Abstract
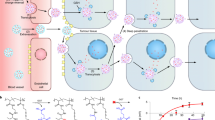
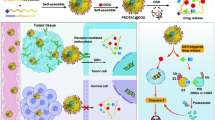
Polymer-directed enzyme prodrug therapy (PDEPT) is a novel two-step antitumour approach using a combination of a polymeric prodrug and polymer-enzyme conjugate to generate cytotoxic drug selectively at the tumour site. In this study the polymeric prodrug N-(2-hydroxypropyl) methacrylamide (HPMA) copolymer-Gly-Phe-Leu-Gly-doxorubicin conjugate PK1 (currently under Phase II clinical evaluation) was selected as the model prodrug, and HPMA copolymer-cathepsin B as a model for the activating enzyme conjugate. Following polymer conjugation (yield of 30–35%) HPMA copolymer-cathepsin B retained ~20–25% enzymatic activity in vitro. To investigate pharmacokinetics in vivo, 125I-labelled HPMA copolymer-cathepsin B was administered intravenously (i.v.) to B16F10 tumour-bearing mice. HPMA copolymer-cathespin B exhibited a longer plasma half-life (free cathepsin B t1/2α = 2.8 h; bound cathepsin B t1/2α = 3.2 h) and a 4.2-fold increase in tumour accumulation compared to the free enzyme. When PK1 (10 mg kg–1 dox-equiv.) was injected i.v. into C57 mice bearing subcutaneously (s.c.) palpable B16F10 tumours followed after 5 h by HPMA copolymer-cathepsin B there was a rapid increase in the rate of dox release within the tumour (3.6-fold increase in the AUC compared to that seen for PK1 alone). When PK1 and the PDEPT combination were used to treat established B16F10 melanoma tumour (single dose; 10 mg kg–1 dox-equiv.), the antitumour activity (T/C%) seen for the combination PDEPT was 168% compared to 152% seen for PK1 alone, and 144% for free dox. Also, the PDEPT combination showed activity against a COR-L23 xenograft whereas PK1 did not. PDEPT has certain advantages compared to ADEPT and GDEPT. The relatively short plasma residence time of the polymeric prodrug allows subsequent administration of polymer-enzyme without fear of prodrug activation in the circulation and polymer-enzyme conjugates have reduced immunogenicity. This study proves the concept of PDEPT and further optimisation is warranted. © 2001 Cancer Research Campaign http://www.bjcancer.com
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Ashihara Y, Kono T, Yamazaki S and Inada Y (1978) Modification of E. coli L-asparaginase with polyethylene glycol: disappearance of binding ability to anti-asparaginase serum. Biochem Biophys Res Commun 83: 385–391
Bagshawe KD (1987) Antibody directed enzymes revive anti-cancer prodrugs concept. Br J Cancer 56: 531–532
Bagshawe KD, Sharma SK, Springer CJ and Antoniw P (1995) Antibody directed enzyme prodrug therapy (ADEPT): a pilot scale clinical trial. Tumour Target 1: 17–29
Baillie-Johnson H, Twentyman PR, Fox NE, Walls GA, Workman P, Watson JV, Johnson N, Reeve JG and Bleehen NM (1985) Establishment and characterisation of cell lines from patients with lung cancer (predominantly small cell carcinoma). Br J Cancer 52: 495–504
Brocchini S and Duncan R (1999) Polymer drug conjugates: drug release from pendent linkers. In: Encyclopaedia of Controlled Release (Ed. E Mathiovitz), 786–816 Wiley: New York
Connors TA (1995) The choice of prodrugs for gene directed enzyme prodrug therapy of cancer. Gene Therapy 2: 702–709
Connors TA (1996) Is there a future for cancer chemotherapy?. Ann Oncol 7: 445–452
