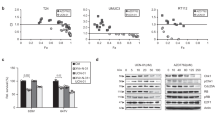
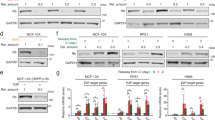
Abstract
The retinoblastoma gene, RB, participates in the regulation of the G1/S-phase transition and in p53-mediated apoptosis. We have previously reported that stably transfected RB functions as a growth and tumour suppressor in HTB9 human bladder carcinoma cells, which carry a mutation of the p53 gene at codon 280 and lack RB expression. To elucidate the potential role of RB in the regulation of p53-mediated apoptosis, we transfected a wt p53 expression plasmid under the control of the human cytomegalovirus promoter into parental and RB-transfected HTB9 cells. The p53+/RB– cells were susceptible to apoptosis under various experimental conditions: 1) incubation in serum-free culture for 72 h, 2) short-term (6 h) or long-term (48 h) exposure to etoposide, and 3) culturing in soft agar. In contrast, p53+/RB+ cells were significantly resistant to apoptosis under similar conditions and exhibited efficient growth arrest, as measured by laser scanning cytometry. Tumorigenicity in nude mice of parental HTB9 cells was lost by exogenous expression of wt p53. Likewise, none of mice injected subcutaneously with either p53–/RB+ or p53+/RB+ cells developed tumours, indicating that RB allows suppression of tumorigenesis, regardless of p53 status. These results suggest that the growth-inhibitory function of RB may overcome the ability of wt p53 to induce apoptosis. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Almasan A, Yin Y, Kelly RE, Lee EY-HP, Bradley A, Li W, Bertino JR and Wahl GM (1995) Deficiency of retinoblastoma protein leads to inappropriate S-phase entry, activation of E2F-responsive genes, and apoptosis. Proc Natl Acad Sci USA 92: 5436–5440
Bartek J, Bartkova J and Lukas J (1997) The retinoblastoma protein pathway in cell cycle control and cancer. Exp Cell Res 237: 1–6
Chomczynski P and Sacchi N (1987) Single-step method of RNA isolation by acid guanidium thiocyanate-phenol-chloroform extraction. Anal Biochem 162: 156–159
Clarke AR, Purdie CA, Harrison DJ, Morris RG, Bird CC, Hooper ML and Wyllie AH (1993) Thymocyte apoptosis induced by p53-dependent and independent pathways. Nature 362: 849–852
DeGregori J, Leone G, Miron A, Jakoi L and Nevins JR (1997) Distinct roles for E2F proteins in cell growth control and apoptosis. Proc Natl Acad Sci USA 94: 7245–7250
Del Bino G and Darzynkiewicz Z (1991) Camptothecin, tenioside, or 4′-(9-acridinylamino)-3-methanesulfon-m-anisidide, but not mitoxantrone or doxorubicin, induces degradation of nuclear DNA in the S phase of HL-60 cells. Cancer Res 51: 1165–1169
Haapajärvi T, Kivinen L, Pitkänen K and Laiho M (1995) Cell cycle-dependent effects of UV-radiation on p53 expression and retinoblastoma protein phosphorylation. Oncogene 11: 151–159
Hartwell LH and Kastan MB (1994) Cell cycle control and cancer. Science 266: 1821–1828
Haupt Y, Rowan S and Oren M (1995) p53-mediated apoptosis in HeLa cells can be overcome by excess pRB. Oncogene 10: 1563–1571
Kamentsky LA and Kamentsky LD (1991) Microscope-based multiparameter laser scanning cytometer yielding data comparable to flow cytometry data. Cytometry 12: 381–387
Kowalik TF, DeGregori J, Schwarz JK and Nevins JR (1995) E2F-1 overexpression in quiescent fibroblasts leads to induction of cellular DNA synthesis and apoptosis. J Virol 69: 2491–2500
Kowalik TF, DeGregori J, Leone G, Jakoi L and Nevins JR (1998) E2F-1-specific induction of apoptosis and p53 accumulation, which is blocked by MDM2. Cell Growth Differ 9: 113–118
