Summary
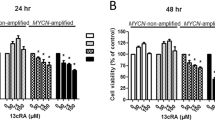
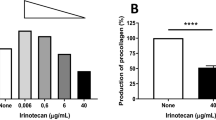
Uncontrolled proliferation and a defect of apoptosis constitute crucial elements in the development and progression of tumours. Among many other biological response modifiers known to influence these mechanisms, the efficacy of retinoids and interferons in the treatment of various malignant entities is currently matter of discussion. In the present study, we have investigated the effects of 9-cis-retinoic acid (9cRA), 13-cis-retinoic acid (13cRA), all-trans-retinoic acid (tRA) and interferon-α on proliferation and apoptosis of human soft tissue sarcoma (STS) cell lines HTB-82 (rhabdomyosarcoma), HTB-91 (fibrosarcoma), HTB-92 (liposarcoma), HTB-93 (synovial sarcoma) and HTB-94 (chondrosarcoma) in relation to p53 genotype as well as p53 expression. HTB-91, HTB-92 and HTB-94 STS cells exhibited mutant p53, whereas wild-type p53 was found in HTB-93 STS cells, and a normal p53 status in HTB-82 STS cells, carrying a silent point mutation only. Interferon-α, irrespective of p53 status, inhibited the proliferation of all five cell lines dose- and time-dependently. Similarly, 9cRA, 13cRA and tRA decreased the proliferation of HTB-82 and HTB-93 STS cells, whereas the proliferation of p53-mutated HTB-91, HTB-92 and HTB-94 STS cells remained unchanged. Furthermore, only 9cRA and tRA were capable of inducing apoptosis in HTB-82 and HTB-93 STS cells, whereas HTB-91, HTB-92 and HTB-94 STS cells did not undergo apoptosis under the influence of 9cRA or tRA. Retinoic acid receptor (RAR)-α and RAR-β mRNA were not detectable by Northern blot analysis in the five STS cell lines, whereas mRNA for the universal retinoic acid receptor, RAR-γ, was expressed in all STS cell lines indicating that retinoid resistance was not associated with a lack of RAR expression. Apoptosis was not induced by interferon-α or 13cRA in any of the five STS cell lines tested. Our results indicate that within the panel of tested STS cell lines, inhibition of proliferation and induction of apoptosis result from different mechanisms which differ in their dependence upon the presence of intact p53.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Aas, T., Borresen, A. L., Geisler, S., Smith-Sorensen, B., Johnson, H., Varhaug, J. E., Akslen, L. A. & Lonning, P. E. (1996). Specific p53 mutations are associated with de novo resistance to doxorubicin in breast cancer patients. Nat Med 2: 811–814.
Agarwal, M. L., Agarwal, A., Taylor, W. R. & Stark, G. R. (1995). p53 controls both the G2/M and the G1 cell cycle checkpoints and mediates reversible growth arrest in human fibroblasts. Proc Natl Acad Sci USA 92: 8493–8497.
Antman, K., Crowley, J., Balcerzak, S. P., Rivkin, S. E., Weiss, G. R., Elias, A., Natale, R. B., Cooper, R. M., Barlogie, B., Trump, D. L., Doroshow, J. H., Aisner, J., Pugh, R. P., Weiss, R. B., Cooper, B. A., Clamond, G. H. & Baker, L. H. (1993). An intergroup phase III randomized study of doxorubicin and dacarbacine with or without ifosfamide and mesna in advanced soft tissue and bone sarcomas. J Clin Oncol 11: 1276–1285.
Antman, K. H. (1997). Adjuvant therapy of sarcomas of soft tissue. Semin Oncol 24: 556–560.
Berggren Soderlund, M., Johannesson, G. & Fex, G. (1995). Expression of human all-trans-retinoic acid receptor beta and its ligand-binding domain in Escherichia coli. Biochem J 308: 353–359.
Bergh, J., Norberg, T., Sjogren, S., Lindgren, A. & Holmberg, L. (1995). Complete sequencing of the p53 gene provides prognostic information in breast cancer patients, particularly in relation to adjuvant systemic therapy and radiotherapy. Nat Med 1: 1029–1034.
