Abstract
Objective:
Offspring of diabetes patients may suffer from tissue iron deficiency. Erythrocyte zinc protoporphyrin/heme (ZnPP/H) ratios measure impaired iron status. The aim of the study was to examine whether cord ZnPP/H ratios were associated with pregnancy glycemic control.
Methods:
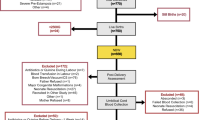
ZnPP/H was measured in cord blood from 31 pregnancies with insulin-treated diabetes (diabetes group) and compared to population normal values. Maternal glycemic control was assessed by daily glucose log, glycosylated hemoglobin and birth weight.
Results:
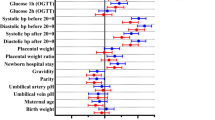
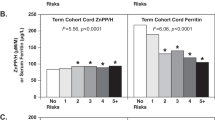
Median cord ZnPP/H was higher in the diabetes group than the population normal values (106 (65.2 to 146.8)?μ M/M vs 68.2 (37.6 to 98.8)?μ M/M, P<0.0001). Ratios were directly correlated to surrogates of control (glycosylated hemoglobin, P=0.05, and birth weight, P<0.04). Cord ZnPP/H ratios from pregnancies with pre-existing and gestational diabetes were similar.
Conclusion:
Because cord ZnPP/H was higher in large offspring of diabetic pregnancy, it might identify greater iron utilization for fetal erythropoiesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Widness JA, Susa JB, Garcia JF, Singer DB, Sehgal P, Oh W et al. Increased erythropoiesis and elevated erythropoietin in infants born to diabetic mothers and in hyperinsulinemic rhesus fetuses. J Clin Invest 1981; 67: 637–642.
Georgieff MK, Landon MB, Mills MM, Hedlund BE, Faassen AE, Schmidt RL et al. Abnormal iron distribution in infants of diabetic mothers: spectrum and maternal antecedents. J Pediatr 1990; 117: 455–461.
Widness JA, Teramo KA, Clemons GK, Voutilainen P, Stenman U-H, McKinlay S et al. Direct relationship of antepartum glucose control and fetal erythropoietin in human Type 1 (insulin-dependent) diabetic pregnancy. Diabetologia 1990; 33: 378–383.
Schwartz R, Teramo KA . Effects of diabetic pregnancy on the fetus and newborn. Semin Perinatol 2000; 24: 120–135.
Petry CD, Eaton MA, Wobkin JD, Mills MM, Johnson DE, Georgieff MK . Iron deficiency of liver, heart, and brain in newborn infants of diabetic mothers. J Pediatr 1992; 121: 109–114.
Stehbens JA, Baker GL, Kitchell M . Outcomes at ages 1, 3, and 5 years of children born to diabetic mothers. Am J Obstet Gynecol 1977; 127: 408–413.
Lozoff B, Jimenez E, Wolf A . Long term developmental outcome of infants with iron deficiency. N Engl J Med 1991; 325: 687–694.
Lozoff B, Jimenez E, Hagen J, Mollen E, Wolf AW . Poorer behavioral and developmental outcome more than 10 years after treatment for iron deficiency in infancy. Pediatrics 2000; 105: e51.
Nelson CA, Wewerka SW, Thomas KM, Tribby-Waldbridge S, deRegnier R-A, Geogieff MK . Neurocognitive sequelae of infants of diabetic mothers. Behav Neurosci 2000; 114: 950–956.
deUngria M, Rao R, Wobken JD, Luciana M, Nelson CA, Georgieff MK . Perinatal iron deficiency decreases cytochrome c oxidase (CytOx) activity in selected regions of neonatal rat brain. J Pediatr 2000; 48: 169–176.
deRegnier R-A, Nelson CA, Thomas KM, Wewerka S, Georgieff MK . Neurophysiologic evaluation of auditory recognition memory in healthy newborn infants and infants of diabetic mothers. J Pediatr 2000; 137: 777–784.
Siddappa AM, Georgieff MK, Wewerka SW, Worwa C, Nelson CA, deRegnier R-A . Iron deficiency alters auditory recognition memory in newborn infants of diabetic mothers. Pediatr Res 2004; 55: 1034–1041.
Labbe' RF, Finch CA, Smith NJ, Doan RN, Sood SK, Madan N . Erythrocyte protoporphyrin/heme ratio in the assessment of iron status. Clin Chem 1979; 25: 87–92.
