Abstract
Aim:
To investigate the neuroprotective effect of resveratrol in an experimental spinal cord injury (SCI) model in rats.
Methods:
Male Wistar albino rats weighing 200–250 g were randomized into six groups. Weight-drop trauma was performed for SCI. Group 1 underwent laminectomy alone. Group 2 underwent laminectomy followed by SCI. Groups 3, 4, 5, and 6 underwent laminectomy followed by SCI and received resveratrol (100 mg/kg), methylprednisolone (MP) (30 mg/kg), resveratrol (100 mg/kg) plus MP (30 mg/kg), and ethanol (2%), respectively. The rats were divided into two subgroups for biochemical analysis (killed at 24 h after surgery) and for neurobehavioral and histopathological evaluation (killed at 6 weeks after surgery). Posttraumatic neurological recovery after surgery was recorded weekly.
Results:
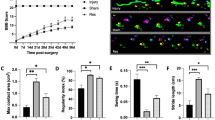
Groups 3 and 5 revealed significantly lower malondialdehyde, nitric oxide, xanthine oxidase, and higher glutathione levels than group 4 (P<0.05). Neurological recovery rates were significantly better in groups 3 and 5 than group 4 (P<0.05). When spinal trauma size ratios were compared, there was no significant difference between treatment groups.
Conclusion:
Resveratrol treatment revealed better biochemical recovery in the acute stage of trauma than MP treatment. Although resveratrol and combined treatment revealed better neurobehavioral recovery than MP treatment; resveratrol, MP, and combined treatment modalities improved histopathological recovery at the same level in the final stage of the experiment. Future studies involving different doses of resveratrol and different doses combinations with MP could promise better results as each drug has a different anti-oxidative mechanism of action.
Similar content being viewed by others
Article PDF
References
Dolan ES, Tator CH . The treatment of hypotension due to acute experimental spinal cord compression injury. Surg Neurol 1980; 13: 380–4.
Ducker TB, Salcman M, Perot PL Jr, Ballantine D . Experimental spinal cord trauma I: correlation of blood flow, tissue oxygen and neurologic status in the dog. Surg Neurol 1978; 10: 60–3.
Hall ED . Lipid peroxidants in acute central nervous system injury. Ann Emerg Med 1993; 22: 1022–7.
Anderson DK, Hall ED . Pathophysiology of spinal cord trauma. Ann Emerg Med 1993; 22: 987–92.
Hall ED . The neuroprotective pharmacology of methylprednisolone. J Neurosurg 1992; 76: 13–22.
Shi MM, Kugelman A, Iwamoto T, Tian L, Forman HJ . Quinone induced oxidative stress elevates glutathione and induces glutamylcysteine synthetase activity in rat lung epithelial L2 cells. J Biol Chem 1994; 269: 26512–7.
Tator CH, Fehlings MG . Review of the secondary injury theory of acute spinal cord trauma with emphasis on vascular mechanisms. J Neurosurg 1991; 75: 15–26.
Young W . Secondary injury mechanisms in acute spinal cord injury. J Emerg Med 1993; 11: 13–22.
Allen AR . Remarks on the histopathological changes in the spinal cord due to impact. An experimental study. J Nerv Ment Dis 1914; 41: 141–7.
Collins WF . A review and update of experimental and clinical studies of spinal cord injury. Paraplegia 1983; 21: 204–19.
Nash HH, Borke RC, Anders JJ . Ensheating cells and methyl-prednisolone promote axonal regeneration and functional recovery in the lesioned adult rat spinal cord. J Neurosci 2002; 22: 7111–20.
Oudega M, Vargas CG, Weber AB, Kleitman N, Bunge MB . Long-term effects of methylprednisolone following transection of the adult rat spinal cord. Eur J Neurosci 1999; 11: 2453–64.
Ji B, Li M, Budel S, Pepinsky RB, Walus L, Engber TM, et al. Effect of combined treatment with methylprednisolone and soluble Nogo-66 receptor after rat spinal cord injury. Eur J Neurosci 2005; 22: 587–94.
