Abstract
Aim:
To evaluate the analgesic effects of 2 celecoxib derivatives and their inhibitory effects on cyclooxygenase (COX).
Methods:
Four antinociceptive assays were used: the acetic acid-induced writhing test, hot plate test, hot tail-flick test and formalin test. Three doses were used in the analgesic assays and ED50 values were calculated. For the selectivity assay, macrophages were incubated with test compounds at various concentrations and then stimulated with calcimycin or lipopolysaccharide (LPS). The amounts of 6-keto-prostaglandin F1α (6-keto-PGF1α) and prostaglandin E2 (PGE2) in the supernatant were examined by radioimmunoassay (RIA). The selectivity of the test compounds was expressed as the IC50, COX-1/IC50,COX-2 value.
Results:
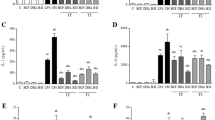
Celecoxib and its 2 derivatives had a significant analgesic effect. The ED50 values of celecoxib, PC-406 and PC-407 were 94.2, 67.9, and 63.3 mg/kg, respectively, for the acetic acid-induced writhing test; 104.7, 89.1, and 30.0 mg/kg, respectively, for the hot tail-flick response test; 60.7, 56.7, and 86.2 mg/kg, respectively, for the hot plate response test; 67.1, 55.8, and 68.8 mg/kg, respectively, for the formalin-induced response. That is, the ED50 of PC-406 was the lowest for the formalin and hot plate tests, which focus on changes above the spinal cord level; however, the ED50 of PC-407 was lowest for the tail-flick and writhing tests, which focus on changes at the spinal cord level. Celecoxib and PC-407 inhibited COX-1 with IC50 values of 39.8 and 27.5 nmol/L, respectively. PC-406 inhibited COX-1 with an IC50 value of more than 1000 nmol/L. The IC50 values for the effect of celecoxib, PC-406 and PC-407 on COX-2 were 4.8, 8.9, and 1.9 nmol/L respectively. The IC50,COX-1/IC50,COX-2 ratios for celecoxib and PC-407 were 8.3 and 14.4, respectively. For PC-406, the ratio was greater than 112.2.
Conclusion:
Derivatives of celecoxib via substitution with an isopropyl or naphthyl group at the 5 position in the pyrazole ring still have analgesic effects and the ability to selectively inhibit COX-2. Substitution with a naphthyl group may have more effect on the peripheral pain pathway, whereas substitution with an isopropyl group may have more effect on the central pain pathway. This phenomenon occurs partly because substitution with an isopropyl group is more beneficial for COX-2 selectivity than is substitution with a naphthyl group.
Similar content being viewed by others
Article PDF
References
Crofford LJ . COX-1 and COX-2 tissue expression: implications and predictions. J Rheumatol 1997; 24 Suppl 49: 15–9.
Lane JM . Anti-inflammatory medications: selective COX-2 inhibitors. J Am Acad Orthop Surg 2002; 10: 75–8.
Zhang BL, Mei QB, He W, Zhou SY . Synthesis of celecoxib. Chin J New Drug 2002; 11: 859–61.
Zhang BL, Mei QB, Zhou SY, He W, Chen CS . Quantitative structure-activity relationship of the 1, 5-diarylpyrazole class of cyclooxygenase-2 selective inhibitors. J Fourth Milit Med Univ 2003; 24: 1523–5.
Ewell RD, Spencer PS . Antinociceptive activity of narcotic agonist and partial agonist analgesics and other agents in the tail-immersion test in mice and rats. Neuropharmacology 1976; 15: 683–8.
Baker AK, Hoffmann VL, Meert TF . Dextromethorphan and ketamine potentiate the antinociceptive effects of mu- but not delta- or kappa-opioid agonists in a mouse model of acute pain. Pharmacol Biochem Behav 2002; 74: 73–86.
Gierse JK, Hauser SD, Creely DP, Koboldt C, Rangwala SH, Isakson PC, et al. Expression and selective inhibition of the constitutive and inducible forms of human cyclo-oxygenase. Biochem J 1995; 305: 479–84.
Brideau C, Kargman S, Liu S, Dallob AL, Ehrich EW, Rodger IW, et al. A human whole blood assay for clinical evaluation of biochemical efficacy of cyclooxygenase inhibitors. Inflamm Res 1996; 45: 68–74.
Riendeau D, Percival MD, Boyce S, Brideau C, Charleson S, Cromlish W, et al. Biochemical and pharmacological profile of a tetrasubstituted furanone as a highly selective COX-2 inhibitor. Br J Pharmacol 1997; 121: 105–17.
Lazer ES, Miao CK, Cywin CL, Sorcek R, Wong HC, Meng Z, et al. Effect of structural modification of enol-carboxamide-type nonsteroidal antiinflammatory drugs on COX-2/COX-1 selectivity. J Med Chem 1997; 40: 980–9.
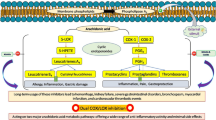
Naraba H, Murakami M, Matsumoto H, Shimbara S, Ueno A, Kudo I, et al. Segregated coupling of phospholipases A2, cyclooxygenases, and terminal prostanoid synthases in different phases of prostanoid biosynthesis in rat peritoneal macrophages. J Immunol 1998; 160: 2974–82.
Shen F, Bai AP, Guo ZR, Cheng CF . Inhibitory effect of 3,4-diaryl-3-pyrrolin-2-one derivatives on cyclooxygenase 1 and 2 in murine peritoneal macrophages. Acta Pharmacol Sin 2002; 23: 762–8.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the National Natural Science Foundation of China No 30400548).
Rights and permissions
About this article
Cite this article
Lu, Zh., Xiong, Xy., Zhang, Bl. et al. Evaluation of 2 celecoxib derivatives: analgesic effect and selectivity to cyclooxygenase-2/1. Acta Pharmacol Sin 26, 1505–1511 (2005). https://doi.org/10.1111/j.1745-7254.2005.00222.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2005.00222.x
Keywords
This article is cited by
-
Cost-utility analysis of imrecoxib compared with diclofenac for patients with osteoarthritis
Cost Effectiveness and Resource Allocation (2021)