Abstract
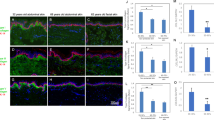
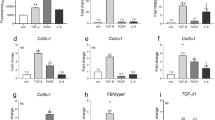
Organ cultures of human skin were incubated for 8 days under growth factor-free conditions or exposed to 10 ng ml–1 of human recombinant epidermal growth factor (EGF) during the incubation period. Normal histological features were preserved in the absence of growth factor, while epithelial cells underwent a proliferative response and invaded the underlying stroma in the presence of exogenous EGF. The same concentrations of EGF that induced stromal invasion also resulted in up-regulation of matrix metalloproteinase-9 (MMP-9; 92-kD gelatinase B) in organ culture and keratinocyte monolayer culture, and expression of MMP-1 (interstitial collagenase) in organ culture and fibroblast monolayer culture. When skin organ cultures were exposed to a potent, irreversible EGF–receptor tyrosine kinase (EGF–RTK) antagonist along with EGF, abnormal histological features were reversed, and MMP-9 production was suppressed. In contrast, EGF-RKT antagonism had only a modest inhibitory effect on MMP-1 production. Culture fluid from keratinocytes grown in monolayer culture stimulated fibroblast proliferation and MMP-1 elaboration. Treatment of fibroblasts with the same EGF–RTK antagonist inhibited keratinocyte-induced fibroblast proliferation but had only a modest inhibitory effect (approximately 20% inhibition) on MMP-1 production. In contrast, treatment of dermal fibroblasts with Interleukin-1 Receptor Antagonist had no effect on keratinocyte-induced fibroblast growth but strongly inhibited MMP-1 production (greater than 70% inhibition). These data indicate that stromal invasion by epithelial cells in EGF-treated skin is associated with events occurring in both the epidermis and dermis. The direct effect of the exogenous growth factor appears to be primarily on the epidermis. Dermal events reflect, at least in part, a response to factors elaborated in the epidermis. © 2001 Cancer Research Campaign http://www.bjcancer.com
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Bauer EA, Gordon JM, Reddick ME and Eisen AZ (1977) Quantitation and immunochemical localization of human skin collagenase in basal cell carcinoma. J Invest Dermatol 69: 363–367
Boukamp P, Petrussevska RT, Breitkreutz D, Hornung J, Markham A and Fusenig NE (1988) Normal keratinization in a spontaneously immortalized aneuploid human keratinocyte cell line. J Cell Biol 106: 761–771
Boyce S and Ham R (1983) Normal human epidermal keratinocytes. In: Webber M and Sekely L (eds) In Vitro Models for Cancer Research, p 245–274 CRC Press: Boca Raton, FL
Brodland DG (1998) Basal Cell Carcinoma: Features associated with metastasis. In:Cutaneous Oncology: Pathophysiology, Diagnosis and Management, SJ Miller and ME Maloney (ed.). Blackwell Science, Inc.: Malden MA pp 657–663
Clark W (1991) Tumor progression and the nature of cancer. Brit J Cancer 64: 631–644
Elder JT, Tavakkol A, Klein SB, Zeigler ME, Wicha M and Voorhees JJ (1990) Protooncogene expression in normal and psoriatic skin. J Invest Dermatol 94: 19–25
Elder JT (1994) Transforming growth factor-alpha and related growth factors. In: Epidermal Growth Factors and Cytokines. T Luger and T Schwarz (editors), Marcel Dekker Inc, New York, 205–240
Fisher GJ, Wang Z-Q, Datta S, Varani J and Voorhees JJ (1997) Pathophysiology and retinoic acid prevention of sun-induced premature skin aging. New Eng J Med 337: 1419–1428
Fligiel SEG and Varani J (1993) In situ epithelial cell invasion in organ culture. Invasion & Metastasis 13: 225–233
Gibbs DF, Apel IJ, Warner RO, Weiss SJ, Johnson KJ and Varani J (1999) Characterization of matrix metalloproteinases produced by rat alveolar macrophages. Amer J Respir Cell Molecular Biol 20: 1136–1144
Goslen JB and Bauer EA (1986) Basal cell carcinoma and collagenase. J Dermatol Surg Oncol 12: 812–817
Gottlieb AB, Chang CK, Posnett ND, Fanelli B and Tam JP (1988) Detection of TGF-a in normal, malignant and hyper-proliferative human keratinocytes. J Exp Med 167: 670–675
Herouy Y (2001) Matrix metalloproteinases in skin pathology. Int J Molec Medicine 7: 3–12
Maas-Szabowski N, Shimotoyodome A and Fusenig NE (1999) Keratinocyte growth regulation in fibroblast cocultures via a double paracrine mechanism. J Cell Sci 112: 1843–1853
Maas-Szabowski N, Stark H-J and Fusenig NE (2000) Keratinocyte growth regulation in defined organotypic cultures through IL-1-induced keratinocyte growth factor expression in resting fibroblasts. J Invest Dermatol 114: 1075–1084
Mackay AR, Ballin M, Pelina M, Farina AR, Nason AM, Hartzler JL and Thorgeirsson UP (1992) Effect of phorbol ester and cytokines on matrix metalloproteinase and tissue inhibitor of metalloproteinase expression in tumor and normal cell lines. Invasion & Metastasis 12: 168–184
