Abstract
Background:
Ecological and experimental studies have suggested a relationship between Asian pickled vegetable consumption and oesophageal squamous cell carcinoma (OSCC), but the results of epidemiological studies investigating the association have been inconsistent. We conducted a meta-analysis of observational studies of this association to evaluate the existing evidence.
Methods:
We searched the PubMed, ISI-Web of Science, J-EAST, IndMed, Vip Chinese Periodical, and China National Knowledge Infrastructure databases for all studies published in English or Chinese languages. Pooled results for all studies combined and for several study subgroups were computed.
Results:
A total of 34 studies were included in this analysis. The overall random effects odds ratio (OR) and 95% confidence interval (CI) for pickled vegetable consumption was 2.08 (1.66–2.60), but the results were heterogeneous across studies. After excluding the three most influential studies, the respective numbers were 2.32 (1.92–2.81). Similar to the overall association, the majority of subgroup analyses showed a statistically significant association between consuming pickled vegetables and OSCC risk. There were only three prospective studies.
Conclusion:
Our results suggest a potential two-fold increased risk of oesophageal cancer associated with the intake of pickled vegetables. However, because the majority of data was from retrospective studies and there was a high heterogeneity in the results, further well-designed prospective studies are warranted.
Similar content being viewed by others

Main
Eating pickled vegetables has long been suspected as a cause of oesophageal squamous cell carcinoma (OSCC). Cancer registries established in the 1960s showed extremely high incidence rates of OSCC in certain areas of China, such as Linxian (The Coordinating Group for Research on Etiology of Esophageal Cancer in North China, 1975; Tran et al, 2005). Known causes of OSCC, most notably tobacco and alcohol consumption could not explain this high incidence (Kamangar et al, 2009), so other suggested causes (Yang, 1980), particularly eating pickled vegetables, were investigated.
In high-risk areas of China, pickled vegetables were eaten daily for 9–12 months a year and were an integral part of the diet in some families (Yang, 1980), traditionally prepared by keeping tightly packed moist vegetables in a jar packing for a few weeks or months, allowing fermentation and growth of fungi and yeasts (Yang, 1980; IARC Working Group, 1993) that can generate potentially carcinogenic N-nitroso compounds and mycotoxins (Yang, 1980; Cheng et al, 1981; Zhang et al, 1983). Mutagenicity and carcinogenicity of pickled vegetables have been shown in some experimental and in vitro studies (Cheng et al, 1980; Yang, 1980; Lu et al, 1981).
Although a higher risk of OSCC has been found in areas that used more pickled vegetables (Yang, 1980), case–control and cohort studies have not consistently supported a causal role. IARC Working Group (1993) concluded that there was limited evidence in humans and inadequate evidence in experimental animals for carcinogenicity of pickled vegetables. Since then, many studies have investigated the question, and in this review and meta-analysis we examine the studies of the association of pickled vegetable consumption OSCC risk.
Materials and methods
We conducted a comprehensive search of the PubMed, ISI-Web of Science, Japanese Science, Technology and Medical Literature (J-EAST), Indian Medlars Center (IndMed), Vip Chinese Periodical Database, and China National Knowledge Infrastructure databases for studies published in English or Chinese languages on oesophageal cancer risk in relation to pickled vegetables consumption upto 10 April 2009. The following terms were used to search text words in the PubMed and the ISI-Web of Science databases: (oesophageal OR oesophageal OR oesophagus OR oesophagus) AND (cancer OR carcinoma OR adenocarcinoma OR neoplasm OR neoplasia OR neoplastic) AND (pickle OR pickled OR moldy OR fermented). Combinations of the above terms were used to search keywords in the J-EAST database. For IndMed database, the first two phrases of the above terms were used. Combinations of the following terms were used for the Chinese databases:  (diet),
(diet),  (pickled food),
(pickled food),  (Suan Cai, a kind of pickled vegetable),
(Suan Cai, a kind of pickled vegetable),  (salted vegetable),
(salted vegetable),  (salted vegetable),
(salted vegetable),  (pickled vegetable),
(pickled vegetable),  (pickled vegetable),
(pickled vegetable),  (digestive tract),
(digestive tract),  (oesophagus or oesophageal),
(oesophagus or oesophageal),  (cancer or carcinoma), and
(cancer or carcinoma), and  (tumour). At least two of the authors (FI, JR, or FK) reviewed the publications in English; those in Chinese were reviewed by one author (JR).
