Abstract
We investigated childhood brain tumours by histological subtype in relation to prenatal X-ray among all children, less than 15 years of age, born in Sweden between 1975 and 1984. For each case, one control was randomly selected from the Medical Birth Register, and exposure data on prenatal X-ray were extracted blindly from antenatal medical records. Additional information on maternal reproductive history was obtained from the Medical Birth Register. We found no overall increased risk for childhood brain tumour after prenatal abdominal X-ray exposure (adjusted odds ratio (OR): 1.02, 95% confidence interval (CI): 0.64–1.62); primitive neuroectodermal tumours had the highest risk estimate (OR: 1.88, 95% CI: 0.92–3.83).
Similar content being viewed by others
Main
From 1975 to 2002, the incidence rate of childhood brain tumours (CBTs) in the United States increased from 2.3 to 3.5 cases per 100 000 children (NCI, 2005). A step increase was noted in 1984–1985, for which suggested reasons were better diagnostic procedures with MRI and changes in the histological classification (Smith et al, 1998). However, in a Swedish study between 1973 and 1992, the increase was around 2.6% annually, with no time clustering (Hjalmars et al, 1999). It is still unclear, therefore, whether the observed trend is a true increase or an effect of better diagnostic procedures.
Few risk factors are considered as established: rare genetic disorders and high-dose ionising radiation for cancer therapy and tinea capitis (Ron et al, 1988; Bunin, 2000; Baldwin and Preston-Martin, 2004). Radiological examinations during pregnancy have in some studies been associated with a slightly increased risk of CBT in offspring (Bithell and Stewart, 1975; Rodvall et al, 1990; Shu et al, 1994). However, meta-analyses have found no clear associations (Gurney et al., 1999; Bunin, 2000; Linet et al, 2003).
During recent years, more focus has been on risk factors for the subtypes of CBT, since their cellular origins as well as geographic distribution differ. The few previously existing studies on prenatal X-ray and subtypes of CBT are based on retrospectively collected information and exposure frequency was low (Bunin et al, 1994; Schuz et al, 2001).
The aim with this study was to assess any association between prenatal exposure to diagnostic X-ray and primary CBT in a population-based case–control study, including subanalyses according to histological subtype. To avoid selection and recall bias, we performed a nation-wide study, where information on X-ray had been prospectively recorded in antenatal records.
Materials and methods
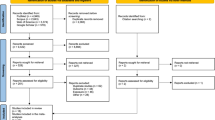
All children born between 1975 and 1984 in Sweden were eligible for this study. We used the nation-wide Swedish Cancer Register to identify children with brain tumours and for histological classification. The Cancer Register uses the 7th revision of the International Classification of Diseases (ICD-7) to facilitate comparison over time. Cases were children less than 15 years of age, diagnosed with brain tumour (ICD-7, code 193). The histological subtypes are defined through the three-digit pathohistological diagnosis code assigned by a pathologist. Controls were randomly selected children from the Medical Birth Register and frequency matched to cases by gender and year of birth. Controls had to be alive and residents in Sweden until the age of 15 years, which was ensured by using information from the Cause of Death Register and the Register of Population and Population Changes.
Medical information was abstracted from antenatal records, kept at local delivery archives and completed with data from the Medical Birth Register. Individual record linkage between registers and antenatal records was made possible by the Personal Identification Number (PIN), which is assigned to each Swedish resident and included in registers and medical records.
Sources of data
The Medical Birth Register contains prospectively collected information on more than 99% of all births in Sweden since 1973 (Cnattingius et al, 1990). The information is provided through antenatal, obstetrical and neonatal records, which are filled in by midwives and physicians. The register includes data on maternal demographics, reproductive history and complications during pregnancy, delivery and the neonatal period. The register contains no data on X-ray examinations.
The Cancer Register was founded in 1958 and contains information about clinical and histological diagnosis and date and place of living at diagnosis. It is updated annually and has reliable information on more than 97% of all patients with cancer (Mattsson, 1977).
The Cause of Death Register contains data on dates and causes of death for Swedish citizens since 1961. The coverage is more than 99.5% and data are updated annually.
The antenatal records are kept in the hospital archives and include information on X-ray and ultrasound examinations and complement the Medical Birth Register in smoking habits, pregnancy symptoms and diagnoses.