Macleod K, Hu Y and Jacks T (1996) Loss of Rb activates both p53-dependent and independent cell death pathways in the developing mouse nervous system. EMBO J 15: 6178–6188
Morgenbesser SD, Williams BO, Jacks T and DePinho RA (1994) p53-dependent apoptosis produced by Rb-deficiency in the developing mouse lens. Nature 371: 72–74
National Institutes of Health (1996). Guide for the Care and Use of Laboratory Animals, http://grants.nih.gov/grants/policy
Pan H, Yin C, Dyson NJ, Harlow E, Yamasaki L and Van Dyke T (1998) Key roles for E2F1 in signaling p53-dependent apoptosis and in cell division within developing tumours. Mol Cell 2: 283–292
Pomerantz J, Schreiber-Agus N, Liegeois NJ, Silverman A, Alland L, Chin L, Potes J, Chen K, Orlow I, Lee HW, Cordon-Cardo C and DePinho RA (1998) The Ink4a tumour suppressor gene product, p19Arf, interacts with MDM2 and neutralizes MDM2’s inhibition of p53. Cell 92: 713–723
Qin X-Q, Livingston DM, Kaelin WG and Adams PD (1994) Deregulated transcription factor E2F-1 expression leads to S-phase entry and p53-mediated apoptosis. Proc Natl Acad Sci USA 91: 10918–10922
Resnicoff M, Burgand JL, Rotman HL, Abraham D and Baserga R (1995) Correlation between apoptosis, tumorigenesis, and levels of insulin-like growth factor I receptors. Cancer Res 55: 3739–3741
Shaw P, Bovey R, Tardy S, Sahli R, Sordat B and Costa J (1992) Induction of apoptosis by wild-type p53 in a human colon tumour-derived cell line. Proc Natl Acad Sci USA 89: 4495–4499
Singleton JR, Randolph AE and Feldman EL (1996) Insulin-like growth factor I receptor prevents apoptosis and enhances neuroblastoma tumourigenesis. Cancer Res 55: 3739–3741
Takahashi R, Hashimoto T, Xu H-J, Hu S-X, Matsui T, Miki T, Bigo-Marshall H, Aaronson SA and Benedict WF (1991) The retinoblastoma gene functions as a growth and tumour suppressor in human bladder carcinoma cells. Proc Natl Acad Sci USA 88: 5257–5261
Tamura T, Aoyama N, Saya H, Haga H, Futami S, Miyamoto M, Koh T, Ariyasu T, Tachi M, Kasuga M and Takahashi R (1995) Induction of Fas-mediated apoptosis in p53-transfected human colon carcinoma cells. Oncogene 11: 1939–1946
Thomas PS (1980) Hybridization of denatured RNA and small DNA fragments transferred to nitrocellulose. Proc Natl Acad Sci USA 77: 5201–5205
Tsai KY, Hu Y, Macleod KF, Crowley D, Yamasaki L and Jacks T (1998) Mutation of E2F-1 suppresses apoptosis and inappropriate S phase entry and extends survival of Rb-deficient mouse embryos. Mol Cell 2: 293–304
Waldman T, Lengauer C, Kinzler KW and Vogelstein B (1996) Uncoupling of S phase and mitosis induced by anticancer agents in cells lacking p21. Nature 381: 713–716
White E, Cipriani R, Sabbatini P and Denton A (1991) Adenovirus E1B 19-kilodalton protein overcomes the cytotoxicity of E1A proteins. J Virol 65: 2968–2978
Wyllie FS, Haughton MF, Bond JA, Rowson JM, Jones CJ and Wynford-Thomas D (1996) S phase cell-cycle arrest following DNA damage is independent of the p53/p21WAF1 signalling pathway. Oncogene 12: 1077–1082
Yamasaki L (1998) Growth regulation by the E2F and DP transcription factor families. InCell Cycle Control, Pagano M (ed) pp 199–227, Springer–Verlag: Berlin
Yamasaki L, Bronson R, Williams BO, Dyson NJ, Harlow E and Jacks T (1998) Loss of E2F-1 reduces tumorigenesis and extends the lifespan of Rb1(+/–) mice. Nature Genet 18: 360–364
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Shinohara, H., Zhou, J., Yoshikawa, K. et al. Retinoblastoma protein-initiated cellular growth arrest overcomes the ability of cotransfected wild-type p53 to induce apoptosis. Br J Cancer 83, 1039–1046 (2000). https://doi.org/10.1054/bjoc.2000.1411
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1411