Abstract
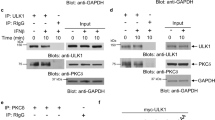
Interferon (IFN)-α affects the growth, differentiation and function of various cell types by transducing regulatory signals through the Janus tyrosine kinase/signal transducers of activation and transcription (Jak/STAT) pathway. The signalling pathways employing the mitogen-activated ERK-activating kinase (MEK) and the extracellular-regulated kinase (ERK) are critical in growth factors signalling. Engagement of the receptors, and subsequent stimulation of Ras and Raf, initiates a phosphorylative cascade leading to activation of several proteins among which MEK and ERK play a central role in routing signals critical in controlling cell development, activation and proliferation. We demonstrate here that 24–48 h following treatment of transformed T- and monocytoid cell lines with recombinant human IFN-α2b both the phosphorylation and activity of MEK1 and its substrates ERK1/2 were reduced. In contrast, the activities of the upstream molecules Ras and Raf -1 were not affected. No effect on MEK/ERK activity was observed upon short-term exposure (1–30 min) to IFN. The anti-proliferative effect of IFN-α was increased by the addition in the culture medium of a specific inhibitor of MEK, namely PD98059. In conclusion, our results indicate that IFN-α regulates the activity of the MEK/ERK pathway and consequently modulates cellular proliferation through a Ras/Raf-independent mechanism. Targeting the MEK/ERK pathway may strengthen the IFN-mediated anti-cancer effect. © 2000 Cancer Research Campaign
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Ahn NG, Campbell JS, Seger R, Jensenn AM, Graves LM and Krebs EG (1993) Metabolic labeling of mitogen-activated protein kinase kinase in A431 cells demonstrates phosphorylation on serine and threonine residues. Proc Natl Acad Sci USA 90: 5143–5147
Anderson NG, Maller JL, Tonks NK and Sturgill TW (1990) Requirement for integration of signals from two distinct phosphorylation pathways for activation of MAP kinase. Nature 143: 651–653
Arora T, Floyd-Smith G, Espy MJ and Jelinek DF (1999) Dissociation between IFN-alpha-induced anti-viral and growth signaling pathways. J Immunol 162: 3289–3297
Bromberg JF, Horvath CM, Wen Z, Schreiber RD and Darnell JE Jr (1996) Transcriptionally active Stat1 is required for the antiproliferative effects of both interferon alpha and interferon gamma. Proc Natl Acad Sci USA 93: 7673–7678
Burgering BM, Medema RH, Maassen JA, van de Wetering ML, van der Eb AJ, McCormick F and Bos JL (1991) Insulin stimulation of gene expression mediated by p21ras activation. EMBO J 10: 1103–1109
Cowley S, Paterson H, Kemp P and Marshall CJ (1994) Activation of MAP kinase kinase is necessary and sufficient for PC12 differentiation and for transformation of NIH 3T3 cells. Cell 77: 841–852
Darnell, Jr JE (1997) STATs and gene regulation. Science 277: 1630–1635
David M, Petricoin E 3rd, Benjamin C, Pine R, Weber MJ and Larner AC (1995) Requirement for MAP kinase (ERK2) activity in interferon alpha- and interferon beta-stimulated gene expression through STAT proteins. Science 269: 1721–1723
Dent P, Haser W, Haystead TA, Vincent LA, Roberts TM and Sturgill TW (1992) Activation of mitogen-activated protein kinase kinase by v-Raf in NIH 3T3 cells and in vitro. Science 257: 1404–1407
Gardner AM, Vaillancourt RR, Lange-Carter CA and Johnson GL (1994) MEK-1 phosphorylation by MEK kinase, Raf and mitogen-activated protein kinase: analysis of phosphopeptides and regulation of activity. Mol Biol Cell 5: 193–201
Gomez N and Cohen P (1991) Dissection of the protein kinase cascade by which nerve growth factor activates MAP kinases. Nature 353: 170–173
Grimley PM, Fang H, Rui H, Petricoin EF 3rd, Ray S, Dong F, Fields KH, Hu R, Zoon KC, Audet S and Beeler J (1998) Prolonged STAT1 activation related to the growth arrest of malignant lymphoma cells by interferon-alpha. Blood 91: 3017–3027
Haystead CM, Gregory P, Shirazi A, Fadden P, Mosse C, Dent P and Haystead TA (1994) Insulin activates a novel adipocyte mitogen-activated protein kinase kinase kinase that shows rapid phasic kinetics and is distinct from c-Raf. J Biol Chem 269: 12804–12808
Herberman RB (1997) Effect of α-interferon on immune function. Semin Oncol 24: S9-78–S9-80
Howe LR, Leevers SJ, Gomez N, Nakielny S, Cohen P and Marshall CJ (1992) Activation of the MAP kinase pathway by the protein kinase Raf. Cell 71: 335–342
Itoh T, Kaibuchi K, Masuda T, Yamamoto T, Matsuura Y, Maeda A, Shimizu K and Takai Y (1993) A protein factor for Ras p21-dependent activation of mitogen-activated protein (MAP) kinase through MAP kinase kinase. Proc Natl Acad Sci USA 90: 975–979
Khokhlatchev AV, Canagarajah B, Wilsbacher J, Robinson M, Atkinson M, Goldsmith E and Cobb MH (1998) Phosphorylation of the MAP kinase ERK2 promotes its homodimerization and nuclear translocation. Cell 93: 605–615