(tumour). At least two of the authors (FI, JR, or FK) reviewed the publications in English; those in Chinese were reviewed by one author (JR).
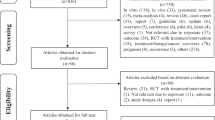
We read the abstracts of the retrieved articles; in case of any doubt, we also reviewed the full texts of those articles. Furthermore, references cited in all relevant original and review articles were searched manually. We included only studies with case–control or cohort design, the former limited to those that used control subjects without upper gastrointestinal cancers. Using these approaches, we found 55 full-text articles, among which 2 (Wang et al, 1992; Chen et al, 2003) reported the results from 3 and 2 individual studies, respectively, making the total number retrieved 58, of these 24 were excluded because: (i) no risk estimates or crude numbers of cases and controls for the exposure-of-interest were reported (Shen et al, 1999; Wang et al, 2006); (ii) the method of pickling vegetables differed from those in East and Southeast Asia (Onuk et al, 2002; Sapkota et al, 2008); (iii) results were only reported in combination with other foods (Hou et al, 1999; Lu et al, 2000; Wang et al, 2004; Xie et al, 2005; Pan et al, 2006) or in combination with other cancers (Li et al, 1989; Yu et al, 1993; Tong et al, 2001; Zhang et al, 2001; one of the three studies reported in Chen et al, 2003); and (iv) results were also reported in other publications (Chu and Ye, 1992; Chu et al, 1992a, 1992b; Guo et al, 1994; Cheng et al, 1995; Li et al, 2001; Xu et al, 2002; Li et al, 2007; Feng et al, 2008a; another one of the three studies reported in Chen et al, 2003). Finally, 34 studies remained in the meta-analysis. Although many studies reported the results for oesophageal cancer without specifying any histological subtype, as OSCC constitutes the large majority in China and other places where such pickled vegetables are highly consumed (Kamangar et al, 2006, 2009; Eslick, 2009), our findings mainly relate to OSCC.
Statistical analysis
Wherever available, we extracted details of author, place and year of publication, the actual number of pickled vegetable users as well as non-users by case status, the crude and adjusted odds ratios (OR) and 95% confidence intervals (CI), study design, and the method of selecting controls.
Owing to high heterogeneity across studies, we used random-effects models (DerSimonian–Laird method) to calculate OR and 95% CI for all studies combined. When an individual study presented both crude and adjusted ORs and 95% CIs, we used the maximally adjusted results. If results on multiple consumption categories were given, we collapsed the categories into a single measure of association using fixed-effects models. Depending on the available data, the reference groups included subjects with no or the lowest amount of pickled vegetable consumption. To reduce the influence of outlier studies, we used the command ‘metainf’ (in STATA software, version 10.0) to identify the three most dominant studies in fixed-effects estimates and repeated the analyses without those studies.
We analysed cohort (including nested case–control) separately from case–control studies. As in our previous meta-analyses (Gorouhi et al, 2008; Islami and Kamangar, 2008), we present the results for larger studies, defined as those with s.e. ⩽0.5 separately. Subgroup analyses were also carried out on studies with population-based controls. As age is a strong risk factor for OSCC, studies using age-matched controls (group- or individual-matched) were examined separately. As the association with pickled vegetables could be confounded by other factors, for example socio-economic status, we examined studies with adjusted ORs (95% CIs) separately, excluding those with adjustments only for age and sex. We also calculated summary OR (95% CI) for the subgroup that reported the numbers or proportions of cases and controls who consumed pickled vegetables, because we were more certain that the calculation of OR (95% CI) was correct in these studies.