Anderson DK, Saunders RS, Demediuk P, Dugan LL, Braughler JM, Hall ED, et al. Lipid hydrolysis and peroxidation in injured spinal cord: partial protection with methylprednisolone or vitamin E and selenium. Cent Nerv Syst Trauma 1985; 2: 257–67.
Soleas GJ, Diamandis EP, Goldberg DM . Wine as a biological fluid: history, production, and role in disease prevention. J Clin Lab Anal 1997; 11: 287–313.
Daniel O, Meier MS, Schlatter J, Frischhnecht P . Selected phenolic compounds in cultivated plants: ecologic functions, health implications, and modulation by pesticides. Environ Health Perspect 1999; 107: 109–14.
Sobolev VS, Cole RJ . Trans-resveratrol content in commercial peanuts and peanut products. J Agric Food Chem 1999; 47: 1435–9.
Inoue H, Umesono K, Nishimori T, Hirata Y, Tanabe T . Gluco-corticoid-mediated suppression of the promoter activity of the cyclooxygenase-2 gene is modulated by expression of its receptor in vascular endothelial cells. Biophys Biophys Res Commun 1999; 254: 292–8.
Sinha K, Chaudray G, Gupta YK . Protective effect of resveratrol against oxidative stres in middle cerebral artery. Life Sci 2002; 71: 655–65.
Inoue H, Tanabe T, Umesono K . Feedback control of cyclooxygenase-2 expression through PPARγ. J Biol Chem 2000; 275: 28028–32.
Huang SS, Tsai MC, Chih CL, Hung LM, Tsai SK . Resveratrol reduction of infarct size in Long-Evans rats subjected to focal cerebral ischemia. Life Sci 2001; 69: 1057–65.
Bloomfield Rubins H, Davenport J, Babikian V, Brass LM, Collins D, Wexler L, et al. VA-HIT Study Group. Reduction in stroke with gemfibrozil in men with coronary heart disease and low HDL cholesterol: The Veterans Affairs HDL Intervention Trial (VA-HIT). Circulation 2001; 103: 2828–33.
Frémont L . Biological effects of resveratrol. Life Sci 2000; 66: 663–73.
Kimura Y, Okuda H, Arichi S . Effects of stilbenes on arachidonate metabolism in leukocytes. Biochim Biophys Acta 1985; 834: 275–8.
Allen AR . Surgery of experimental lesion of spinal cord equivalent to crush injury of fracture dislocation of spinal column. A preliminary report. JAMA 1911; 57: 878–80.
Farooque M, Isaksson J, Jackson D, Olsson Y . Clomethiazole improves hind limb function and reduces neuronal damage after severe spinal cord injury in rat. Acta Neuropathol 1999; 98: 22–30.
Gale K, Kerasidis H, Wrathall JR . Spinal cord contusions in the rat: behavioral analysis of functional neurologic impairment. Exp Neurol 1985; 88: 123–34.
Rivlin AS, Tator CH . Objective clinical assessment of motor function after experimental spinal cord injury in the rat. J Neurosurg 1977; 47: 577–81.
Huang L, Mehta MP, Nanda A, Zhang JH . The role of multiple hyperbaric oxygenation in expanding therapeutic Windows after acute spinal cord injury in rats. J Neurosurg 2003; 99: 198–205.
Mihara M, Uchiyama M . Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal Biochem 1978; 86: 271–8.
Elman GL . Tissue sulphydryl groups. Arch Biochem Biophys 1959; 82: 70–7.
Jungersten L, Edlund A, Petersson AS, Wennmalm A . Plasma nitrate as an index of nitric oxide formation in man: analyses of kinetics and confounding factors. Clin Physiol 1996; 16: 369–79.
Zeballos GA, Bernstein RD, Thompson CI, Forfia PR, Seyedi N, Shen W, et al. Pharmacodynamics of plasma nitrate/nitrite as an indication of nitric oxide formation in conscious dogs. Circulation 1995; 91: 2982–8.