Borden, E. C., Amato, D. A., Edmonson, J. H., Ritch, P. S. & Shiraki, M. (1990). Randomized comparison of doxorubicin and vindesine to doxorubicin for patients with metastatic soft-tissue sarcomas. Cancer 66: 862–867.
Bracey, T. S., Miller, J. C., Preece, A. & Paraskeva, C. (1995). γ-Radiation-induced apoptosis in human colorectal adenoma and carcinoma cell lines can occur in the absence of wild-type p53. Oncogene 10: 2391–2396.
Brodowicz, T., Kotz, R., Wiltschke, C., Zielinski, C. C. & Ritschl, P. (1997). Re: isolated limb perfusion with high-dose tumor necrosis factor-alpha in combination with interferon-γ and melphalan for non-resectable extremity soft tissue sarcomas: a multicenter trial. J Clin Oncol 15: 2174–2175.
Canman, C. E., Gilmer, T. M., Coutts, S. B. & Kastan, M. B. (1995). Growth factor modulation of p53-mediated growth arrest versus apoptosis. Gene Dev 9: 600–611.
Crouch, G. D. & Helman, L. J. (1991). All-trans-retinoic acid inhibits the growth of human rhabdomyosarcoma cell lines. Cancer Res 51: 4882–4887.
Delia, D., Aiello, A., Lombardi, L., Pelicci, P. G., Grignani, F. r., Grignani, F. a., Formelli, F., Menard, S., Costa, A., Veronesi, U. & Pierotti, M. A. (1993). N-(4-hydroxyphenyl) retinamide induces apoptosis of malignant hemopoietic cell lines including those unresponsive to retinoic acid. Cancer Res 53: 6036–6041.
Di Leonardo, A., Linke, S. P., Clarkin, K. & Wahl, G. M. (1994). DNA damage triggers a prolonged p53-dependent G1 arrest and long-term induction of Cip1 in normal human fibroblasts. Genes Dev 8: 2540–2551.
Dmitrovsky, E. (1997). N-(4-hydroxyphenyl)retinamide activation of a distinct pathway signaling apoptosis. J Natl Cancer Inst 89: 1179–1181.
Elledge, R. M., Gray, R., Mansour, E., Yu, Y., Clark, G. M. & Ravdin, P. (1995). Accumulation of p53 protein as a possible predictor of response to adjuvant combination chemotherapy with cyclophosphamide, methotrexate, fluorouracil, and prednisone for breast cancer. J Natl Cancer Inst 87: 1254–1256.
Engers, R., van Roy, F., Heymer, T., Ramp, U., Moll, R., Dienst, M., Friebe, U., Pohl, A., Gabbert, H. E. & Gerharz, C. D. (1996). Growth inhibition in clonal subpopulations of a human epithelioid sarcoma cell line by retinoic acid and tumour necrosis factor alpha. Br J Cancer 73: 491–498.
Fisher, G. J., Datta, S. C. & Voorhees, J. J. (1998). Retinoic acid receptor-γ in human epidermis preferentially traps all-trans retinoic acid as its ligand rather than 9-cis retinoic acid. J Invest Dermatol 110: 297–300.
Gabbert, H. E., Gerharz, C. D., Biesalski, H. K., Engers, R. & Luley, C. (1988). Terminal differentiation and growth inhibition of a rat rhabdomyosarcoma cell line (BA-HAN-1C) in vitro after exposure to retinoic acid. Cancer Res 48: 5264–5269.
Gherlinzoni, F., Bacci, F., Picci, P., Capanna, R., Calderoni, P., Lorenzi, E. G., Bernini, M., Emiliani, E., Barbieri, E., Normand, A. & Campanacci, M. (1986). A randomized trial for the treatment of high-grade soft-tissue sarcomas of the extremities: preliminary observations. J Clin Oncol 4: 552–558.
Giandomenico, V., Lancillotti, F., Fiorucci, G., Percario, Z. A., Rivabene, R., Malorni, W., Affabris, E. & Romeo, G. (1997). Retinoic acid and IFN inhibition of cell proliferation is associated with apoptosis in squamous carcinoma cell lines: role of IRF-1 and TGase II-dependent pathways. Cell Growth Differ 8: 91–100.