Study sample
A power calculation was performed in the planning phase of the study. By including 600 cases and an equal number of controls, and assuming a power of 80%, a two-sided 5% significance level and an abdominal X-ray exposure frequency of 10%, we should be able to detect an odds ratio (OR) of at least 1.7. In the Cancer Register, we identified 601 children born between 1975 and 1984 with a diagnosis of brain tumour before the age of 15 years. For 62 of the 601 cases, the PINs were incomplete or information on hospital of birth was missing, which made it impossible to identify their antenatal records. Of the remaining 539 cases, we were able to retrieve antenatal records for 512 (95%). For the 539 control subjects, we found 524 records (97%).
Data collection and data management
Exposure data were extracted blindly with regard to case or control status from both antenatal records and registers. All data were assessed by one of the authors (KS) before transfer to the database. From the antenatal records, we included information on pregnancy complications and other current diseases and data on X-ray exposure. From the Medical Birth Register, we retrieved information on maternal age and reproductive history, gestational age at birth, birth weight and mode of delivery. Information concerning smoking habits was collected from the Medical Birth Register (data included from 1982) and completed by data from the antenatal records.
Radiological examinations were divided into abdominal and non-abdominal examinations. Abdominal examinations included pelvimetry, X-ray for foetal position and others. Risk for CBT was evaluated for all types of brain tumours combined and according to subtype. Each diagnosis was classified into one of the following subtypes: low-grade astrocytoma (astrocytoma grades I–II, benign opticusglioma), high-grade astrocytoma (astrocytoma grades III–IV), primitive neuroectodermal tumour (PNET) defined as category IC–D in the modified WHO classification by Rorke et al (1985), ependymoma (Rorke IA3, IA4), germ cell tumour (Rorke VI, VIId) or others (rare and not completely specified tumours).
We included information on numbers of X-ray examinations and the gestational week when they were performed. Gestational age was divided into trimesters: the first (gestational weeks 2–14), second (weeks 15–28), and third (weeks 29–45). Gestational age was calculated from data on last menstrual period, which was the standard procedure in Sweden at this time (Hogberg and Larsson, 1997). Birth weight and head circumference are presented as small, appropriate or large for gestational length, according to the gender-specific Swedish reference curves for birth weight and head circumference for gestational age (Niklasson et al, 1991).
Statistical analyses
Logistic regression was performed to evaluate the association between prenatal exposure to diagnostic X-ray and incidence of CBT. Estimates of ORs and 95% confidence intervals (95% CIs) were calculated. In advance, we identified potential confounding factors that could interfere with both exposure and outcome, and for which it was possible to obtain information either from the registers or the antenatal records. The following confounders were included in the adjusted analyses: maternal age at birth, parity, multiple birth, mother's country of birth (Nordic country (Sweden, Norway, Denmark, Finland and Iceland) or non-Nordic country), hypertension during pregnancy, mode of delivery, breech position, gestational age at birth, birth weight, head circumference at birth and level of hospital (primary, secondary or tertiary level).
The statistical analyses were conducted with the SAS 9.1 software package.
The study was approved by the Ethics Committees at Karolinska Institutet and Uppsala University.
Results
In children with CBT, 50.4% were boys and 49.6% girls. As we matched for gender, the distribution between sexes among controls was similar. There were no differences in maternal smoking habits during pregnancy between cases and controls (36 vs 37% smokers, respectively). However, the information on smoking habits was missing for 36.5% of the sample. Further maternal and foetal characteristics are presented in Table 1. Among children with CBT, it was more common to be the first-born child (P=0.01) and to be born at a primary or secondary level hospital (P=0.04) than for controls. There were no other significant differences in maternal and neonatal characteristics between cases and controls (Table 1).
Overall, 21.1% (n=108) of the mothers to children diagnosed with CBT were exposed to X-ray during pregnancy compared to 21.2% (n=111) of mothers to the children randomly selected as controls. For abdominal X-ray, the exposure frequency was 10.7% (n=55) for cases and 9.4% (n=48) for controls. Beside pelvimetry and X-ray for foetal position, abdominal X-ray included three scintigraphy of placenta, four scintigraphy of kidney, four plain abdominal X-rays and one colon examination. For both cases and controls, more than 96% of the abdominal examinations were performed in the last trimester. Non-abdominal examinations included 107 pulmonary and one each of skeletal, sinus and dental X-ray. Only three individuals in each group were exposed to X-ray more than once during the pregnancy.
Table 2 presents the distribution of CBT subtypes and age at diagnosis. Median age for diagnosis for all CBTs was 8 years. For subtypes, ependymomas had the lowest median age at diagnosis (4 years) and high-grade astrocytomas had the highest (9 years).