Delgado C, Francis GE and Fisher D (1992) The uses and properties of PEG-linked proteins. Crit Rev Therap Drug Carrier Sys 9: 249–304
Dreyer G and Ray W (1910) III The blood volume of mammals as determined by experiments upon rabbits, guinea-pigs and mice and its relationship to body weight and surface area expressed in a formula. Philosophical Transactions of The Royal Society for Science London, Section B 201: 133–137
Duncan R (1992) Drug-polymer conjugates: potential for improved chemotherapy. Anticancer drugs 3: 175–210
Duncan R (1999) Polymer conjugates for tumour targeting and intracytoplasmic delivery. The EPR effect as a common gateway?. Pharm Sci and Technol Today 2: 441–449
Duncan R, Cable HC, Lloyd JB, Rejmanova P and Kopecek J (1984) Polymers containing enzymatically degradable bonds, 7. Design of oligopeptide side chain in poly N-(2-hydroxypropyl)methacrylamide copolymers to promote efficient degradation by lysosomal enzymes. Makromol Chem 184: 1997–2008
Duncan R, Dimitrijevic S and Evagorou EG (1996) The role of polymer conjugates in the treatment and diagnosis of cancer. S.T.P. Pharma Sci 6: 237–263
Dvorak HF, Nagy JA, Dvorak JT and Dvorak AM (1988) Identification and characterisation of the blood vessels of solid tumours that are leaky to circulating macromolecules. Am J Pathol 133: 95–109
Flanagan PA, Rihova B, Subr V, Kopecek J and Duncan R (1990) Immunogenicity of protein-N-(2-hydroxypropyl)methacrylamide copolymer conjugates measured in A/J and B10 mice. J Bioact Compat Polymers 5: 151–166
Ferry DR, Seymour LW, Anderson D, Hesselwood S, Julyan P, Poyner R, Guest P, Doran J and Kerr DJ (1999) Phase I trial of liver targeted HPMA copolymer doxorubicin PK2, pharmacokinetics, spect imaging of123I-PK2 and activity in hepatoma. Proceedings of the American Society for Clinical Oncology 21: 4–13
Francis GE, Delgado C and Fisher D (1992) PEG-modified proteins, In: Stability of proteins pharmaceuticals (Part B): In vivo pathways of degradation and strategies for protein stabilisation, TJ Ahem, MC Manning (Eds.), New York, Plenum Press, 235–263
Ho DH, Brown NS, Yen A, Holmes R, Keating M, Abuchowski A, Newman RA and Krakoff IH (1986) Clinical pharmacology of polyethylene glycol-L-asparaginase. Drug Metabol Dispos 14: 349–352
Matsumura Y and Maeda H (1986) A new concept for macromolecular therapeutics in cancer chemotherapy: mechanism of tumour tropic accumulation of proteins and antitumour agent SMANCS. Cancer Res 46: 6387–6392
McNeish IA, Searle PF, Young LS and Kerr DJ (1997) Gene directed enzyme prodrug therapy for cancer. Adv Drug Del Rev 26: 173–184
Melton RG, Knox RJ and Connors TA (1996) Antibody-Directed Enzyme Prodrug Therapy (ADEPT). Drugs of The Future 21: 167–181
Muggia FM (1999) Doxorubicin-polymer conjugates: further demonstration of the concept of enhanced permeability and retention. Clin Cancer Res 5: 7–8
Pendri A, Conover CD and Greenvald RB (1998) Antitumour activity of paclitaxel-2-glycinate conjugated to poly(ethylene glycol): a water soluble prodrug. Anti-Cancer Drug Design 13: 387–395
Ram Z, Culver KW, Walbridge S, Blaese RM and Oldfield EH (1993) In situ retroviral-mediated gene transfer for the treatment of brain tumours in rats. Cancer Res 53: 83–88
Rihova B, Bilej M, Vetvicka V, Ulbrich K, Strohalm J, Kopecek J and Duncan R (1989) Biocompatibility of N-(2-hydroxypropyl)methacrylamide copolymers containing adriamycin. Immunogenicity, effect of haematopoietic stem cells in bone marrow in vivo and effect on mouse splenocytes and human peripheral blood lymphocytes in vitro. Biomaterials 10: 335–342