Harant, H., Lindley, I., Uthman, A., Ballaun, C., Kruptiza, G., Grunt, T., Huber, H. & Dittrich, C. (1995). Regulation of interleukin-8 gene expression by all-trans retinoic acid. Biochem Biophys Res Commun 210: 898–906.
Harvey, M., Sands, A. T., Weiss, R. S., Hegi, M. E., Wiseman, R. W., Pantazis, P., Giovanella, B. C., Tainsky, M. A., Bradley, A. & Donehower, L. A. (1993). In vitro growth characteristics of embryo fibroblasts isolated from p53-deficient mice. Oncogene 8: 2457–2467.
Hockenbery, D., Nunez, G., Milliman, C., Schreiber, R. D. & Korsmeyer, S. J. (1990). Bcl-2 is an inner mitochondrial membrane protein that blocks programmed cell death. Nature 348: 334–336.
Kastan, M. B., Onyekwere, O., Sidransky, D., Vogelstein, B. & Craig, R. W. (1991). Participation of p53 in the cellular response to DNA damage. Cancer Res 51: 6304–6311.
Kerr, J. F. R., Wyllie, A. H. & Currie, A. R. (1972). Apoptosis: a basic biological phenomenon with wide ranging implications in tissue kinetics. Br J Cancer 26: 239–257.
Lehman, T. A., Bennett, W. P., Metcalf, R. A., Welsh, J. A., Ecker, J., Modali, R. V., Ullrich, S., Romano, J. W., Appella, E., Testa, J. R., Gerwin, B. I. & Harris, C. C. (1991). P53 mutations, ras mutations, and p53-heat shock 70 protein complexes in human lung carcinoma cell lines. Cancer Res 51: 4090–4096.
Lotan, R. (1995). Retinoids and apoptosis: implications for cancer chemoprevention and therapy. J Natl Cancer Inst 87: 1655–1657.
Lovat, P. E., Irving, H., Annicchiarico-Petruzzelli, M., Bernassola, F., Malcolm, A. J., Pearson, A. D., Melino, G. & Redfern, C. P. (1997). Apoptosis of N-type neuroblastoma cells after differentiation with 9-cis-retinoic acid and subsequent washout. J Natl Cancer Inst 89: 446–452.
Lutzker, S. G. & Levine, A. J. (1996). A functionally inactive p53 protein in teratocarcinoma cells is activated by either DNA damage or cellular differentiation. Nature Med 2: 804–810.
Mangelsdorf, D. J., Thummel, C., Beato, M., Herrlich, P., Schutz, G., Umesono, K., Blumberg, B., Kastner, P., Mark, M., Chambon, P. & Evans, R. M. (1995). The nuclear receptor superfamily: the second decade. Cell 83: 835–839.
Oridate, N., Suzuki, S., Higuchi, M., Mitchell, M. F., Hong, W. K. & Lotan, R. (1997). Involvement of reactive oxygen species in N-(4-hydroxyphenyl)retinamide-induced apoptosis in cervical carcinoma cells. J Natl Cancer Inst 89: 1191–1198.
Ravaud, A., Nguyen, B. B. & Coindre, J. M. (1990). Adjuvant chemotherapy with CYVADIC in high-risk soft tissue sarcoma: a randomized prospective trial. In Adjuvant Therapy of Cancer VI, Salmon SE WB Saunders: Philadelphia 556–566.
Repa, J. J., Berg, J. A., Kaiser, M. E., Hanson, K. K., Strugnell, S. A. & ClagettDame, M. (1997). One-step immunoaffinity purification of recombinant human retinoic acid receptor γ. Protein Expres Purif 9: 319–330.
Rosenberg, S. A., Tepper, J., Glatstein, E., Costa, J., Baker, A., Brennan, M., De Moss, E. V., Seipp, C., Sindelar, W. F., Sugarbaker, P. & Wesley, R. (1982). The treatment of soft-tissue sarcoma of the extremities: prospective randomized evaluations of (1) limb-sparing surgery plus radiation therapy compared with amputation and (2) the role of adjuvant chemotherapy. Ann Surg 196: 305–315.