Ogiso Y, Oikawa T, Kondo N, Kuzumaki N, Sugihara T and Ohura T (1988) Expression of proto-oncogenes in normal and tumor tissue of human skin. J Invest Dermatol 90: 841–844
Pilcher BK, Dumin JA, Schwartz MJ, Mast BA, Schultz GS, Parks WC and Welgus HG (1999) Keratinocyte collagenase-1 expression requires an epidermal growth factor receptor autocrine mechanism. J Biol Chem 274: 10372–10381
Shingu M, Nagai Y, Isayama T, Naono T, Nobunaga M and Nagai Y (1993) The effects of cytokines on metalloproteinase inhibitors (TIMP) and collagenase production by human chondorcytes and TIMP production by synovial cells and endothelial cells. Clin Exp Immunol 94: 145–149
Smaill JB, Rewcastle GW, Loo JA, Greis KD, Chan OH, Reyner EL, Lipka E, Showalter HD, Vincent PW, Elliott WL and Denny WA (2000) Tyrosine kinase inhibitors. 17. Irreversible inhibitors of the epidermal growth factor receptor: 4-(phenylamino)quinazoline-and 4-(phenylamino)pyrido[3,2-d] pyrimidine-6-acrylamides bearing additional solubilizing functions. J Med Chem 43: 1380–1397
Stetler-Stevenson WG, Aznavoorian S and Liotta LA (1993) Tumor cell interactions with the extracellular matrix during invasion and metastassis. Ann Rev Cell Biol 9: 541–573
Stoll S, Garner W and Elder JT (1997) Heparin-binding ligands mediate autrocrine epidermal growth factor receptor activation in skin organ culture. J Clin Invest 100: 1271–1281
Stoscheck CM and King LE jr (1986) Functional and structural characteristics of EGF and its receptor and their relationship to transforming proteins. J Cell Biochem 31: 135–152
Varani J, Fligiel SEG, Schuger L, Perone P, Inman DR, Griffiths CEM and Voorhees JJ (1993) Effects of all-trans retinoic acid and Ca2+ on human skin in organ culture. Am J Pathology 142: 189–198
Varani J, Perone P, Griffith CEM, Inman DR, Fligiel SEG and Voorhees JJ (1994) All-trans-retinoic acid (RA) stimulates events in organ cultured human skin that underlie repair. J Clin Invest 94: 1747–1756
Varani J, Perone P, Inman DR, Burmeister W, Schollenberger SB, Sitrin RJ and Johnson KJ (1995) Human skin in organ culture: Elaboration of proteolytic enzymes under growth factor-free and growth factor-containing conditions. Amer J Pathol 146: 210–217
Varani J, Hattori Y, Chi Y, Schmidt T, Perone P, Zeigler ME, Fader DJ and Johnson TM (2000) Collagenolytic and gelatinolytic matrix metalloproteinases and their inhibitors in basal cell carcinoma of skin: Comparison with normal skin. Brit J Cancer 82: 657–665
Wan YS, Belt A, Wang ZQ, Voorhees JJ and Fisher G (2001) Transmodulation of epidermal growth factor receptor mediates IL-1 beta-induced MMP-1 expression in human keratinocytes. International J Molec Medicine 7: 329–334
Weedon D (1997). Skin Pathology, Churchill Livingstone: New York. pp 635–671
Wells A (1999) EGF Receptor. International J Biochem Cell Biol 31: 637–643
Werb Z (1997) ECM and cell surface proteolysis: regulating cellular ecology. Cell 91: 439–442
Zeigler ME, Krause S, Karmiol S and Varani J (1996a) Growth factor-induced epidermal invasion of the dermis in human skin organ culture: Dermal invasion correlated with epithelial cell motility. Invasion & Metastasis 16: 3–10
Zeigler ME, Dutcheshen NT, Gibbs DF and Varani J (1996b) Growth factor-induced epidermal invasion of the dermis in human skin organ culture: Expression and role of matrix metalloproteinases. Invasion & Metastasis 16: 11–18
Zeigler ME, Chi Y, Schmidt T and Varani J (1999) Role of ERK and JNK pathways in regulating cell motility and matrix metalloproteinase 9 production in growth factor-stimulated human epidermal keratinocytes. J Cell Physiol 180: 271–284
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Moon, S., Dame, M., Remick, D. et al. Induction of matrix metalloproteinase-1 (MMP-1) during epidermal invasion of the stroma in human skin organ culture: keratinocyte stimulation of fibroblast MMP-1 production. Br J Cancer 85, 1600–1605 (2001). https://doi.org/10.1054/bjoc.2001.2122
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2001.2122
Keywords
This article is cited by
-
Expression of matrix metalloproteinase-1, -9, -13, and tissue inhibitor of metalloproteinases-1 in basal cell carcinomas of the eyelid
Graefe's Archive for Clinical and Experimental Ophthalmology (2012)
-
Essential role of microfibrillar-associated protein 4 in human cutaneous homeostasis and in its photoprotection
Scientific Reports (2011)
-
Keratinocyte-releasable factors increased the expression of MMP1 and MMP3 in co-cultured fibroblasts under both 2D and 3D culture conditions
Molecular and Cellular Biochemistry (2009)
-
The role of stratifin in fibroblast–keratinocyte interaction
Molecular and Cellular Biochemistry (2007)
-
Keratinocyte stimulation of matrix metalloproteinase-1 production and proliferation in fibroblasts: regulation through mitogen-activated protein kinase signalling events
British Journal of Cancer (2002)