In Table 3, the ORs for prenatal X-ray are presented by tumour subtype. Since there were no differences between cases and controls in frequency or timing of exposure, analyses are restricted to children to mothers exposed to prenatal X-ray, regardless of numbers of examinations or time of exposure. Being prenatally exposed to abdominal X-ray was not associated with an increased overall risk of brain tumours compared with being unexposed (adjusted OR: 1.02, 95% CI: 0.64–1.62). When stratifying according to histological subgroups, we found that PNET had the highest risk estimates (adjusted OR: 1.88, CI: 0.92–3.83). Astrocytoma low and high grade had no increased ORs (adjusted OR: 0.72, CI: 0.36–1.42 and adjusted OR: 1.06, CI: 0.39–2.86, respectively). As there were only 44 cases with ependymoma, it was not possible to perform multivariate analyses to adjust for possible confounders, and only crude risk estimates are presented. For similar reasons, germ cell tumours (n=17) were included in all cancers and not analysed separately.
Discussion
Prenatal X-ray exposure does not seem to increase the overall risk of CBT. However, for the subgroup of PNET, we found an increased OR of 1.88 (95% CI: 0.92–3.83). PNETs are found among the youngest children and is classified as an embryonic tumour. Medulloblastoma is the most common tumour in the PNET group, has a peak age of occurrence of 7 years of age and is located in the cerebellum. The other, and more rare tumours in the PNET group, may occur in the neonatal period (in rare cases congenitally), have a peak age at diagnosis of about 5 years of age and are mainly supratentorial (Sarkar et al, 2005).
It has been suggested that each cell type has a ‘window of vulnerability’ during which neoplastic transformation may occur (VandenBerg, 2001). Consequently, gestational age at exposure to X-rays might be of importance when evaluating risks for CBT. Since 96% of the abdominal X-ray examinations in this study were performed in the third trimester, it was not possible to evaluate the impact of time of exposure. However, the human brain grows and develops during the whole foetal period and there is currently no evidence that foetuses should be more sensitive to carcinogenic factors in the early gestational period.
In the present study, mean foetal absorbed radiation dose was hard to estimate since the X-ray examinations were not standardised and performed at several hospitals over a 10-year period. Foetal absorbed dose for an abdominal X-ray examination has been reported to vary between 0.1 mGy for a pelvimetry with one anterior–posterior and one lateral film (Axelsson and Ohlsen, 1979) to more than 4 mGy (Osei and Faulkner, 1999; ICRP, 2000). In dosimeter studies, radiation doses have been found to vary by a factor of 30 or more for the same type of examination depending on variations in radiological techniques, choice of film, etc, (Badr et al, 1997; ICRP, 2000). It is reported that prenatal exposure to a radiation dose of over 10 mGy may increase the risk of childhood cancer (Doll and Wakeford, 1997). Furthermore, it is suggested that there is no ‘safe’ threshold limit but instead a linear relationship between exposed dose and risk increase (Doll and Wakeford, 1997; National Research Council, 2006). Risk increases can be expressed as excess relative risk (ERR), and from previous studies of prenatal X-ray exposure, it is estimated that the ERR for childhood cancer for an absorbed dosage of 10 mGy is approximately 0.5 (Wakeford and Little, 2003). Although there are uncertainties concerning such risk estimates, the low radiation doses in the present study suggest that an ERR of more than 0.5 would not be expected.
The study had sufficient power to detect an OR of at least 1.7 for the overall risk of CBT. However, for the analyses stratified by histological subtypes, a larger population would probably have been needed to find statistically significant increased risks. For the subgroup PNET, which included 105 cases, we had a power of 52% to detect an OR of 1.9 at the 5% significance level. Since there are approximately 10–15 new cases of PNET among Swedish children annually, several more years had to be included to reach sufficient power. As the Medical Birth Register was founded in 1973, 2 years before the start of this study, this register could not have been used to reach the warranted number of cases with PNET, which would be needed to increase power. A study large enough to have sufficient statistical power for the subanalyses might be performed by pooling data from the Nordic countries all of which have population-based national registries and similar health-care systems.
The strengths of this study include the population-based design, the large number of cases of CBTs, the prospective and blinded data collection and the small number of missed cases. We were forced to exclude 5% of the cases and 3% of the controls, as we could not trace their antenatal records. Since the antenatal records are filled in and filled before any of the included cases were diagnosed with CBT, the exclusions should not have affected the risk estimates.
