Abstract
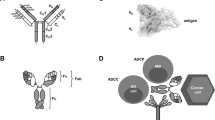
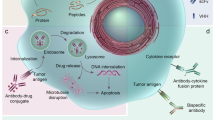
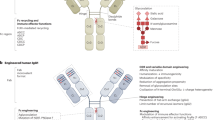
Antibody-based therapeutics are beginning to realize the promise enclosed in their early denomination as “magic bullets”. Initial disappointment has turned into clinical and commercial success, and engineered antibodies currently represent over 30% of biopharmaceuticals in clinical trials. Recent structural and functional data have allowed the design of a new generation of therapeutic antibodies, with strategies ranging from complement-mediated and antibody-dependant cellular cytotoxicity enhancement to improved cytotoxic payloads using toxins, drugs, radio nuclides and viral delivery. This review considers the structure of different types of recombinant antibodies, their mechanism of action and how their efficacy has been increased using a broad array of approaches. We will also focus on the additional benefits offered by the use of gene therapy methods for the in vivo production of therapeutic antibodies.
Similar content being viewed by others
Article PDF
References
Kohler G, Milstein C . Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 1975; 256: 495–7.
Carter P . Improving the efficacy of antibody-based cancer therapies. Nat Rev Cancer 2001; 1: 118–29.
Hudson PJ, Souriau C . Engineered antibodies. Nat Med 2003; 9: 129–34.
Brekke OH, Sandlie I . Therapeutic antibodies for human diseases at the dawn of the twenty-first century. Nat Rev Drug Disc 2003; 2: 52–62.
Shields RL, Namenuk AK, Hong K, Meng YG, Rae J, Briggs J, et al. High resolution mapping of the binding site on human IgG1 for Fc gamma RI, Fc gamma RII, Fc gamma RIII, and FcRn and design of IgG1 variants with improved binding to the Fc gamma R. J Biol Chem 2001; 276: 6591–604.
Idusogie EE, Wong PY, Presta LG, Gazzana-Santoro H, Totpal K, Ultsch M, et al. Engineered antibodies with increased activity to recruit complement. J Immunol 2001; 166: 2571–5.
Umanña P, Jean-Mairet J, Moudry R, Am Stutz H, Bailey JE . Engineered glycoforms of an anti neuroblystoma IgG1 with optimized antibody-dependent cellular cytotoxic activity. Nat Biotechnol 1999; 17: 176–80.
Ghetie V, Popov S, Borvak J, Radu C, Matesoi D, Medesan C, et al. Increasing the serum persistence of an IgG fragment by random mutagenesis. Nat Biotechnol 1997; 15: 637–40.
Pluckthun A . Mono- and bivalent antibody fragments produced in Escherichia coli: engineering, folding and antigen binding. Immunol Rev 1992; 130: 151–88.
Holliger P, Prospero T, Winter G . “Diabolizes”: small bivalent and bi specific antibody fragments. Proc Natl Acad Sci USA 1993; 90: 6444–8.
Kontermann RE . Recombinant bi specific antibodies for cancer therapy. Acta Pharmacol Sin 2005; 26: 1–9.
Natarajan A, Xiong CY, Albrecht H, De Nardo GL, De Nardo SJ . Characterization of site-specific Sc Fv Pagination for tumor-targeting pharmaceuticals. Bioconjug Chem 2005; 16: 113–21.
Smallshaw JE, Ghetie V, Rizo J, Fulmer JR, Trahan LL, Ghetie MA, et al. Genetic engineering of an immunotoxin to eliminate pulmonary vascular leak in mice. Nat Biotechnol 2003; 21: 387–91.
Goldenberg DM . Advancing role of radiolabeled antibodies in the therapy of cancer. Cancer Immunol Immunother 2003; 52: 281–96.
Xu G, McLeod HL . Strategies for enzyme/pro drug cancer therapy. Clin Cancer Res 2001; 7: 3314–24.
Sharma SK, Pedley RB, Bhatia J, Boxer GM, El-Emir E, Qureshi U, et al. Sustained tumor regression of human colorectal cancer xenografts using a multifunctional mannosylated fusion protein in antibody-directed enzyme pro drug therapy. Clin Cancer Res 2005; 11: 814–25.
Shabat D, Lode HN, Pertl U, Reisfeld RA, Rader C, Lerner RA, et al. In vivo activity in a catalytic antibody-pro drug system: antibody catalyzed etoposide pro drug activation for selective chemotherapy. Proc Natl Acad Sci USA 2001; 98: 7528–33.