Studies from Linxian, a high-risk area in China, showed very heterogeneous results, consumption among controls ranged from 0 to approximately 60%, and point-estimates of association varied widely on both sides of null. The observed heterogeneity may be because of the mass public health campaigns in the 1970s in Linxian and some other regions, which advised people to avoid consumption of pickled vegetables to reduce the risk of oesophageal cancer (Sakaguchi et al, 2005; Tran et al, 2005). In consequence, some subjects may have reduced their intake of pickled vegetables or under-reported their use in the survey. We therefore, compared studies from Linxian vs all others, and from Mainland China vs all others (including Hong Kong, Taiwan, Japan, and India). As these public health campaigns subsided in the 1990s, we also carried out subgroup analyses for earlier studies (initiated in 1990 or earlier) and more recent studies (initiated after 1990). We investigated whether there was any difference in the risk estimates between English and Chinese papers.
We used Begg's funnel plot to examine small study effects (Sterne et al, 2001), Begg and Mazudmar's method (Begg and Mazumdar, 1994) to calculate P-value for rank correlation and Egger's weighted regression method (Egger et al, 1997) to calculate P-value for publication bias. To examine heterogeneity, the Q-statistic (using Mantel–Haenszel weights) and the I2 statistic were calculated (Higgins et al, 2003). Throughout the paper, two-sided P-values <0.05 were considered as statistically significant.
Results
Summary characteristics of the 34 included studies are presented in Table 1; more detailed information is given in Supplementary Table 1. There were 3 prospective and 31 case–control studies; approximately half of the studies were population based, the other half clinic based. Most studies (n=29) were from Mainland China; 56% (n=19) were in Chinese.
For the overall association, the OR (95% CI) was 2.08 (1.66–2.60). The P-value associated with the Q-statistic was <0.001, and the I2-statistic was 88%, suggesting very high heterogeneity (Table 2). A forest plot and Begg's funnel plot for overall associations are shown in Figure 1, wherein the distribution of dots appeared to be non-symmetric, and P-value for bias in Egger's weighted regression method was 0.01, suggesting publication bias. However, there was no evidence for bias using Begg and Mazumdar's method (P-value for rank correlation=0.30). After excluding the three studies with the highest influence on overall results (Hu et al, 1994; Kinjo et al, 1998; Tran et al, 2005), the OR (95% CI) changed to 2.32 (1.92–2.81).
Table 2 shows the results of subgroup analyses, the majority of which showed a significant association between consumption of pickled vegetables and oesophageal cancer risk. Results from prospective studies (n=3) were insignificant (OR=1.52; 95% CI 0.82–1.63), whereas case–control studies showed highly significant results (OR=2.18; 95% CI 1.75–2.73). After excluding small studies, defined as those having a s.e. >0.5, the summary OR (95% CI) was 2.09 (1.67–2.63). Summary ORs (95% CIs) for studies with population-based controls, age-matched controls, and adjusted results were comparable to those of large studies (Table 2). Studies that reported actual data on pickled vegetable consumption among cases and controls showed OR (95% CI) of 2.55 (2.01–3.24).
Studies from Linxian (n=4), showed a marginally significant association (OR=1.51; 95% CI 0.98–2.33), whereas the summary OR (95% CI) for those conducted elsewhere was 2.17 (1.70–2.78). Studies initiated in 1990 or earlier showed a moderate association (OR=1.73; 95% CI 1.14–2.62), while those initiated after 1990 showed a more than two-fold increased risk (OR=2.28; 95% CI 1.89–2.76). The summary ORs (95% CI) for studies from China (Mainland) and elsewhere were 2.10 (1.63–2.69) and 1.96 (1.12–3.43), respectively. The summary ORs (95% CIs) for studies retrieved from English and Chinese literature were 2.10 (1.47–3.00) and 2.10 (1.65–2.67), respectively.
Discussion
We found an approximately two-fold increased risk of OSCC associated with pickled vegetable use in the overall analysis and in the majority of subgroup analyses. However, this association was insignificant in prospective studies or in studies from Linxian. There was also very high heterogeneity in the strength of associations between studies.
None of our multiple subgroup analyses substantially reduced heterogeneity, the I2-index for heterogeneity remaining above 60%. However, case–control studies, those with age-matched controls, studies that reported the prevalence of use in cases and controls, studies outside Linxian, and studies conducted after 1990, when public health campaigns against pickled vegetable use had subsided, all showed stronger associations.