Kirk CJ and Miller RA (1998) Analysis of Raf-1 activation in response to TCR activation and costimulation in murine T-lymphocytes: effect of age. Cell Immunol 190: 33–42
Kyriakis JM, App H, Zhang XF, Banerjee P, Brautigan DL, Rapp UR and Avruch J (1992) Raf-1 activates MAP kinase-kinase. Nature 358: 417–421
Lund TC, Medveczky MM and Medveczky PG (1999) Interferon-alpha induction of STATs1, -3 DNA binding and growth arrest is independent of Lck and active mitogen-activated kinase in T cells. Cell Immunol 192: 133–139
Madhani HD and Fink GR (1998) The riddle of MAP kinase signaling specificity. Trends Genet 14: 151–155
Matsuda S, Gotoh Y and Nishida E (1993) Phosphorylation of Xenopus mitogen-activated protein (MAP) kinase kinase by MAP kinase kinase kinase and MAP kinase. J Biol Chem 268: 3277–3281
Pages G, Lenormand P, L'Allemain G, Chambard JC, Meloche S and Pouyssegur J (1993) Mitogen-activated protein kinases p42mapk and p44mapk are required for fibroblast proliferation. Proc Natl Acad Sci USA 90: 8319–8323
Papin C, Denouel-Galy A, Laugier D, Calothy G and Eychene A (1998) Modulation of kinase activity and oncogenic properties by alternative splicing reveals a novel regulatory mechanism for B-Raf. J Biol Chem 273: 24939–24947
Pawson T and Scott JD (1997) Signaling through scaffold, anchoring, and adaptor proteins. Science 278: 2075–2080
Pfeffer LM, Dinarello CA, Herberman RB, Williams BR, Borden EC, Bordens R, Walter MR, Nagabhushan TL, Trotta PP and Pestka S (1998) Biological properties of recombinant α-interferon: 40th Anniversary of the discovery of Interferons. Cancer Res 58: 2489–2499
Posada J, Yew N, Ahn NG, Vande Woude GF and Cooper JA (1993) Mos stimulates MAP kinase in Xenopus oocytes and activates a MAP kinase kinase in vitro. Mol Cell Biol 13: 2546–2553
Pumiglia KM and Decker SJ (1997) Cell cycle arrest mediated by the MEK/mitogen-activated protein kinase pathway. Proc Natl Acad Sci USA 94: 448–452
Resnitzky D, Tiefenbrun N, Berissi H and Kimchi A (1992) Interferons and interleukin 6 suppress phosphorylation of the retinoblastoma protein growth-sensitive hematopoietic cells. Proc Natl Acad Sci USA 89: 402–406
Rubinstein S, Familletti PC and Pestka S (1981) Convenient assay for interferons. J Virol 37: 755–758
Sakatsume M, Stancato LF, David M, Silvennoinen O, Saharinen P, Pierce J, Larner AC and Finbloom DS (1998) Interferon gamma activation of Raf-1 is Jak1-dependent and p21 ras-independent. J Biol Chem 273: 3021–3026
Schaeffer HJ, Catling AD, Eblen ST, Collier LS, Krauss A and Weber MJ (1998) MP1: a MEK binding partner that enhances enzymatic activation of the MAP kinase cascade. Science 281: 1668–1671
Sebolt-Leopold JS, Dudley DT, Herrera R, Van Becelaere K, Wiland A, Gowan RC, Tecle H, Barret SD, Bridges A, Przybranowski,, Leopold WR and Saltiel AR (1999) Blockade of the MAP kinase pathway suppresses growth of colon tumours in vitro. Nat Med 5: 810–816
Seger R and Krebs EG (1995) The MAPK signaling cascade. FASEB J 9: 726–735
Stancato LF, Sakatsume M, David M, Dent P, Dong F, Petricoin EF, Krolewski JJ, Silvennoinen O, Saharinen P, Pierce J, Marshall CJ, Sturgill T, Finbloom DS and Larner AC (1997) Beta interferon and oncostatin M activate Raf-1 and mitogen-activated protein kinase through a JAK1-dependent pathway. Mol Cell Biol 17: 3833–3840
Sun WH, Pabon C, Alsayed Y, Huang PP, Jandeska S, Uddin S, Platanias LC and Rosen ST (1998) Interferon-alpha resistance in a cutaneous T-cell lymphoma cell line is associated with lack of STAT1 expression. Blood 91: 570–576
Takenaka K, Moriguchi T and Nishida E (1998) Activation of the protein kinase p38 in the spindle assembly checkpoint and mitotic arrest. Science 280: 599–602
Uddin S, Fish EN, Sher DA, Gardziola C, White MF and Platanias LC (1997) Activation of the phosphatidylinositol 3-kinase serine kinase by IFN-alpha. J Immunol 158: 2390–2397
Whitmarsh AJ and Davis RJ (1998) Structural organization of MAP-kinase signaling modules by scaffold proteins in yeast and mammals. Trends Biochem Sci 23: 481–485
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Romerio, F., Riva, A. & Zella, D. Interferon-α 2b reduces phosphorylation and activity of MEK and ERK through a Ras/Raf-independent mechanism. Br J Cancer 83, 532–538 (2000). https://doi.org/10.1054/bjoc.2000.1263
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1263
Keywords
This article is cited by
-
EMT and Inflammation: Crossroads in HCC
Journal of Gastrointestinal Cancer (2023)
-
Direct and indirect effects of IFN-α2b in malignancy treatment: not only an archer but also an arrow
Biomarker Research (2022)
-
PML-II regulates ERK and AKT signal activation and IFNα-induced cell death
Cell Communication and Signaling (2021)