Ozbek E, Turkoz Y, Gokdeniz R, Davarci M, Ozugurlu F . Increased nitric oxide production in the spermatic vein of patients with varicocele. Eur Urol 2000; 37: 172–5.
Prajda N, Weber G . Malignant transformation-linked imbalance: decreased xanthine oxidase activity in hepatomas. FEBS Lett 1975; 59: 245–9.
Yang YB, Piao YJ . Effects of resveratrol on secondary damages after acute spinal cord injury in rats. Acta Pharmacol Sin 2003; 24: 703–10.
Koc RK, Akdemir H, Kurtsoy A, Pasaoglu H, Kavuncu I, Pasaoglu A, et al. Lipid peroxidation in experimental spinal cord injury. Comparison of treatment with Ginkgo biloba, TRH and methyl-prednisolone. Res Exp Med (Berl) 1995; 2: 117–23.
Cayli SR, Kocak A, Yilmaz U, Tekiner A, Erbil M, Ozturk C, et al. Effect of combined treatment with melatonin and methylprednisolone on neurological recovery after experimental spinal cord injury. Eur Spine J 2004; 13: 724–32.
Braughler JM, Hall ED . Correlation of methylprednisolone levels in cat spinal cord with its effects on (Na+K)-ATPase, lipid peroxidation, and alpha motor neuron function. J Neurosurg 1982; 56: 838–44.
Braughler JM, Hall ED . Current application of “high dose” steroid therapy for CNS injury. A pharmacological perspective. J Neurosurg 1985; 62: 806–10.
Young W, Flamm ES . Effects of high-dose corticosteroid therapy on blood flow, evoked potentials and extracellular calcium in experimental spinal cord injury. J Neurosurg 1982; 57: 667–73.
Young W, Bracken MB . The second national acute spinal cord injury study. J Neurotrauma 1992; 9 ( Suppl 1): 397–405.
Kristal BS, Koopmans SJ, Jackson CT, Ikeno Y, Park BS, Yu BP . Oxidant-mediated repression of the mitochondrial transcription in diabetic rats. Free Radic Biol Med 1997; 22: 813–22.
Wang Q, Xu J, Rottinghaus GE, Simonyi A, Lubahn D, Sun GY, et al. Resveratrol protects against global cerebral ischemic injury in gerbils. Brain Res 2002; 958: 439–47.
Cai YJ, Fang JG, Ma LP, Yang L, Liu ZL . Inhibition of free radical-induced peroxidation of rat liver microsomes by resveratrol and its analogues. Biochim Biophys Acta 2003; 1637: 31–8.
Caimi G, Carollo C, Lo Presti R . Chronic renal failure: oxidative stres, endothelial dysfunction and wine. Clin Nephrol 2004; 62: 331–5.
Dernek S, Ikizler M, Erkasap N, Ergun B, Koken T, Yilmaz K . Cardioprotection with resveratrol pretreatment: improved benefical affects over Standard treatment in rat hearts after global ischemia. Scand Cardiovasc J 2004; 38: 245–54.
Kiziltepe U, Turan NN, Han U, Ulus AT, Akar F . Resveratrol, a red wine polyphenol, protects spinal cord from ischemia-reperfusion injury. J Vasc Surg 2004; 40: 138–45.
Olas B, Nowak P, Wachowicz B . Resveratrol protects against peroxynitrite-induced thiol oxidation in blood platelets. Cell Mol Biol Lett 2004; 9: 577–87.
Tadolini B, Juliano C, Piu L, Franconi F, Cabrini L . Resveratrol inhibition of lipid peroxidation. Free Radic Res 2000; 33: 104–14.
Chanvitayapongs S, Draczynska-Lusiak B, Sun AY . Amelioration of oxidative stress by antioxidants and resveratrol in PC12 cells. Neuroreport 1997; 8: 1499–502.
