.1111/j.1745-7254.2006.00303.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2006.00303.x
Keywords
This article is cited by
-
Ginsenosides emerging as both bifunctional drugs and nanocarriers for enhanced antitumor therapies
Journal of Nanobiotechnology (2021)
-
Ginsenosides, ingredients of the root of Panax ginseng, are not substrates but inhibitors of sodium-glucose transporter 1
Journal of Natural Medicines (2017)
-
Preparation and characterization of mucoadhesive enteric-coating ginsenoside-loaded microparticles
Archives of Pharmacal Research (2015)
-
Increased effects of ginsenosides on the expression of cholesterol 7α-hydroxylase but not the bile salt export pump are involved in cholesterol metabolism
Journal of Natural Medicines (2013)
-
Efficient biotransformation for preparation of pharmaceutically active ginsenoside Compound K by Penicillium oxalicum sp. 68
Annals of Microbiology (2013)