Abstract
Aim:
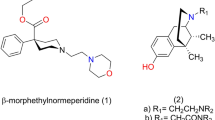
To further investigate the analgesic pharmacophore of (+)-meptazinol.
Methods:
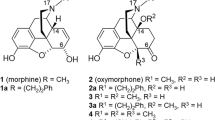
Two different opioid pharmacophores, Pharm-I and Pharm-II, were established from structures of nine typical opiates and meperidine by using molecular modeling approaches according to their different structure activity relationship properties. They were further validated by a set of conformationally constrained arylpiperidines. Two conformers of (+)-meptazinol (Conformer-I and Conformer-II) detected in solution were then fitted into the pharmacophores, respectively, by Fit Atoms facilities available in SYBYL, a computational modeling tool kit for molecular design and analysis.
Results:
Conformer-I fit Pharm-I from typical opiates well. However, Conformer-II fit none of these pharmacophores. Instead, it was found to be similar to another potent analgesic, benzofuro[2,3-c] pyridin-6-ol, whose pharmacophore was suggested to hold the transitional state between the two established pharmacophores. Unlike typical analgesics derived from 4-aryl piperidine (eg, meperidine) with one conformer absolutely overwhelming, the (+)-meptazinol exists in two conformers with similar amounts in solution. Furthermore, both conformers can not transform to each other freely in ordinary conditions based on our NMR results.
Conclusion:
(+)-meptazinol was suggested to be an opioid with mixed analgesic pharmacophores, which may account for the complicated pharmacological properties of meptazinol.
Similar content being viewed by others
Article PDF
References
Hoskin PJ, Hanks GW . Opioid agonist-antagonist drugs in acute and chronic pain states. Drugs 1991; 41: 326–44.
Green D . Current concepts concerning the mode of action of meptazinol as an analgesic. Postgrad Med J 1983; 59: 9–12. Suppl.
Spiegel K, Pasternak GW . Meptazinol: a novel mu-1 selective opioid analgesic. J Pharmacol Exp Ther 1984; 228: 414B.
Li W, Hao JL, Tang Y, Chen Y, Qiu ZB . Structural comparisons of meptazinol with opioid analgesics. Acta Pharmacol Sin 2005; 26: 334–8.
Tillack JV, Seccombe RC, Kennard CHL, Oh PWT . Analgetics. I. Crystal structure of pethidine hydrochloride, 4-carbethoxy-1-4-phenylpiperidine hydrochloride, C15H21NO2·HCl. Recl Trav Chim Pays-Bas 1974; 93: 164–5.
Casy AF, Dewar GH, Al-Deeb OAA . Conformational equilibra of hydrochloride salts of pethidine, ketobemidone, and related central analgesics of the 4-arylpiperidine class. J Chem Soc Perkin Trans 2 1989; 2: 1243–7.
Daum SJ, Martini CM, Kullnig RK, Clarke RL . Analgesic activity of the epimeric tropane analogs of meperidine. Physical and pharmacological study. J Med Chem 1975; 18: 496–501.
Zimmerman DM, Cantrell BE, Swartzendruber JK, Jones ND, Mendelsohn LG, Leander JD, et al. Synthesis and analgesic properties of N-substituted trans-4a-aryldecahydro isoquinolines. J Med Chem 1988; 31: 555–60.
Froimowitz M, Salva P, Hite GJ, Gianntsos G, Suzdak P, Heyman R . Conformational properties of α- and β-azabicyclane opiates. The effect of conformation on pharmacological activity. J Comput Chem 1984; 5; 291–8.
Hutchison AJ, De Jesus R, Wiliams M, Simke JP, Neale RF, Jackson RH, et al. Benzofuro[2,3-c]pyridine-6-ols: synthesis, affinity for opioid subtypes, and antinociceptive activity. J Med Chem 1989; 32: 2221–6.
Froimowitz M, Pick CG, Pasternak GW . Phenylmorphans and analogs-opioid receptor subtype selectivity and effect of conformation on activity. J Med Chem 1992; 35: 1521–5.
Dewar MJS, Zoebisch EG, Healy EF, Stewart JJP . AM1: a new general purpose quantum mechanical model. J Am Chem Soc 1985; 107: 3902–9.
Casy AF, Parfitt RT . Opioid analgesics: Chemistry and receptor. Plenum Press: New York and London, 1986, p 485.
Tyers MB . A classification of opiate receptors that mediate antinociception in animals. Br J Pharmacol 1980; 69: 503–12.
Zimmerman DM, Nickander R, Horng JS, Wong DT . New structural concepts for narcotic-antagonists defined in a 4-phenylpi-peridine series. Nature 1978; 275: 332–4.
Portoghese PS, Alreja BD, Larson DL . Allylprodine analogs as receptor probes - evidence that phenolic and nonphenolic ligands interact with different subsites on identical opioid Receptors. J Med Chem 1981; 24: 782–7.
Casy AF, Ogungbamila FO . Phenolic analogs of reversed esters of pethidine. J Pharm Pharmacol 1985; 37: 121–3.
Horn AS, Rodgers JR . The enkephalins and opiates: structure-activity relations. J Pharm Pharmacol 1977; 29: 257–65.
Hodgson DJ, Rychlewska U, Eliel EL, Manoharan M, Knox DE, Olefirowicz EM . Rotational conformation of the phenyl moiety in geminally substituted phenylcyclohexanes with equatorial phenyl. J Org Chem 1985; 50: 4838–43.
Allinger NL, Tribble MT . Conformational analysis LXXVIII. The conformation of phenyl cyclohexane and related molecules. Tetrahedron Lett 1971; 12: 3259–62.
Burke TRJ, Bajwa BS, Jacobson AE, Rice KC, Streaty RA, Klee WA . Probes for narcotic receptor mediated phenomena 7. Synthesis and pharmacological properties of irreversible ligands specific for mu and delta opiate receptors. J Med Chem 1984; 27: 1570–4.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the National Natural Science Foundation of China (No 30271539, 2003-2005).
Rights and permissions
About this article
Cite this article
Li, W., Wang, Xh., Lau, Cw. et al. Conformational re-analysis of (+)-meptazinol: an opioid with mixed analgesic pharmacophores. Acta Pharmacol Sin 27, 1247–1252 (2006). https://doi.org/10.1111/j.1745-7254.2006.00375.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2006.00375.x