Draczynska-Lusiak B, Chen YM, Sun AY . Oxidized lipoprotein, activate NF-κB binding activity and apoptosis in PC 12 cells. Neuroreport 1998; 9: 527–32.
Zini R, Morin C, Berteli A, Berteli AA, Tillement JP . Effects of resveratrol on the rat brain respiratory chain. Drugs Exp Clin Res 1999; 25: 87–97.
Mizutani K, Ikeda K, Kawai Y, Yamori Y . Protective effect of resveratrol on oxidative damage in male and female stroke-prone spontaneously hypertensive rats. Clin Exp Pharmacol Physiol 2001; 28: 55–9.
Virgili M, Contestabile A . Partial neuroprotection of in vivo exitotoxic brain damage by chronic administration of the red wine antioxidant agents, trans-resveratrol in rats. Neurosci Lett 2000; 281: 123–6.
Kaplan S, Bisleri G, Morgan JA, Cheema FH, Oz MC . Resveratrol, a natural red wine polyphenol, reduces ischemia-reperfusion induced spinal cord injury. Ann Thorac Surg 2005; 80: 2242–9.
Barut S, Canbolat A, Bilge T, Aydin Y, Cokneseli B, Kaya U . Lipid peroxidation in experimental spinal cord injury: time-level relationship. Neurosurg Rev 1993; 16: 53–9.
Constantini S, Young W . The effects of methylprednisolone and the ganglioside GM1 on acute spinal cord injury in rats. J Neurosurg 1994; 80: 97–111.
Sinha K, Deogonakar M, Jagannathan NR, Gupta YK . Effect of melatonin on ischemia reperfusion injury induced by middle cerebral artery occlusion in rats. Eur J Pharmacol 2001; 428: 185–92.
Hile R, Nishino T . Flavoprotein structure and mechanism. 4. Xanthine oxidase and xanthine dehydrogenase. FASEB J 1995; 9: 995–1003.
Corte ED, Stirpe F . The regulation of rat liver xanthine oxidase. Involvement of thiol groups in the conversion of the enzyme activity from dehydrogenase (type D) into oxidase (type O) and purification of the enzyme. Biochem J 1972; 126: 739–45.
Moncada S, Higgs A . The L-arginine-nitric oxide pathway. N Engl J Med 1993; 329: 2002–12.
Peunova N, Enikolopov G . Amplification of calcium-induced gene transcription by nitric oxide in neuronal cells. Nature 1993; 364: 450–3.
Goldberger ME, Bregman BS, Vierck CJ Jr, Brown M . Criteria for assessing recovery of function after spinal cord injury: Behavioral methods. Exp Neurol 1990; 107: 113–7.
Von Euler M, Akesson E, Samuelsson EB, Seiger A, Sundström E . Motor performance score: a new algorithm for accurate behavioral testing of spinal cord injury in rats. Exp Neurol 1996; 137: 242–5.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ates, O., Cayli, S., Altinoz, E. et al. Effects of resveratrol and methylprednisolone on biochemical, neurobehavioral and histopathological recovery after experimental spinal cord injury. Acta Pharmacol Sin 27, 1317–1325 (2006). https://doi.org/10.1111/j.1745-7254.2006.00416.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2006.00416.x
Keywords
This article is cited by
-
The Role of Resveratrol on Spinal Cord Injury: from Bench to Bedside
Molecular Neurobiology (2024)
-
Comparative evaluation of natural neuroprotectives and their combinations on chronic immobilization stress-induced depression in experimental mice
3 Biotech (2023)
-
Effects of resveratrol in the signaling of neuropathic pain involving P2X3 in the dorsal root ganglion of rats
Acta Neurologica Belgica (2021)
-
Resveratrol suppresses glial activation and alleviates trigeminal neuralgia via activation of AMPK
Journal of Neuroinflammation (2016)
-
Efficacy of N-acetylcysteine on neuroclinical, biochemical, and histopathological parameters in experimental spinal cord trauma: comparison with methylprednisolone
European Journal of Trauma and Emergency Surgery (2014)