In two previous case–control studies on CBT subtypes, no increased risk after prenatal X-ray could be detected, neither for all CBTs nor for PNET specifically (Bunin et al, 1994; Schuz et al, 2001). In these studies, retrospective interviews on exposure data were used and the risk of recall bias could not be excluded. In both studies, less than 5% of the study population were exposed to prenatal X-ray, whereas we had an exposure rate of 21%. The present study is, to our knowledge, the first study on X-ray and CBT subtypes with prospectively collected information from antenatal records. The antenatal records in Sweden are standardised and are considered of high quality (Cnattingius et al, 1990).
Potential weaknesses are that some X-ray examinations may not have been noted in the records. However, we assume that such negligence was rare and we do not suspect that any possible misclassification of exposure would be different between cases and controls. Although we have performed multivariate analyses and adjusted for potential confounding factors, some other conditions that vary with both exposure and outcome could possibly have affected the results. Maternal smoking has, in some studies, been associated with a small increase in risk of CBT for the child (Brooks et al, 2004). However, as smoking was not included in the Medical Birth Register until 1982 and earlier data from the antenatal records were incomplete, information on smoking was missing for 37% of the study population. Therefore, it was not possible to include smoking in the adjusted analyses. On the other hand, for the 63% for whom we had information on maternal smoking; the distribution between smokers and non-smokers was similar between cases and controls. Furthermore, low birth weight is closely correlated to maternal smoking and adjustments for birth weight had only minor effects on the results.
Ultrasound has, during the last two decades, become the examination of choice for determining foetal status (position, multiple births, etc), and antenatal pelvimetry is rarely indicated. Nevertheless, abdominal radiation still occurs, for instance in CT scan on maternal indications. Risks with foetal diagnostic radiation could probably be extrapolated to the early childhood period, since the brain still develops and grows rapidly during the first years of life.
Our most important finding might not be the clinical implication, but in relation to the theory that prenatal or neonatal radiation might affect the various neural cell types differently. PNET are presumed to arise from undifferentiated neural stem cells and little is known regarding the molecular genetic events initiating carcinogenesis (Biegel, 1997). An interesting hypothesis would be that neural stem cells could be extra sensitive during their differentiation and that DNA damage caused by radiation could initiate malignant transformation, leading to the development of embryonic CNS tumours such as PNET. If an increased risk of PNET after radiation exposure is confirmed in future studies, this hypothesis could be tested further in laboratory or animal studies, as part of the complex work of mapping the pathogenesis of childhood brain cancer.
In conclusion, we found no increased risk for CBT after prenatal X-ray exposure when analysed as one group. When analysing according to subgroups, the embryonic tumours defined as PNET showed the highest risk estimates.
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Axelsson B, Ohlsen H (1979) Radiation doses in low-dose pelvimetry using rare-earth screens. Acta Radiol Oncol Radiat Phys Biol 18: 470–480
Badr I, Thomas SM, Cotterill AD, Pettett A, Oduko JM, Fitzgerald M, Adam EJ (1997) X-ray pelvimetry–which is the best technique? Clin Radiol 52: 136–141
Baldwin RT, Preston-Martin S (2004) Epidemiology of brain tumors in childhood – a review. Toxicol Appl Pharmacol 199: 118–131
Biegel JA (1997) Genetics of pediatric central nervous system tumors. J Pediatr Hematol Oncol 19: 492–501
Bithell JF, Stewart AM (1975) Pre-natal irradiation and childhood malignancy: a review of British data from the Oxford Survey. Br J Cancer 31: 271–287
Brooks DR, Mucci LA, Hatch EE, Cnattingius S (2004) Maternal smoking during pregnancy and risk of brain tumors in the offspring. A prospective study of 1.4 million Swedish births. Cancer Causes Control 15: 997–1005
Bunin G (2000) What causes childhood brain tumors? Limited knowledge, many clues. Pediatr Neurosurg 32: 321–326