Two of the three cohort studies showed no increased risk. The results of dietary cohort studies are usually more reliable than retrospective case–control studies, because the results of the former are not subject to recall bias or interviewer bias. However, in this analysis, there are reasons to dispute the summary results from the cohort studies. One of these studies (Tran et al, 2005) with a large sample size was from Linxian in a time when strong recommendations had been made against pickled vegetable use. Such use in that study was very low (close to zero), which may be due to residents having temporarily discontinued using this food item, or may have felt uncomfortable about reporting their consumption. In either case, the results would be flawed due to exposure misclassification.
After excluding the three most influential studies (Hu et al, 1994; Kinjo et al, 1998, 2003; Tran et al, 2005) the results remained highly heterogeneous. One of these studies (Tran et al, 2005) was conducted in the very high-risk regions of Linxian and, as discussed above, may have been subject to exposure misclassification. We tested for publication bias using two methods. Although the P-value from Egger's weighted regression method suggested a publication bias, but that from Begg and Mazumdar's method did not suggest such a bias. The main reason for the low P-value from Egger's method may not be publication bias, but rather due to the null results from large studies conducted in Linxian in the 1980s vs increased risks found in smaller studies from other places.
Although the above results do not prove an association, they strongly suggest that the use of pickled vegetables increases OSCC risk. This association is more likely if the studies in Linxian, at the time of mass public health campaigns or soon after, were subject to exposure misclassification. On the other hand, the majority of data come from case–control studies, which are subject to recall or interviewer bias. We therefore believe that the association needs further investigations in well-designed prospective studies, with validated questionnaires and trained interviewers.
Extracts of pickled vegetables have shown mutagenic and transforming activities as well as promoting effects in vitro (Cheng et al, 1981), inducing hyperplasia and dysplasia in oesophageal epithelium and tumours in rodents (Yang, 1980). Heavy infestation of pickled vegetables with fungi is reported from Linxian (Yang, 1980). Some common fungi can reduce nitrate to nitrite and also increase amines content of pickled vegetables (Li et al, 1986). This may facilitate the formation of N-nitroso compounds, which are strong oesophageal carcinogens in several animal models (Kamangar et al, 2009). Roussin red methyl ester, a non-alkylating nitroso compound with promoting effect in vitro, has been identified in pickled vegetables samples from Linxian in much higher concentrations than in samples from a low-incidence areas (Cheng et al, 1981; Zhang et al, 1983). Fumonisin mycotoxins have been shown to cause liver and kidney tumours in rodents (Kamangar et al, 2009). Furthermore, a synergistic interaction between mouldy food and N-nitroso compounds has been reported (Yang, 1980).
This meta-analysis has several strengths. We covered several databases, including those indexing publications in the areas where pickled vegetables are highly consumed; we searched publications in Chinese as well as in English. The meta-analysis presents results from 34 studies with a large number of cases and controls. As in any meta-analysis of observational studies, however, combining studies conducted in different populations with various qualities of design to obtain summary ORs and 95% CIs may be misleading, and summary statistics needs to be interpreted with caution. Few prospective studies have investigated pickled vegetables use and OSCC, so most data were from case–control studies, in which food intake data may be subject to recall and interviewer bias.
In summary, our results suggest a two-fold higher risk of OSCC associated with the intake of pickled vegetables. However, because most data were from retrospective studies with high heterogeneity in the results, further well-designed prospective studies with validated questionnaires are warranted.