Nissim A, Gofur Y, Vessillier S, Adams G, Chernajovsky Y . Methods for targeting biologicals to specific disease sites. Trends Mol Med 2004; 10: 269–74.
Hombach A, Heuser C, Abken H . Simultaneous targeting of IL2 and IL12 to Hodgkin's lymphoma cells enhances activation of resting NK cells and tumor cell lysis. Int J Cancer. 2005; 115: 241–7.
Gillies SD, Lan Y, Williams S, Carr F, Forman S, Raubitschek A, et al. An anti-CD20–IL-2 immunocytokine is highly efficacious in a SCID mouse model of established human B lymphoma. Blood. 2005; 105: 3972–8.
Halin C, Rondini S, Nilsson F, Berndt A, Kosmehl H, Zardi L, et al. Enhancement of the antitumor activity of interleukin-12 by targeted delivery to neovasculature. Nat Biotechnol 2002; 20: 264–9.
Sanz L, Blanco B, Alvarez-Vallina L . Antibodies and gene therapy: teaching old ‘magic bullets’ new tricks. Trends Immunol 2004; 25: 85–91.
Pelegrin M, Gros L, Dreja H, Piechaczyk M . Monoclonal antibody-based genetic immunotherapy. Curr Gene Ther 2004; 4: 347–56.
Arafat WO, Gomez-Navarro J, Buchsbau DJ, Xiang J, Wang M, Casado, et al. Effective single chain antibody (scFv) concentrations in vivo via adenoviral vector mediated expression of secretory scFv. Gene Ther 2002; 9: 256–62.
Sanz L, Kristensen P, Russell SJ, Ramirez Garcia JR, Alvarez-Vallina L . Generation and characterization of recombinant human antibodies specific for native laminin epitopes: potential application in cancer therapy. Cancer Immunol Immunother 2001; 50: 557–65.
Sanz L, Garcia-Bermejo L, Blanco FJ, Kristensen P, Feijoo M, Suarez E, et al. A novel cell binding site in the coiled-coil domain of laminin involved in capillary morphogenesis. EMBO J 2003; 22: 1508–17.
Sanz L, Kristensen P, Blanco B, Facteau S, Russell SJ, Winter G, et al. Single-chain antibody-based gene therapy: inhibition of tumor growth by in situ production of phage-derived human antibody fragments blocking functionally active sites of cell-associated matrices. Gene Ther 2002; 9: 1049–53.
Blanco B, Holliger P, Vile R, Alvarez-Vallina L . Induction of human T lymphocyte cytotoxicity and inhibition of tumor growth by tumor-specific body-based molecules secreted from gene-modified bystander cells. J Immunol 2003; 171: 1070–7.
Baxenavis CN, Papamichail M . Targeting of tumor cells by lymphocytes engineered to express chimeric receptor genes. Cancer Immunol Immunother 2004; 53: 893–903.
Álvarez-Vallina L . Genetic approaches for antigen-selective cell therapy. Curr Gene Ther 2001; 4: 385–97.
Brentjens RJ, Latouche JB, Santos E, Marti F, Gong MC, Lyddane C et al. Eradication of systemic B-cell tumors by genetically targeted human T lymphocytes co-stimulated by CD80 and interleukin-15. Nat Med 2003; 9: 279–86.
Uherek C, Tonn T, Uherek B, Becker S, Schnierle B, Klingemann HG, et al. Retargeting of natural killer-cell cytolytic activity to ErbB2–expressing cancer cells results in efficient and selective tumor cell destruction. Blood 2002; 100: 1265–73.
Alvarez-Vallina L, Hawkins RE . Antigen-specific targeting of CD28-mediated T cell co-stimulation using chimeric single-chain antibody variable fragment-CD28 receptors. Eur J Immunol 1996; 26: 2304–9.
Finney HM, Lawson AD, Bebbington CR, Weir AN . Chimeric receptors providing both primary and costimulatory signaling in T cells from a single gene product. J Immunol 1998; 161: 2791–7.
Sanz L, Qiao J, Vile R, Alvarez-Vallina L . Antibody engineering, virus retargeting and cellular immunotherapy: one ring to rule them all? Curr Gene Ther 2005; 5: 63–70.