Bunin G, Buckley J, Boesel C, Rorke L, Meadows A (1994) Risk factors for astrocytic glioma and primitive neuroectodermal tumor of the brain in young children: a report from the Children's Cancer Group. Cancer Epidemiol Biomarkers Prev 3: 197–204
Cnattingius S, Ericson A, Gunnarskog J, Kallen B (1990) A quality study of a medical birth registry. Scand J Soc Med 18: 143–148
Doll R, Wakeford R (1997) Risk of childhood cancer from fetal irradiation. Br J Radiol 70: 130–139
Gurney JG, Smith MA, Bunin GR (1999) CNS and miscellaneous intracranial and intraspinal neoplasms ICCC III, p 60–61. In SEER Pediatric Monography, http://seer.cancer.gov/ National Cancer Institute, Bethesda, MD, USA
Hjalmars U, Kulldorff M, Wahlqvist Y, Lannering B (1999) Increased incidence rates but no space–time clustering of childhood astrocytoma in Sweden, 1973–1992: a population-based study of pediatric brain tumors. Cancer 85: 2077–2090
Hogberg U, Larsson N (1997) Early dating by ultrasound and perinatal outcome. A cohort study. Acta Obstet Gynecol Scand 76: 907–912
ICRP (2000) Pregnancy and medical radiation. In Annals of the ICRP, Valentin J (ed), p 18. Stockholm, Sweden: The International Commission on Radiological Protection
Linet MS, Wacholder S, Zahm SH (2003) Interpreting epidemiologic research: lessons from studies of childhood cancer. Pediatrics 112: 218–232
Mattsson B (1977) The completeness of registration in the Swedish Cancer Register. Statistical Reports HS1977, Report no. 15 Stockholm: National Board of Health and Welfare
National Research Council (2006) Health Risks from Exposure to Low Levels of Ionizing Radiation: BEIR VII-Phase 2. Public summary, p10. National Academy of Sciences, Washington, DC
NCI (2005) NCI Research on Childhood Cancers, Vol 2007. www.cancer.gov
Niklasson A, Ericson A, Fryer JG, Karlberg J, Lawrence C, Karlberg P (1991) An update of the Swedish reference standards for weight, length and head circumference at birth for given gestational age (1977–1981). Acta Paediatr Scand 80: 756–762
Osei E, Faulkner K (1999) Fetal doses from radiological examinations. Br J Radiol 72: 773–780
Rodvall Y, Pershagen G, Hrubec Z, Ahlbom A, Pedersen NL (1990) Prenatal X-ray exposure and childhood cancer in Swedish twins. Int J Cancer 46: 362–365
Ron E, Modan B, Boice Jr JD, Alfandary E, Stovall M, Chetrit A, Katz L (1988) Tumors of the brain and nervous system after radiotherapy in childhood. N Engl J Med 319: 1033–1039
Rorke LB, Gilles FH, Davis RL, Becker LE (1985) Revision of the World Health Organization classification of brain tumors for childhood brain tumors. Cancer 56: 1869–1886
Sarkar C, Deb P, Sharma MC (2005) Recent advances in embryonal tumours of the central nervous system. Childs Nerv Syst 21: 272–293
Schuz J, Kaletsch U, Kaatsch P, Meinert R, Michaelis J (2001) Risk factors for pediatric tumors of the central nervous system: results from a German population-based case–control study. Med Pediatr Oncol 36: 274–282
Shu XO, Jin F, Linet MS, Zheng W, Clemens J, Mills J, Gao YT (1994) Diagnostic X-ray and ultrasound exposure and risk of childhood cancer. Br J Cancer 70: 531–536
Smith MA, Freidlin B, Ries LA, Simon R (1998) Trends in reported incidence of primary malignant brain tumors in children in the United States. J Natl Cancer Ins 90: 1269–1277
VandenBerg S (2001) The developing brain and cellular targets for neoplastic transformation. In Brain Tumors: An Encyclopedic Approach, Kaye AH, Laws Jr ER (eds), Vol 2001. London: Churchill Livingstone, pp 9–10
Wakeford R, Little MP (2003) Risk coefficients for childhood cancer after intrauterine irradiation: a review. Int J Radiat Biol 79: 293–309
Acknowledgements
This study was funded by grants from The Swedish Council for Working Life and Social Research (project 2002-0421), the County Council of Uppsala (Uppsala Läns Landsting) 90543/10894 and Uppsala University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Stålberg, K., Haglund, B., Axelsson, O. et al. Prenatal X-ray exposure and childhood brain tumours: a population-based case–control study on tumour subtypes. Br J Cancer 97, 1583–1587 (2007). https://doi.org/10.1038/sj.bjc.6604046
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6604046
Keywords
This article is cited by
-
Childhood and parental diagnostic radiological procedures and risk of childhood brain tumors
Cancer Causes & Control (2014)
-
Maternal exposure to medical radiation and Wilms tumor in the offspring: a report from the Children’s Oncology Group
Cancer Causes & Control (2009)
-
Prenatal ultrasound and the risk of childhood brain tumour and its subtypes
British Journal of Cancer (2008)