Schifman RB, Rivers SL, Finley PR, Thies C . RBC zinc protoporphyrin to screen blood donors for iron deficiency anemia. JAMA 1982; 248: 2012–2015.
NCCLS. Erythrocyte Protoporphyrin Testing: Approved Guideline (NCCLS Document C42-A). National Committee on Clinical Laboratory Standards: Villanova, PA, 1996.
Labbe' RF, Dewanji A . Iron assessment tests: transferrin receptor vis-a-vis zinc protoporphyrin. Clin Biochem 2004; 37: 165–174.
Rettmer RL, Carlson TH, Origenes ML, Jack RM, Labbe' RF . Zinc protoporphyrin/heme ratio for diagnosis of preanemic iron deficiency. Pediatrics 1999; 104: e37.
Arbuckle TE, Wilkins R, Sherman GJ . Birth weight percentiles by gestational age in Canada. Obstet Gynecol 1993; 81: 39–48.
Carpenter MW, Coustan DR . Criteria for screening tests for gestational diabetes. Am J Obstet Gynecol 1982; 144: 768–773.
Hastka J, Lasserre JJ, Schwarzbeck A, Strauch M, Hehlmann R . Washing erythrocytes to remove interferents in measurements of zinc protoporphyrin by front-face hematofluorometry. Clin Chem 1992; 38: 2184–2189.
Lott DG, Labbe' RF, Widness JA . Erythrocyte zinc protoporphyrin is elevated with prematurity and fetal hypoxemia. Pediatrics 2005; 116: 414–422.
Winzerling JJ, Kling PJ . Iron deficient erythropoiesis in premature infants measured by blood zinc protoporphyrin/heme. J Pediatr 2001; 139: 134–136.
Juul SE, Zerzan JC, Strandjord TP, Woodrum DE . Zinc protoporphyrin/heme as an indicator of iron status in NICU patients. J Pediatr 2003; 142: 273–278.
Rizzo TA, Metzger BE, Dooley SL, Nam CH . Early malnutrition and child neurobehavioral development: insights from the study of children of diabetic mothers. Child Dev 1997; 68: 26–38.
Nylund L, Lunell NO, Lewander R, Persson B, Sarby B . Uteroplacental blood flow in diabetic pregnancy: measurements with indium 113m and computer-linked gamma camera. Am J Obstet Gynecol 1982; 144: 298–302.
Petry CD, Wobkin JD, McKay H, Eaton MA, Seybold VS, Johnson DE et al. Placental transferrin receptor in diabetic pregnancies with increased fetal iron demand. Am J Physiol 1994; 267: E507–E517.
Shannon K, Davis JC, Kitzmiller JL, Fulcher SA, Koenig HM . Erythropoiesis in infants of diabetic mothers. Pediatr Res 1986; 20: 161–165.
Lubetzky R, Ben-shachar S, Mimouni FB, Dollberg S . Mode of delivery and neonatal hematocrit. Am J Perinatol 2000; 17: 163–165.
Monzon CM, Beaver BD, Dillon TD . Evaluation of erythrocyte disorders with mean corpuscular volume (MCV) and red cell distribution width (RDW). Clin Pediatr 1987; 26: 632–638.
Novak RW . Red blood cell distribution width in pediatric microcytic anemias. Pediatrics 1987; 80: 251–254.
Homko CJ, Reece EA . To screen or not to screen for gestational diabetes mellitus – the clinical quagmire. Clin Perinatol 2001; 28: 407–417.
Coustan DR . Management of gestational diabetes: a self-fulfilling prophecy. JAMA 1996; 275: 1199.
Acknowledgements
We acknowledge the assistance of John A Widness, MD, Michael K Georgieff, MD, David G Lott, MD and Carrie Daniel. This work was supported by grant from the American Heart Association, Southwest Affiliate (SW-GS-16-98) (PJK) and a gift from the Sparks family to the University of Arizona Children's Research Center (PJK).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lesser, K., Schoel, S. & Kling, P. Elevated zinc protoporphyrin/heme ratios in umbilical cord blood after diabetic pregnancy. J Perinatol 26, 671–676 (2006). https://doi.org/10.1038/sj.jp.7211600
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.jp.7211600
Keywords
This article is cited by
-
Impact and interactions between risk factors on the iron status of at-risk neonates
Journal of Perinatology (2022)
-
Maternal pregnancy weight gain and cord blood iron status are associated with eosinophilia in infancy
Journal of Perinatology (2015)
-
Neonatal iron status is impaired by maternal obesity and excessive weight gain during pregnancy
Journal of Perinatology (2014)