Korokhov N, Mikheeva G, Krendelshchikov A, Belousova N, Simonenko V, Krendelshchikov V, et al. Targeting of adenovirus via genetic modification of the viral capsid combined with a protein bridge. J Virol 2003; 77: 12931–40.
Volpers C, Thirion C, Biermann V, Hussmann S, Kewes H, Dunant P, et al. Antibody-mediated targeting of an adenovirus vector modified to contain a synthetic immunoglobulin g-binding domain in the capsid. J Virol 2003; 77: 2093–104.
Russell SJ, Hawkins RE, Winter G . Retroviral vectors displaying functional antibody fragments. Nucleic Acids Res 1993; 21: 1081–5.
Somia NV, Zoppe M, Verma IM . Generation of targeted retroviral vectors by using single-chain variable fragment: an approach to in vivo gene delivery. Proc Natl Acad Sci USA 1995; 92: 7570–4.
Chu TH, Dornburg R . Toward highly efficient cell-type-specific gene transfer with retroviral vectors displaying single-chain antibodies. J Virol 1997; 71: 720–5.
Martin F, Neil S, Kupsch J, Maurice M, Cosset F, Collins M . Retrovirus targeting by tropism restriction to melanoma cells. J Virol 1999; 73: 6923–9.
Morizono K, Xie Y, Ringpis GE, Johnson M, Nassanian H, Lee B, et al. Lentiviral vector retargeting to P-glycoprotein on meta-static melanoma through intravenous injection. Nat Med 2005; 11: 346–52.
Hammond AL, Plemper RK, Zhang J, Schneider U, Russell SJ, Cattaneo R . Single-chain antibody displayed on a recombinant measles virus confers entry through the tumor-associated carcinoembryonic antigen. J Virol 2001; 75: 2087–96.
Bucheit AD, Kumar S, Grote DM, Lin Y, von Messling V, Cattaneo RB, et al. An oncocytic measles virus engineered to enter cells through the CD20 antigen. Mol Ther 2003; 7: 62–72.
Peng KW, Donovan KA, Schneider U, Cattaneo R, Lust JA, Russell SJ . Oncocytic measles viruses displaying a single-chain antibody against CD38, a myeloma cell marker. Blood 2003; 101: 2557–62.
Lobato MN, Rabbits TH . Intracellular antibodies and challenges facing their use as therapeutic agents. Trends Mol Med 2003; 9: 390–6.
Stocks MR . Intra bodies: production and promise. Drug Discov Today 2004; 9: 960–6.
Jendreyko N, Popkov M, Beerli RR, Chung J, McGavern DB, Rader C et al. Intra-diabolizes: biospecific, tetravalent antibodies for the simultaneous functional knockout of two cell surface receptors. J Biol Chem 2003; 278: 47812–9.
Rousselet N, Mills L, Jean D, Tellez C, Bar-Eli M, Frade R . Inhibition of tumorigenicity and metastasis of human melanoma cells by anti-cathepsin L single chain variable fragment. Cancer Res 2004; 64: 146–51.
Popkov M, Jendreyko N, McGavern DB, Rader C, Barbas CF III . Targeting tumor angiogenesis with adenovirus-delivered anti-Tie-2 intra body. Cancer Res 2005; 65: 972–81.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by grants from the Fondo de Investigación Sanitaria (grant 02/1144), from the Comunidad Autónoma de Madrid (grant GR/SAL/0214/2004) and from the 5th framework of the European Community (grant EC QLK3-CT-1999-00386).
Rights and permissions
About this article
Cite this article
Sanz, L., Cuesta, Á., Compte, M. et al. Antibody engineering: facing new challenges in cancer therapy. Acta Pharmacol Sin 26, 641–648 (2005). https://doi.org/10.1111/j.1745-7254.2005.00135.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2005.00135.x
Keywords
This article is cited by
-
Intramolecular trimerization, a novel strategy for making multispecific antibodies with controlled orientation of the antigen binding domains
Scientific Reports (2016)
-
In vivo secretion of anti-CD3 × anti-tumor bispecific antibodies by gene-modified cells: over a decade of T-cell engagement
Molecular Therapy (2015)
-
Preparation of Fab-Fragments of GD2-Specific Antibodies and Analysis of Their Antitumor Activity In Vitro
Bulletin of Experimental Biology and Medicine (2013)
-
Development trends for therapeutic antibody fragments
Nature Biotechnology (2009)
-
Recent advances in the generation of human monoclonal antibody
Cytotechnology (2007)