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Begg CB, Mazumdar M (1994) Operating characteristics of a rank correlation test for publication bias. Biometrics 50: 1088–1101
Chen KL, Yin HY, Yi DT, Lan YG, Wu BZ, Wang ZS, Che XX, Fan CJ, Wu DX (1995) The association between smoking, drinking and dietary habit and the incidence of esophageal cancer in low risk area. Bull Chin Cancer 4: 7–9
Chen W, Sun XD, Fan JH, Yang ZM, Lian SY, Qiao YL (2003) The study for risk factors and changing rule of esophageal cancer in Linxian. Cancer Res Clin 15: 5–7
Chen ZY, Li YC, Zhu XP, Cai MR (2004) A case-control study on the relationship between dietary factors and esophageal cancer in Rugao. Bull Chin Cancer 13: 506–508
Chen ZY, Zhao KG, Zhou GX, Liu JB, Cai MR, Li ZM, Li YC, Peng DF (2000) A case-control study on the main risk factors of esophageal cancer in Rugao, Jiangsu Province. Cancer Res Prev Treat 27: 240–242
Cheng KK, Day NE, Duffy SW, Lam TH, Fok M, Wong J (1992) Pickled vegetables in the aetiology of oesophageal cancer in Hong Kong Chinese. Lancet 339: 1314–1318
Cheng KK, Duffy SW, Day NE, Lam TH (1995) Oesophageal cancer in never-smokers and never-drinkers. Int J Cancer 60: 820–822
Cheng SJ, Sala M, Li MH, Courtois I, Chouroulinkov I (1981) Promoting effect of Roussin's red identified in pickled vegetables from Linxian China. Carcinogenesis 2: 313–319
Cheng SJ, Sala M, Li MH, Wang MY, Pot-Deprun J, Chouroulinkov I (1980) Mutagenic, transforming and promoting effect of pickled vegetables from Linxian county, China. Carcinogenesis 1: 685–692
Chitra S, Ashok L, Anand L, Srinivasan V, Jayanthi V (2004) Risk factors for esophageal cancer in Coimbatore, southern India: a hospital-based case-control study. Indian J Gastroenterol 23: 19–21
Chu JJ, Xu YC, Ye BF, Yao FL, Hu X, Wu JN (1992a) Study on the main risk factors of esophageal cancer in Huai’an. Jiangsu Med J 18: 376–377
Chu JJ, Ye BF (1992) Ridge regression analysis of the etiological factors of esophageal cancer in Huai’an city. J Xuzhou Med Coll 12: 85–88
Chu JJ, Ye BF, Xu YC (1992b) Study on the main risk factors of esophageal cancer. J Nanjing Med Coll 12: 309–312, 317
Chu JJ, Ye BF, Xu YC, Hu X, Wu JN (1993) Case-control on the main risk factors of esophageal cancer in the residents of Huaian city. Tumor 13: 49–52
Egger M, Davey SG, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315: 629–634
Eslick GD (2009) Epidemiology of esophageal cancer. Gastroenterol Clin North Am 38: 17–25, vii
Feng XX, Duan PF, Wang LB, Zhang JB, Lu ZX (2008a) Study on the relationship between polymorphisms of XPA gene and susceptibility of esophageal cancer. Zhonghua Liu Xing Bing Xue Za Zhi 29: 930–933
Feng XX, Li ZF, Wang LB, Zhang JB, Lu ZX (2008b) Study on the relationship between polymorphisms of NQO1 gene and susceptibility to esophageal cancer. Chin J Dis Control Prev 12: 112–114
Gao CM, Haruhiko S, Toshiro T, Wu JZ, Li ZY, Ding JH, Liu YT, Li SP, Su P, Hu X, Xu TL, Kazuo T (2001) hOGG1 genotypes, life style and the risk of esophageal and stomach cancers. Bull Chin Cancer 10: 500–502
Gao CM, Takezaki T, Ding JH, Li MS, Tajima K (1999) Protective effect of allium vegetables against both esophageal and stomach cancer: a simultaneous case-referent study of a high-epidemic area in Jiangsu Province, China. Jpn J Cancer Res 90: 614–621
Gorouhi F, Islami F, Bahrami H, Kamangar F (2008) Tumour-necrosis factor-A polymorphisms and gastric cancer risk: a meta-analysis. Br J Cancer 98: 1443–1451