Rosso, R., Sertoli, M. R., Queirolo, P., Sanguineti, O., Barzacchi, M. C., Mariani, G. L., Miglio, L., Venturini, M. & Toma, S. (1992). An outpatient phase I study of a subcutaneous interleukin-2 and intramuscular α-2a-interferon combination in advanced malignancies. Ann Oncol 3: 559–563.
Ryan, J. J., Danish, R., Gottlieb, C. A. & Clarke, M. F. (1993). Cell cycle analysis of p53-induced cell death in murine erythroleukemia cells. Mol Cell Biol 13: 711–719.
Sarcoma Meta-analysis Collaboration (1997). Adjuvant chemotherapy for localised resectable soft-tissue sarcoma of adults: meta-analysis of individual data. Lancet 350: 1647–1654.
Sarkis, A. S., Bajorin, D. F., Reuter, V. E., Herr, H. W., Netto, G., Zhang, Z. F., Schultz, P. K., Cordon-Cardo, C. & Scher, H. I. (1995). Prognostic value of p53 nuclear overexpression in patients with invasive bladder cancer treated with neoadjuvant MVAV. J Clin Oncol 13: 1384–1390.
Shao, Z. M., Dawson, M. I., Li, X. S., Rishi, A. K., Sheikh, M. S., Han, Q. X., Ordonez, J. V., Shroot, B. & Fontana, J. A. (1995). p53 independent G0/G1 arrest and apoptosis induced by a novel retinoid in human breast cancer cells. Oncogene 11: 493–504.
Slichenmyer, W. J., Nelson, W. G., Slebos, R. J. & Kastan, M. B. (1993). Loss of a p53-associated G1 checkpoint does not decrease cell survival following DNA damage. Cancer Res 53: 4164–4168.
Stewart, N., Hicks, G. G., Paraskevas, F. & Mowat, M. (1995). Evidence for a second cell cycle block at G2/M by p53. Oncogene 10: 109–115.
Tallman, M. S., Andersen, J. W., Schiffer, C. A., Appelbaum, F. R., Feusner, J. H., Ogden, A., Shepherd, L., Willman, C., Bloomfield, C. D., Rowe, J. M. & Wiernik, P. H. (1997). All-trans-retinoic acid in acute promyelocytic leukemia. N Engl J Med 337: 1021–1028.
Thompson, C. B. (1995). Apoptosis in the pathogenesis and treatment of disease. Science 267: 1456–1462.
Toma, S., Isnardi, L., Raffo, P., Dastoli, G., De Francisci, E., Riccardi, L., Palumbo, R. & Bollag, W. (1997). Effects of all-trans-retinoic acid and 13-cis-retinoic acid on breast-cancer cell lines: growth inhibition and apoptosis induction. Int J Cancer 70: 619–627.
van der Leede, B. M., van den Brink, C. E. & van der Saag, P. T. (1993). Retinoic acid receptor and retinoid X receptor expression in retinoic acid-resistant human tumor cell lines. Mol Carcinog 8: 112–122.
Wu, Y. N., Gadina, M., Tao-Cheng, J. H. & Youle, R. J. (1994). Retinoic acid disrupts the Golgi apparatus and increases the cytosolic routing of specific protein toxins. J Cell Biol 125: 743–753.
Yonish-Rouach, E., Resnitzky, D., Lotem, J., Sachs, L., Kimchi, A. & Oren, M. (1991). Wild-type p53 induces apoptosis of myeloid leukaemic cells that is inhibited by interleukin-6. Nature 352: 345–347.
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Brodowicz, T., Wiltschke, C., Kandioler-Eckersberger, D. et al. Inhibition of proliferation and induction of apoptosis in soft tissue sarcoma cells by interferon-α and retinoids. Br J Cancer 80, 1350–1358 (1999). https://doi.org/10.1038/sj.bjc.6690528
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690528
Keywords
This article is cited by
-
Ethnozoological importance of Eisenia fetida and experimental validation of its anticancer activity in ascites Dalton’s lymphoma (DL) bearing mice
Future Journal of Pharmaceutical Sciences (2024)
-
Vascular endothelial growth factor acts in an autocrine manner in rhabdomyosarcoma cell lines and can be inhibited with all-trans-retinoic acid
Oncogene (2005)