Satchi R and Duncan R (1998) PDEPT: Polymer Directed Enzyme Prodrug Therapy- in vitro and. in vivo 10: 83
Seymour LW, Ulbrich K, Strohalm J, Kopecek J and Duncan R (1990) The pharmacokinetics of polymer-bound adriamycin. Biochem Pharmacol 39: 1125–1131
Seymour LW, Ulbrich K, Styger PS, Brereton M, Subr V, Strohalm J and Duncan R (1994) Tumour tropism and anti-cancer efficacy of polymer-based doxorubicin prodrugs in the treatment of subcutaneous murine B16F10 melanoma. Br J Cancer 70: 636–641
Sharma SK, Bagshawe KD, Melton RG and Sherwood RF (1992) Human immune response to monoclonal antibody-enzyme conjugates in ADEPT pilot clinical trial. Cell Biophys 21: 109–120
Skinner A, Tutton PJM and O'Brien PE (1990) Microvascular architecture of experimental colon tumours in the rat. Cancer Res 50: 2411–2417
Springer CJ and Niculesco-Duvaz I (1996) Gene-directed enzyme prodrug therapy (GDEPT): choice of prodrugs. Adv Drug Del Rev 22: 351–364
Springer CJ, Bagshawe KD, Sharma SK, Searle F, Boden JA, Antoniw P, Burke PJ, Rogers GT, Sherwood RF and Melton RG (1991) Ablation of human choriocarcinoma xenografts in nude mice by antibody-directed enzyme prodrug therapy (ADEPT) with three novel compounds. Eur J Cancer 27: 1361–1366
Svensson PH, Wallace MP and Senter P (1994) Synthesis and characterisation of monoclonal antibody-β-lactamase conjugates. Bioconjugate Chem 5: 262–267
Ten Bokkel Hunink WW, Terwogt JM, Dubbelman R, Valkenet L, Zurlo MG, Schellens JHM and Beijnen JH (1998) Phase I and pharmacokinetics study of PNU 166945, a polymer formulated paclitaxel. 3rd Int Symp Polymer Therap 12,
Ulbrich K, Strohalm J, Subr V, Plocova D, Duncan R and Rihova B (1996) Polymeric conjugates of drugs and antibodies for site-specific drug delivery. Macromol Symp 103: 177–192
United Kingdom Co-ordinating Committee on Cancer Research (UKCCR) guidelines for the welfare of animals in experimental neoplasia (Second Edition) (1998). Br J Cancer 77: 1–10
Vasey PA, Kaye SB, Morrison R, Twelves C, Wilson P, Duncan R, Thomson AH, Murray LS, Hilditch TE, Murray T, Burtles S, Fraier D, Frigerio E and Cassidy J (1999) Phase I clinical and pharmacokinetic study of PK1 [N-(2-hydroxypropyl) methacrylamide copolymer doxorubicin]: first member of a new class of chemotherapeutic agents-drug-polymer conjugates. Clin Cancer Res 5: 83–94
Wedge SR (1990) Mechanism of action of action of polymer anthracyclines: potential to overcome multidrug resistance. Ph.D. Thesis, Keele University 49–64
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Satchi, R., Connors, T. & Duncan, R. PDEPT: polymer-directed enzyme prodrug therapy. Br J Cancer 85, 1070–1076 (2001). https://doi.org/10.1054/bjoc.2001.2026
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2001.2026
Keywords
This article is cited by
-
Cathepsin B-responsive prodrugs for cancer-targeted therapy: Recent advances and progress for clinical translation
Nano Research (2022)
-
Design of a VLP-nanovehicle for CYP450 enzymatic activity delivery
Journal of Nanobiotechnology (2015)
-
Validation of tumour models for use in anticancer nanomedicine evaluation: the EPR effect and cathepsin B-mediated drug release rate
Cancer Chemotherapy and Pharmacology (2013)