Guo W, Blot WJ, Li JY, Taylor PR, Liu BQ, Wang W, Wu YP, Zheng W, Dawsey SM, Li B (1994) A nested case-control study of oesophageal and stomach cancers in the Linxian nutrition intervention trial. Int J Epidemiol 23: 444–450
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327: 557–560
Hou J, Qiao CY, Meng FS, Zhang GS, He YT, Chen ZF, Liu JB, Song GH, Li SS, Hao SM, Ji HX (1999) A case-control study on risk factor of esophageal cancer in Cixian County of Hebei Province. China Cancer 8: 252–255
Hu J, Nyren O, Wolk A, Bergstrom R, Yuen J, Adami HO, Guo L, Li H, Huang G, Xu X (1994) Risk factors for oesophageal cancer in northeast China. Int J Cancer 57: 38–46
Huang GD, Zheng ZJ, Liu Q, Liu JH (2000) A case-control study on risk factors of esophageal cancer in Hakka in Meizhou city. Guangdong Med J 21: 882–883
Hung HC, Huang MC, Lee JM, Wu DC, Hsu HK, Wu MT (2004) Association between diet and esophageal cancer in Taiwan. J Gastroenterol Hepatol 19: 632–637
IARC Working Group (1993) Some Naturally Occurring Substances: Food Items and Constituents, Heterocyclic Aromatic Amines and Mycotoxins. IARC Monographs on Evaluation of Carcinogenic Risks to Humans, Vol 56, pp 83–113. IARC Press: Lyon
Islami F, Kamangar F (2008) Helicobacter pylori and esophageal cancer risk: a meta-analysis. Cancer Prev Res (Phila Pa) 1: 329–338
Ji YM (1999) A case-control study on pickled vegetable and esophageal cancer. J Shanxi Med Univ 30: 208–209
Kamangar F, Chow WH, Abnet CC, Dawsey SM (2009) Environmental causes of esophageal cancer. Gastroenterol Clin North Am 38: 27–57, vii
Kamangar F, Dores GM, Anderson WF (2006) Patterns of cancer incidence, mortality, and prevalence across five continents: defining priorities to reduce cancer disparities in different geographic regions of the world. J Clin Oncol 24: 2137–2150
Kinjo Y, Cui Y, Akiba S, Watanabe S, Yamaguchi N, Sobue T, Mizuno S, Beral V (1998) Mortality risks of oesophageal cancer associated with hot tea, alcohol, tobacco and diet in Japan. J Epidemiol 8: 235–243
Li JY, Ershow AG, Chen ZJ, Wacholder S, Li GY, Guo W, Li B, Blot WJ (1989) A case-control study of cancer of the esophagus and gastric cardia in Linxian. Int J Cancer 43: 755–761
Li K, Yu P (2003) Food groups and risk of esophageal cancer in Chaoshan region of China: a high-risk area of esophageal cancer. Cancer Invest 21: 237–240
Li K, Yu P, Zhang ZX, Huang SS, Huang G, Ma XH (2001) Food components and risk of esophageal cancer in chaoshan region of china, a high-risk area of esophageal cancer. Chin J Cancer 20: 160–163
Li MH, Ji C, Cheng SJ (1986) Occurrence of nitroso compounds in fungi-contaminated foods: a review. Nutr Cancer 8: 63–69
Li ZF, Feng XX, Wei ZB (2007) A logistic regression analysis of the risk factors of esophageal carcinoma in Changzhi area. J Changzhi Med Coll 21: 179–182
Liu XM, Wang QS, Zhang YL, Meng ZH (2001a) Etiological fraction and interactions analysis of risk factors in male esophageal cancer in Tianjin. Mod Prev Med 28: 257–259
Liu XM, Wang QS, Zhang YL, Meng ZH, Miao RJ (2001b) A case-control study on the association between esophageal cancer and dietary factors. J Tianjin Med Univ 7: 154–156
Lu JB, Lian SY, Sun XB, Zhang ZX, Dai DX, Li BY, Cheng LP, Wei JR, Duan WJ (2000) A case-control study on the risk factors of esophageal cancer in Linzhou. Chin J Epidemiol 21: 434–436
Lu SH, Camus AM, Tomatis L, Bartsch H (1981) Mutagenicity of extracts of pickled vegetables collected in Linhsien County, a high-incidence area for esophageal cancer in Northern China. J Natl Cancer Inst 66: 33–36
