Abstract
OBJECTIVE: To determine if treatment of Ureaplasma urealyticum (Uu), found at the time of an acute respiratory deterioration, decreases the incidence of chronic lung disease (CLD) in very low birth weight infants (VLBW).
STUDY DESIGN: Between 1996 and 1999, medical records of all mechanically ventilated VLBW infants, who had an acute respiratory deterioration, were reviewed for gestational age (GA), birth weight (BW), gender, presence of CLD, Uu tracheal cultures, and erythromycin treatment.
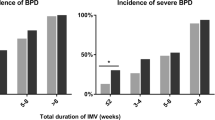
RESULTS: A total of 100 patients met our inclusion criteria (GA: 26.2±1.7 weeks, BW: 737±167.1 g (mean±SD)). Uu was present in 46.3% (38/82) of patients with CLD versus 50% (9/18) of patients without CLD (odds ratio 0.86 (CI: 0.31 to 2.39); p=0.77). Erythromycin treatment was not found to be protective against the development of CLD (odds ratio: 1.46 (CI: 0.25 to 8.31); p=0.66).
CONCLUSION: Following an acute respiratory deterioration, tracheal isolation, and treatment of Uu may not decrease the incidence of CLD in VLBW infants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Sanchez PJ, Regan JA . Vertical transmission of Ureaplasma urealyticum from mothers to preterm infants. Pediatr Infect Dis J 1990;9(6):398–401.
Alfa MJ, Embree JE, Degagne P et al. NT, Hall PF. Transmission of Ureaplasma urealyticum from mothers to full and preterm infants. Pediatr Infect Dis J 1995;14:341–345.
Wang EE, Ohlsson A, Kellner JD . Association of Ureaplasma urealyticum colonization with chronic lung disease of prematurity: results of a meta-analysis. J Pediatr 1995;127(4):640–644.
Ollikainen J, Kiekkaniemi H, Korppi M, Sarkkinnen H, Heinonen K . Ureaplasma urealyticum infection associated with acute respiratory insufficiency and death in premature infants. J Pediatr 1993;122:756–760.
Heggie AD, Jacobs MR, Butler VT, Baley JE, Boxerbaum B . Frequency and significance of isolation of Ureaplasma urealyticum and Mycoplasma hominis from cerebrospinal fluid and tracheal aspirate specimens from low birth weight infants. J Pediatr 1994;124(6):956–961.
Crouse DT, Odrezin GT, Cutter GR et al. Radiographic changes associated with tracheal isolation of Ureaplasma urealyticum from neonates. Clin Infect Dis 1993;17 (Suppl 1):S122–S130.
Farrar HC, Walsh-Sukys MC, Kyllonen K, Blumer JL . Cardiac toxicity associated with intravenous erythromycin lactobionate: two case reports and review of the literature. Pediatr Infect Dis J 1993;12(8):688–691.
Lyon AJ, McColm J, Middlemist L, Fergusson S, Mc Intosh N, Ross PW . Randomised trial of erythromycin on development of chronic lung disease in preterm infants. Arch Dis Child Fetal Neonatal Ed 1998;78:F10–F14.
Jonsson B, Rylander M, Faxelius G . Ureaplasma urealyticum, erythromycin and respiratory morbidity in high-risk preterm neonates. Acta Paediatr 1998;87:1079–1084.
Iles R, Lyon A, Ross P, McIntosh N . Infection with Ureaplasma urealyticum and Mycoplasma hominis and development of chronic lung disease in preterm infants. Acta Paediatr 1996;85:482–484.
Da Silva O, Gregson D, Hammerberg O . Role of Ureaplasma urealyticum and Chlamydia tracheomatis in development of bronchopulmonary dysplasia in very low birth weight infants. Pediatr Infect Dis J 1997;16:364–369.
Saxen H, Hakkarainen K, Pohjavuori M, Miettinen A . Chronic lung disease of preterm infants in Finland is not associated with Ureaplasma urealyticum colonization. Acta Paediatr 1993;82:198–201.
Bowman ED, Dharmalingam A, Fan WQ, Brown F, Garland S . Impact of erythromycin on respiratory colonization of Ureaplasma urealyticum and the development of chronic lung disease in extremely low birth weight infants. Pediatr Infect Dis J 1998;17:615–620.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mhanna, M., Delong, L. & Aziz, H. The Value of Ureaplasma urealyticum Tracheal Culture and Treatment in Premature Infants Following an Acute Respiratory Deterioration. J Perinatol 23, 541–544 (2003). https://doi.org/10.1038/sj.jp.7210978
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.jp.7210978
This article is cited by
-
Association of Ureaplasma urealyticum colonization with development of bronchopulmonary dysplasia: A systemic review and meta-analysis
Journal of Huazhong University of Science and Technology [Medical Sciences] (2014)