Summary
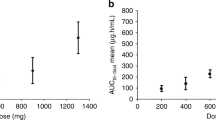
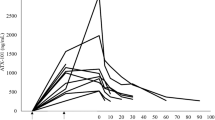
DACA, also known as XR5000, is an acridine derivative active against both topoisomerase I and II. In this phase I study, DACA was given as a 3-h intravenous infusion on 3 successive days, repeated every 3 weeks. A total of 41 patients were treated at 11 dose levels between 9 mg m–2 d–1 and the maximum tolerated dose of 800 mg m–2 day–1. The commonest, and dose-limiting, toxicity was pain in the infusion arm. One patient given DACA through a central venous catheter experienced chest pain with transient electrocardiogram changes, but no evidence of myocardial infarction. At the highest dose levels, several patients also experienced flushing, pain and paraesthesia around the mouth, eyes and nose and a feeling of agitation. Other side-effects, such as nausea and vomiting, myelosuppression, stomatitis and alopecia, were uncommon. There was one minor response but no objective responses. DACA pharmacokinetics were linear and did not differ between days 1 and 3. The pattern of toxicity seen with DACA is unusual and appears related to the mode of delivery. It is possible that higher doses of DACA could be administered using a different schedule of administration.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Abigerges, D., Armand, J-P, Chabot, G. G., Bruno, R., Bissery, M-C, Bayssas, M., Klink-Alaki, M., Clavel, M. & Catimel, G. (1996). Phase I and pharmacology study of intoplicine (RP 60475; NSC 645008), a novel topoisomerase I and II inhibitor, in cancer patients. Anticancer Drugs 7: 166–174.
Atwell, G. J., Rewcastle, G. W., Baguley, B. C. & Denny, W. A. (1987). Potential anti-tumour agents. 50. In vivo solid tumour activity of derivatives of N-[2-(dimethylamino)ethyl]acridine-4-carboxamide. J Med Chem 30: 664–669.
Baguley, B. C., Holdaway, K. M. & Fray, L. M. (1990). Design of DNA intercalators to overcome topoisomerase II-mediated multi-drug resistance. J Natl Cancer Inst 82: 398–402.
Baguley, B. C., Zhuang, L. & Marshall, E. (1995). Experimental solid tumour activity of N-[2-(dimethylamino)ethyl]acridine-4-carboxamide. Cancer Chemother Pharmacol 36: 224–248.
Evans, S. M. H., Young, D., Robertson, I. G. C. & Paxton, J. W. (1992). Intraperitoneal administration of the anti-tumour agent of N-[2-(dimethylamino) ethyl] acridine-4-carboxamide in the mouse: bioavailability, pharmacokinetics and toxicity after a single dose. Cancer Chemother Pharmacol 31: 32–36.
Finlay, G. J. & Baguley, B. C. (1989). Selectivity of N-[2-(dimethylamino) ethyl] acridine-4-carboxamide towards Lewis lung carcinoma and human tumour cell lines in vitro. Eur J Cancer Clin Oncol 25: 271–277.
Finlay, G. J., Marshall, E., Matthews, J. H. L., Paul, K. D. & Baguley, B. C. (1993). In vitro assessment of N-[2-(dimethylamino) ethyl] acridine-4-carboxamide, a DNA-intercalating antitumour drug with reduced sensitivity to multidrug resistance. Cancer Chemother Pharmacol 31: 401–406.
Gupta, R. S., Gupta, R., Eng, B., Lock, R. B., Ross, W. E., Hertzberg, R. P., Caranfa, M. J. & Johns, R. K. (1988). Camptothecin-resistant mutants of Chinese hamster ovary cells containing a resistant form of topoisomerase I. Cancer Res 48: 6404–6410.
Kano, Y., Suzuki, K., Akatsu, M., Suda, K., Inoue, Y., Yoshida, M., Sakamoto, S. & Miura, Y. (1992). Effects of CPT-11 in combination with other anti-cancer agents in culture. Int J Cancer 50: 604–610.
Karato, A., Saski, Y. & Shiraishi, J. et al (1993). Phase I study of CPT-11 and etoposide in patients with refractory solid tumours. J Clin Oncol 11: 2030–2035.
Kaufman, S. H. (1991). Antagonism between camptothecin and topoisomerase II-directed chemotherapeutic agents in a human leukaemia cell line. Cancer Res 51: 1129–1136.
Marshall, E. S., Finlay, G. J., Matthews, J. H. L., Shaw, J. H. F., Nixon, J. & Baguley, B. C. (1992). Microculture-based chemosensitivity testing: a feasibility study comparing freshly explanted human melanoma cells with human melanoma cell lines. J Natl Cancer Inst 84: 340–345.
Paxton, J. W., Young, D., Evans, S. M. H., Kestell, P., Robertson, I. G. C. & Cornford, E. M. (1992). Pharmacokinetics and toxicity of the anti-tumour agent N-[2-(dimethylamino) ethyl]acridine-4-carboxamide after i.v. administration in the mouse. Cancer Chemother Pharmacol 29: 379–384.
Paxton, J. W., Young, D., Evans, S. M. H., Robertson, I. G. C. & Kestell, P. (1993a). Tumour profile of N-[2-(dimethylamino) ethyl]acridine-4-carboxamide after intraperitoneal administration in the mouse. Cancer Chemother Pharmacol 32: 320–322.
Paxton, J. W., Young, D. & Robertson, I. G. C. (1993b). Pharmacokinetics of acridine-4-carboxamide in the rat, with extrapolation to humans. Cancer Chemother Pharmacol 32: 323–325.
Schneider, E., Darkin, S. J., Lawson, P. A., Ching, L-M, Ralph, R. K. & Baguley, B. C. (1988). Cell line selectivity and DNA breakage properties of the anti-tumour agent N-[2-(dimethylamino) ethyl]acridine-4-carboxamide: role of DNA topoisomerase II. Eur J Cancer Clin Oncol 24: 1783–1790.
Tewey, K. M., Chen, G. L., Nelson, E. M. & Liu, L. F. (1984). Intercalative antitumour drugs interfere with the breakage-reunion reaction of mammalian DNA topoisomerase II. J Biol Chem 259: 9182–9187.
Trask, D. K. & Muller, M. T. (1988). Stabalization of type I topoisomerase-DNA covalent complexes by actinomycin D. Proc Natl Acad Sci USA 85: 1417–1421.
Author information
Authors and Affiliations
Consortia
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Twelves, C., Gardner, C., Flavin, A. et al. Phase I and pharmacokinetic study of DACA (XR5000): a novel inhibitor of topoisomerase I and II. Br J Cancer 80, 1786–1791 (1999). https://doi.org/10.1038/sj.bjc.6690598
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690598
Keywords
This article is cited by
-
Plasma pharmacokinetic evaluation of cytotoxic agents radiolabelled with positron emitting radioisotopes
Cancer Chemotherapy and Pharmacology (2008)
-
The role of topoisomerases and RNA transcription in the action of the antitumour benzonaphthyridine derivative SN 28049
Cancer Chemotherapy and Pharmacology (2008)
-
Phase I and pharmacokinetic study of XR11576, an oral topoisomerase I and II inhibitor, administered on days 1–5 of a 3-weekly cycle in patients with advanced solid tumours
British Journal of Cancer (2004)