Onuk MD, Oztopuz A, Memik F (2002) Risk factors for esophageal cancer in eastern Anatolia. Hepatogastroenterology 49: 1290–1292
Pan EC, Yang WZ, Hu X, Kai HT, Wang Y (2006) A case-control study on risk factors of neoplasm of esophagus in Chuzhou district of Huai’an, Jiangsu Province. Chin J Dis Control Prev 10: 114–116
Peng XE, Zhou ZJ, Shi XS, Li GL, Pan PC, Chen ZL, Li WM, Wu QB (2005) Case-control study on risk factors of esophageal cancer in Anxi county. Chin J Public Health 21: 10–12
Phukan RK, Chetia CK, Ali MS, Mahanta J (2001) Role of dietary habits in the development of esophageal cancer in Assam, the north-eastern region of India. Nutr Cancer 39: 204–209
Qi GY, Su SC, You CF, Chen SW, Song Y (2001) A case-control study on the influential factors of esophageal cancer. Chin J Prev Control Chronic non-Commun Dis 9: 15–16
Ren AG, Han XW (1991) Case-control study on dietary and esophageal cancer. Chin J Epidemiol 12: 200–204
Sakaguchi S, Yokokawa Y, Hou J, Zhang XL, Li XP, Li SS, Li XX, Zhu DC, Kamijima M, Yamanoshita O, Nakajima T (2005) Environmental exposure and p53 mutations in esophageal cancer patients in areas of low and high incidence of esophageal cancer in China. Tohoku J Exp Med 207: 313–324
Sapkota A, Hsu CC, Zaridze D, Shangina O, Szeszenia-Dabrowska N, Mates D, Fabianova E, Rudnai P, Janout V, Holcatova I, Brennan P, Boffetta P, Hashibe M (2008) Dietary risk factors for squamous cell carcinoma of the upper aerodigestive tract in central and eastern Europe. Cancer Causes Control 19: 1161–1170
Shen HB, Xu YC, Shen J, Niu JY, Ye BF, Zhou L, Hu X, Wu JN, Chen J (1997) Etiological study on pickled vegetable in Huaian, a high-risk area of esophageal cancer. ACTA Acad Med Nanjing 17: 119–122
Shen YP, Gao YT, Dai Q, Hu X, Xu TL, Xiang YB, Tang ZL, Li WL (1999) A case-control study of esophageal cancer in the people of Huai’an City. Chin J Public Health 15: 961–962
Sterne JA, Egger M, Smith GD (2001) Systematic reviews in health care: investigating and dealing with publication and other biases in meta-analysis. BMJ 323: 101–105
Takezaki T, Gao CM, Wu JZ, Ding JH, Liu YT, Zhang Y, Li SP, Su P, Liu TK, Tajima K (2001) Dietary protective and risk factors for esophageal and stomach cancers in a low-epidemic area for stomach cancer in Jiangsu Province, China: comparison with those in a high-epidemic area. Jpn J Cancer Res 92: 1157–1165
Tao X, Zhu H, Matanoski GM (1999) Mutagenic drinking water and risk of male esophageal cancer: a population-based case-control study. Am J Epidemiol 150: 443–452
The Coordinating Group for Research on Etiology of Esophageal Cancer in North China (1975) The epidemiology and etiology of esophageal cancer in north China. A preliminary report. Chin Med J (Engl) 1: 167–183
Tong WJ, Li J, Yue GQ, Liu XW (2001) Case-control study on risk factors of 120 cases of esophageal cancer and gastric cancer. Chin J Public Health 17: 1093–1094
Tran GD, Sun XD, Abnet CC, Fan JH, Dawsey SM, Dong ZW, Mark SD, Qiao YL, Taylor PR (2005) Prospective study of risk factors for esophageal and gastric cancers in the Linxian general population trial cohort in China. Int J Cancer 113: 456–463
Wang MR, Guo CH, Li MS, Yu GP, Yin XS, Cui GP, Geng CY, Yang JL (1999) A case-control study on the dietary risk factors of upper digestive tract cancer. Chin J Epidemiol 20: 95–97
Wang Y, Luo XP, Wang Q, Xu HQ, Gong YX (1993) A conditional logistic regression analysis on the etiology of esophageal cancer in Chuanbei area. Chin J Cancer Prev Treat 1: 1–5
Wang Y, Pan EC, Hu X, Kai HT, Yang WZ, Li JY, Zhou W (2004) A study on establishing the screening model for neoplasm of esophagus using the relevant risk factors. Jiangsu Prev Med 15: 23–25
Wang YP, Han XY, Su W, Wang YL, Zhu YW, Sasaba T, Nakachi K, Hoshiyama Y, Tagashira Y (1992) Esophageal cancer in Shanxi Province, People's Republic of China: a case-control study in high and moderate risk areas. Cancer Causes Control 3: 107–113
Wang Z, Tang L, Sun G, Tang Y, Xie Y, Wang S, Hu X, Gao W, Cox SB, Wang JS (2006) Etiological study of esophageal squamous cell carcinoma in an endemic region: a population-based case control study in Huaian, China. BMC Cancer 6: 287
Xibib S, Meilan H, Moller H, Evans HS, Dixin D, Wenjie D, Jianbang L (2003) Risk factors for oesophageal cancer in Linzhou, China: a case-control study. Asian Pac J Cancer Prev 4: 119–124
Xie Y, Sun GJ, Hu X, Wang SK, Kai HT, Cui YS (2005) A case-control study on the association between diet and behavior factors and esophageal cancer in the residents of Chuzhou, Huai’an. J Hyg Res 34: 479–480
Xu DZ, Li LS, Zhang XL, Wang B, Sun CS, Yan MX, Wang Y, Yang WX, Chang GS (2002) A case-control study on the etiology of esophageal cancer in Linzhou city. Chin J Epidemiol 23: 320–321
Yang CS (1980) Research on esophageal cancer in China: a review. Cancer Res 40: 2633–2644
Yang CX, Wang HY, Wang ZM, Du HZ, Tao DM, Mu XY, Chen HG, Lei Y, Matsuo K, Tajima K (2005) Risk factors for esophageal cancer: a case-control study in South-western China. Asian Pac J Cancer Prev 6: 48–53
Yu Y, Taylor PR, Li JY, Dawsey SM, Wang GQ, Guo WD, Wang W, Liu BQ, Blot WJ, Shen Q (1993) Retrospective cohort study of risk-factors for esophageal cancer in Linxian, People's Republic of China. Cancer Causes Control 4: 195–202
Zhang GS, He YT, Hou J (2000) A case control study on risk factor of esophageal cancer in Cixian county. Sichuan J Cancer Control 13: 65–67
Zhang WD, An FS, Lin HS, Yu HF, Chen SH, Wang SY (2001) A case-control study on the risk factors of esophageal cancer in Jieyang city of Guangdong in China. Chin J Epidemiol 22: 442–445
Zhang WX, Xu MS, Wang GH, Wang MY (1983) Quantitative analysis of Roussin red methyl ester in pickled vegetables. Cancer Res 43: 339–341
Zhao DL, Yang YD, Chen MH, Hu MX, Zhang QH, Guo XL, Li HQ, Jin SK (2003) Study on risk factors of esophageal cancer in Feicheng city. China J Cancer Prev Treat 10: 27–30
Acknowledgements
This study was supported in part by the Intramural Research Program of the National Cancer Institute, National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on British Journal of Cancer website (http://www.nature.com/bjc)
Supplementary information
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Islami, F., Ren, JS., Taylor, P. et al. Pickled vegetables and the risk of oesophageal cancer: a meta-analysis. Br J Cancer 101, 1641–1647 (2009). https://doi.org/10.1038/sj.bjc.6605372
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6605372
Keywords
This article is cited by
-
Mortality burden and future projections of major risk factors for esophageal cancer in China from 1990 to 2019
General Thoracic and Cardiovascular Surgery (2024)
-
Comparing spatial patterns of 11 common cancers in Mainland China
BMC Public Health (2022)
-
Non-Acid Fluid Exposure and Esophageal Squamous Cell Carcinoma
Digestive Diseases and Sciences (2022)
-
Burden, trends, and risk factors of esophageal cancer in China from 1990 to 2017: an up-to-date overview and comparison with those in Japan and South Korea
Journal of Hematology & Oncology (2020)
-
Primärprävention von Plattenepithelkarzinomen des Ösophagus und des Anus
Der Onkologe (2020)